TRIPLE ONLY: Alcohols
1/17
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
18 Terms
What are alcohols?
A homologous series of compounds which all contain an —OH functional group covalently bonded to a hydrocarbon chain

Describe the ways to oxidise ethanol + their products
Complete combustion: burning in air or oxygen to form CO2 and H2O
Microbial oxidation: reaction with oxygen in the air to form ethanoic acid (vinegar) with the help of microorganisms such as bacteria or yeast
Heating (in water bath) with potassium dichromate(IV) in dilute sulfuric acid to form ethanoic acid
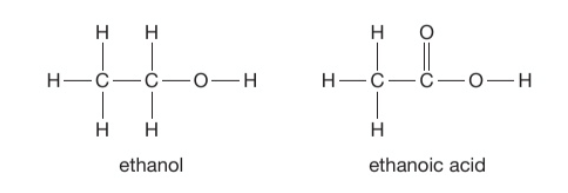
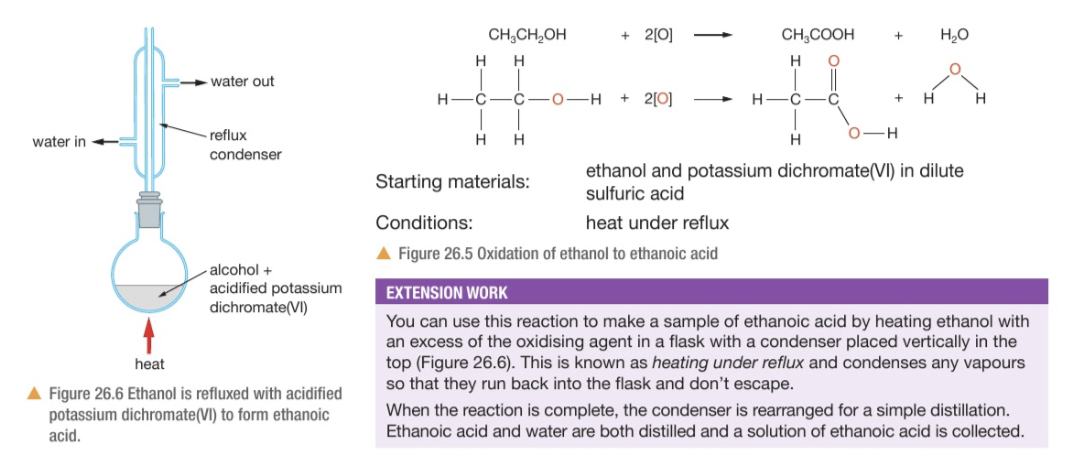
The most common way of oxidising ethanol is heating with potassium dichromate(IV) in dilute sulfuric acid to form ethanoic acid.
What is the oxidising agent? Why is dilute sulfuric acid used?
A mixture of potassium dichromate(VI) and dilute sulfuric acid
It is important for the potassium dichromate(VI) to act as an oxidising agent. Without the H+ ions from the acid, no redox reaction will occur
Colour change in the oxidation reaction of ethanol by heating with potassium dichromate(VI) and dilute sulfuric acid?
What is this colour due to?
Turns from orange to green
Orange = ethanol + potassium dichromate(VI) + dilute sulfuric acid
Green = indicates the presence of Cr3+ ions, which are formed from the reduction of potassium dichromate(VI)
Displayed formula equation for ethanol + potassium dichromate(VI) and dilute sulfuric acid?
Whatever the flip this apparatus is
Ethanol is a biofuel. What is meant by the term biofuel?
A fuel that is made from biological sources, such as sugar can or corn
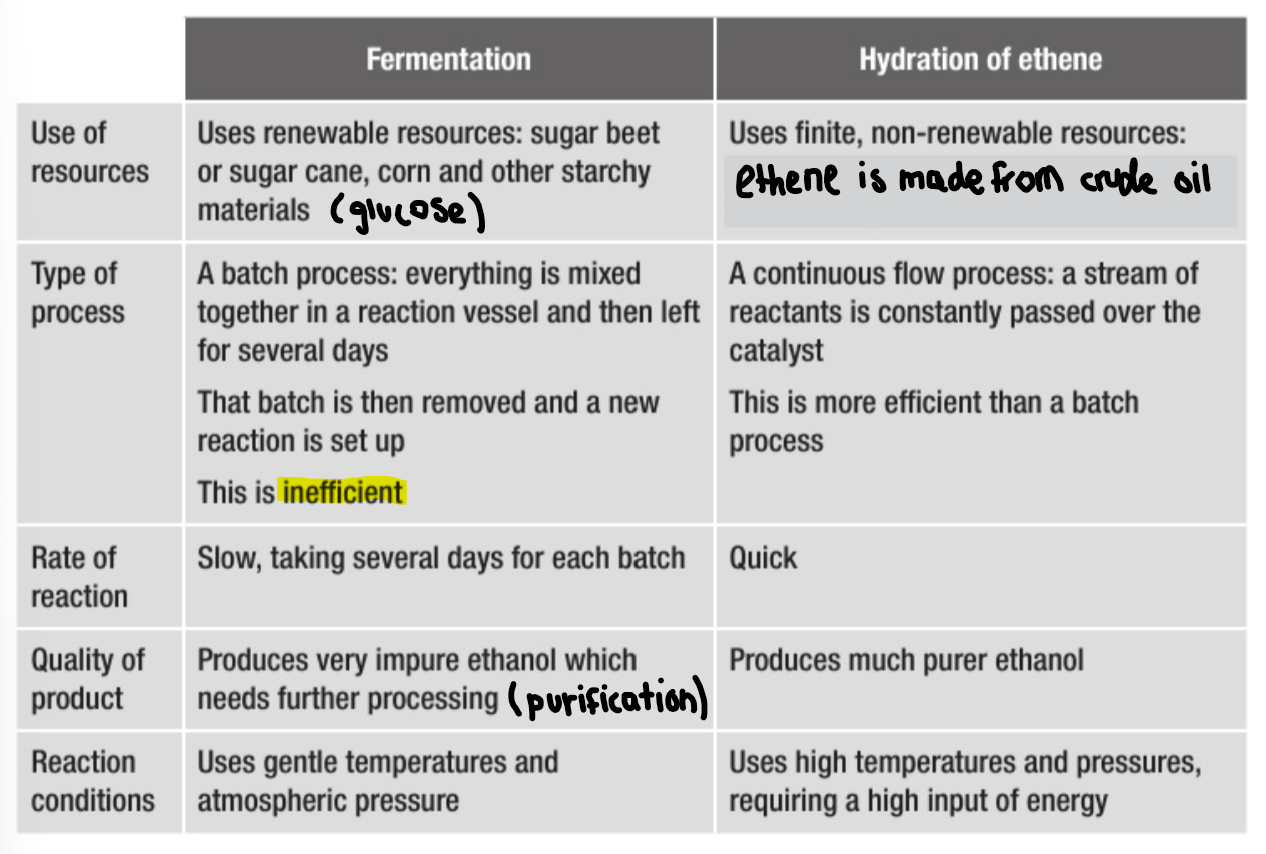
Why are biofuels used in countries?
Countries that have little or no oil industry to produce their own petrol.
Countries that have a climate which is suitable for growing sugar caneç
Countries that want to reduce dependence on fossil fuels, which are finite non-renewable resources
What are the two different processes that are used industrially to make ethanol?
Fermentation
Hydration
Making ethanol by fermentation
Process/details?
Requires aqueous glucose as the reagent
Leave the solution at 30°C at 1 atm for several days in anaerobic conditions
The catalysts are the enzymes in the yeast which convert the sugar into ethanol and carbon dioxide
Equation for fermentation to obtain ethanol
C6H12O6 (aq) → 2C2H5OH (aq) + 2CO2 (g)
Why is the absence of air and the temperature important?
Anaerobic conditions
In the presence of air, enzymes in the yeast produce carbon dioxide and water instead of ethanol
Temperature
The enzymes are proteins and if temp is too high, then they will denature so active site changes shape and substrate no longer fits
At lower temperatures, the reaction is too slow
Why is it impossible to make pure alcohol by fermentation?
What process is needed to obtain pure alcohol from fermentation? How does this work?
Yeast is killed by more than about 15% of alcohol in the mixture.
The alcohol has to be purified by fractional distillation. This takes advantage of the difference in boiling point between ethanol and water.
Making ethanol by hydration
Process/details? Equation?
The reagents are ethene and steam
Temperature: 300
Pressure: 60-70 atm
Catalyst: phosphoric acid (H3PO4)
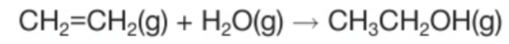
Only a small proportion of the ethene reacts. Why might this be?
Because of this, the end product we get is ethanol, ethene and steam. So what do we do?
It is a reversible reaction (segun Copilot)
Ethanol has a much higher boiling point than ethene, so when the mixture is cooled:
ethanol condenses to a liquid
ethene stays as a gas
This makes it easy to separate them.
The unreacted ethene is recycled and used again for the process
Key point:
What kind of reaction is this? Why?
This is an addition reaction because the water is added to the ethene without removing anything
Compare the two methods of producing ethanol