1. Protozoology
1/23
Earn XP
Description and Tags
All QS - morphology, location, main species, geographical distribution, epizootology,. epidemiology, LC, Pathogenesis, and clinical signs, pathology, diagnosis, treatment and control of parasite. Protozoan - one cell parasites, intracellular/extracellular. These are microscopic one-celled organisms.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
24 Terms
1.Trypanosomosis in humans
Human trypanosomosis = disease by protozoa of genus Trypanosoma. It exist in 2 primary forms - human african & American Trypanosomosis.
Belongs to: phylum - euglenozoa, subphylum - kinetoplasta, class - kinetoplastea, order - Trypanosomatida, Fam: trypanosomatidae.
Transmitted by blood-sucking insects - cyclic transmission. Can affect NS.
Diagnosis: Blood smears, microscopy, PCR & ELISA. Treatment by antiprotozoals, early treatment is more effective. Vector control + reservoir control.
Main difference → African is chancre, winterbottom + CNS, while american is romagna, chagoma - no signs - megasyndrome/heart issues.
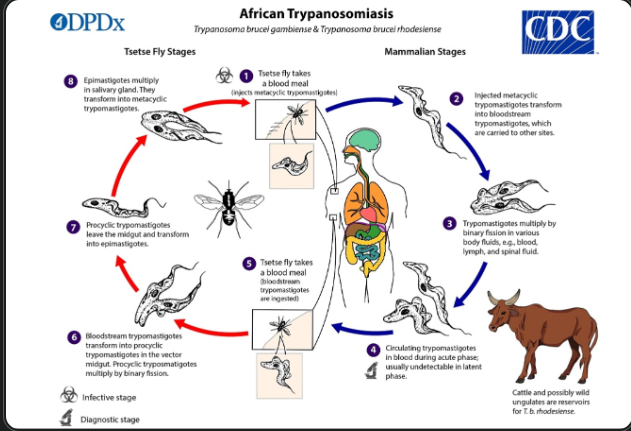
1) Human african trypanosomosis - “African sleeping sickness” - (Salivaria)
Transmission = Salivaria - Injected into the blood via saliva of the Tse-tse fly (Glossina)
Causative agents (T.brucei complex)
T.brucei Gambiense - cause chronic form (west africa), slow progression to CNS involvement
T.brucei rhodesiense - Zoonotic, acute form (East africa), rapid progression to CNS invasion. This is primarily bw. animals - fly, but may affect sometimes humans.
Location: extracellular - blood plasma, lymph, CSF, resp. organs
Host: humans, cattle, pig, car. Zoonotic.
Vector: Tse tse fly (Glossina spp.)
LC: Fly ingests trypomastigotes during blood meal → turns into epimastigotes - multiplies by binary fission → multiplies in salivary glands → infective trypomastigotes are injected during next bite → multiplies in blood, lymph, spinal fluid.
Patho/CS - 3 phases:
Primary lesion: Chancre at bite site in 2-3d, swelling
Parasitemia: parasite in blood → irreg. fever, insomnia, itching. “Winterbottom sign” - enlarged LN. Anemia.
Invasion of CNS: meningitis, encephalitis, severe headache, lethargy, deep sleep → coma → death.
Animals show abortion, infertility, anemia, weight loss, fever.
Diagnosis: Wet mount, Giemsa blood smear (low sensitivity), CSF/LN examination, Hematocrit concentration method.
Serology - limited value - PCR, ELISA
The two species are indistinguishable.
Treatment: Humans: pentamidine - first stage, nifurtimox 2nd stage
Ru: diminazene aceturate - therapy. Isometamidium. Dog/equine: melarsomine (diminazene is toxic).
Control and prevention: vector & reservoir control, insecticides, fly traps, sterile male flies - in Lab (SK).
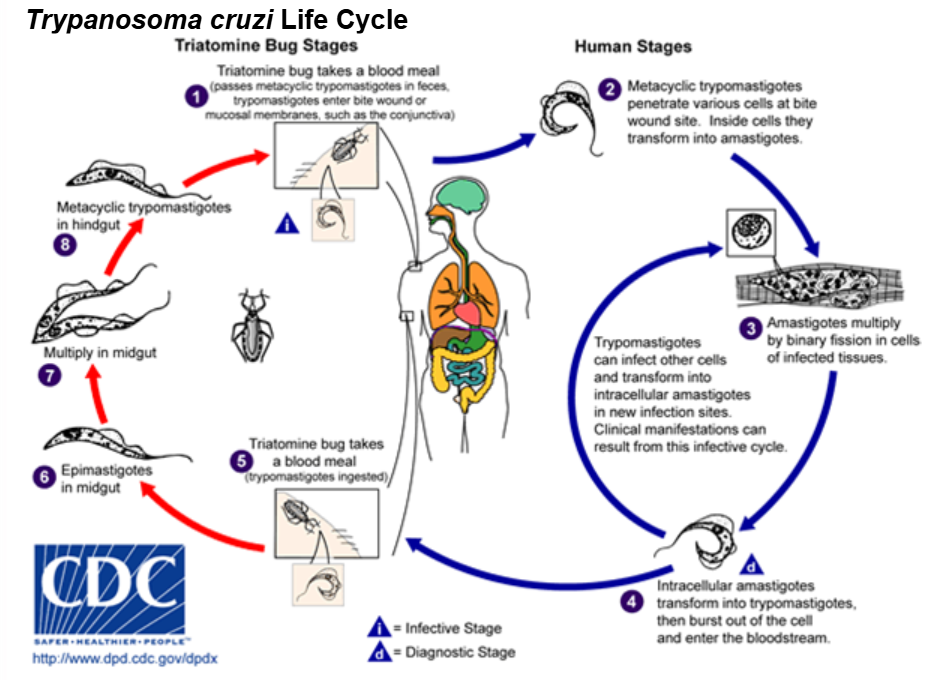
2) American trypanosomosis - “Chaga`s disease/American sleeping sickness” (stercoraria)
Transmission = stercoraria, spread by triatomine bugs (kissing bugs - triatoma, rhodnius) - parasite is passed in bug feces → enters bite wound when person rubs wound/eyes. Transmission can also be by food, blood transfusion & congenital infection.
Causative agent: T. cruzi.
Host: human, car, rodents, opossum, amardillo. Zoonotic
Location: trypomastigotes → blood, amastigotes → tissues/cells, epimastigotes → vector intestine.
LC: bug eats trypomastigotes with blood meal → epimastigotes in midgut → multiples → metacyclic trypomastigotes → passed in feces → enters through bite wound → amastigotes form in tissues, multiplies → trypomastigotes in blood - goes to new sites, Lymph, CNS.
Patho/CS:
Human:
Acute: often mild/asymptomatic, but can show romagna sign (swollen eyelid) or chagoma (skin swelling)
Indeterminate phase - latency until chronic. No signs. Seropositive.
Chronic: occurs 10-30 years later in some, leading to life-threatening heart enlargement (cardiomyopathy) or megasyndromes (megacolon/esophagus).
Dogs up to 1y: acute = death. No signs in transitional period, chronic = myocarditis.
Surviving dogs - hepatosplenomegaly, fever, anemia, resp.
Diagnosis:
Acute - blood smear, concentration methods
Chronic - serology, PCR/ELISA (trypomastigotes may be missing)
Treatment: Benznidazole (dog/man) in acute case.
Chronic for humans - mainly symptomatic treatment.’
Benznidazole for dogs daily 2 months (does not prevent chronic cardiac disease)
Trypanosomosis in animals
Species (Salivaria) - All by trypanosoma.

OBS! Nagana, dourine and surra have very similar CS.
All → fever, anemia, edema, weak, NS/paralysis. Death in severe.
Nagana - VENTRAL edema.
Dourine - GENITAL edema, plagues + skin eruptions
Surra - VENTRAL edema
WOAH notifiable diseases: Surra & Dourine
1) Nagana Disease (African Animal Trypanosomosis)
→ chronic, Africa. cyclic transmission in vector.
Location: blood vessels
LC: fly eats trypomastigotes from blood meal → become epimastigotes → multiply (binary fission) → salivary glands → infective trypomastigotes injected during bite to host → multiplies in blood/lymph/spinal fluid.
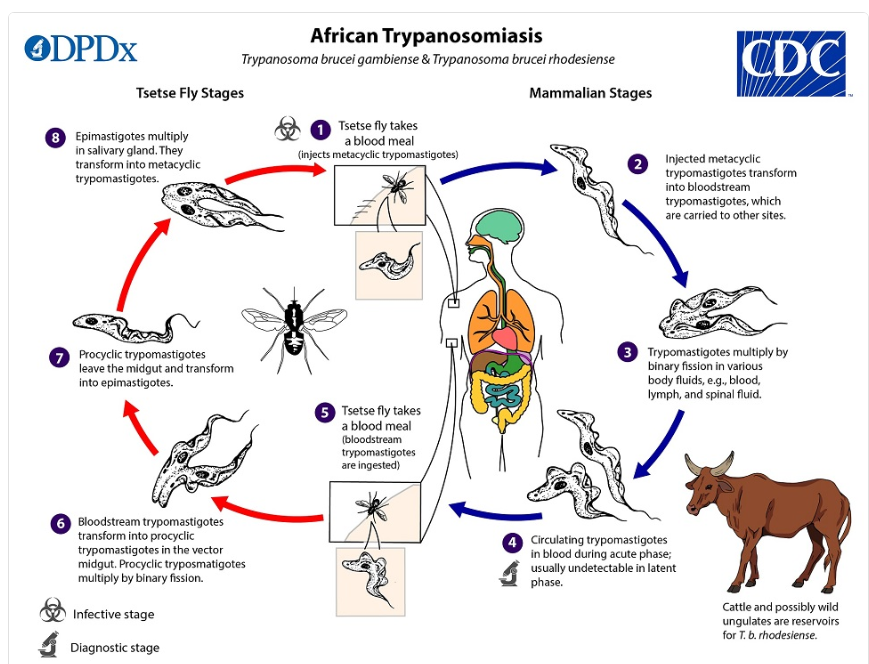
Patho/CS: diarrhea can be present in start stage, anemia, lacrimation, emaciation, death. Non-specific.
Horse: abdominal/scrotal edema, facial nerve paralysis
Dogs: inflammation - pharyngitis, stomatitis, genital/eye edema
Cattle: enlarged prescap. LN, progressive emaciation
Diagnosis: Giemsa blood smear, hematocrit methods, ELISA
Treatment: Melarsomine, diminazine (isometamidium chloride - but toxic for eq/camel). Vector control/monitoring.
2) Surra Disease - chronic, in Africa, latin-america
Evansi - Non-cyclic transmission (Parasite does NOT develop inside vector)
Spread by vector + reused needle, dogs - contaminated meat/blood.
Location: extracellular, plasma, lymph, cerebral fluid
LC: Only in mammalian host - trypomastigotes multiply by binary fission in blood/lymph/spinal fluid
Patho/CS: progressive anemia, patchy hemorrhages in conjunctiva, vaginal mucosa. Locomotor issues, CNS issues. Dogs: death within a week/month.
Diagnosis: blood smears, lymph node aspiration. Hematocrit. ELISA, PCR.
Treatment: Suramin + vector control.
3) Dourine disease (Equine trypanosomiasis) - chronic, often fatal.
Location: free in MM, tissue,blood, cerebrospinal fluid
Vector: NONE! Rarely, it can be carried by blood-sucking flies, even if only mechanically. Mare → foal by MM, milk.
Transmission: sexual by breeding
Geography: endemic in some african, asia and latin-american regions + middle east and eastern Europe.
LC: Binary fission in various tissues.
Patho/CS: Characterized by genital edema, neurological dysfunction, skin lesions (silver-dollar plaques) and death.
Genital stage with edema, swelling, vaginal discharge, fever
Skin stage with silver dollar plaques, skin lesions
Neurological with facial/muscle paralysis, weight loss, death
Diagnosis: Usually undetectable in blood. Rarely found in urethral/vaginal mucus washing or scraping. It is done mainly by CS + serology (ELISA)
Treatment and control: in many countries, treatment is not allowed, not recommended as it can suppress clinical signs. (eq is still carriers)
melarsomine can be used, examine horse before breeding, EU - control of breeding, quarantine + eliminate infected.
Leishmaniosis (cutaneous, visceral, mucocutaneous).
Order: Trypanosomatida, Fam: Trypanosomatidae, Genus: trypanosoma, Leishmania.
Species:

Morphology: Ovoid shape. rod-shaped kinetoplast. Rudimentary flagellum.
Location: MF, Cells of skin, liver, spleen, bone marrow, LN, mucosa. Also leukocytes.
FH: Dogs, humans, rodents. Zoonotic.
Vector: sandflies - cyclic transmission.
Phlebotomus → old world (europe)
Lutzomyia → new world (america)
Worldwide.
LC: Amastigotes in host are eaten by sandfly during blood meal → promastigote in gut of fly → binary fission → to pharynx → transmits promastigotes into host by bite → phagocytosis by MF → reverses back to amastigotes, division, burst of cell.
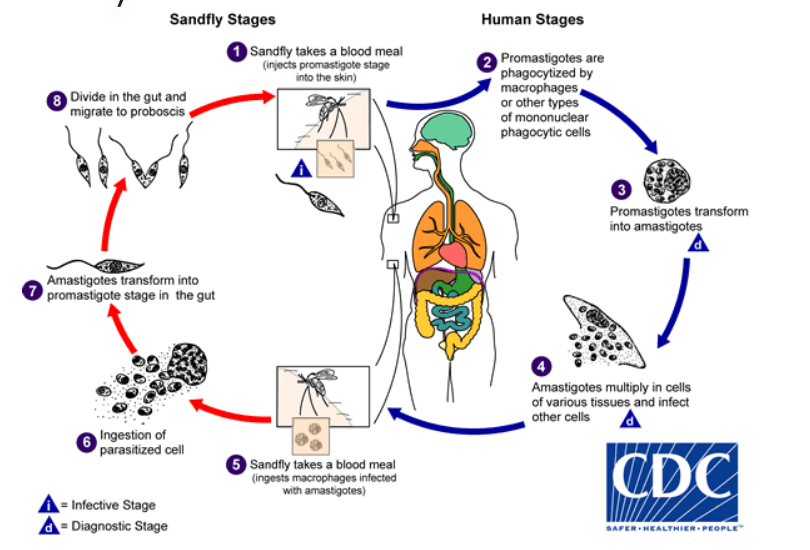
Patho/CS:
Dogs - can respond in 2 ways:
Susceptible dogs: weak cell-mediated immunity (Th2), many Ab, develops disease.
Resistant dogs: strong cell-mediated immunity (Th1 cells), few to no CS.
Many infected dogs (50%) show no signs.
Main clinical forms:
1) Visceral form: Affects internal organs (spleen, liver, bone marrow), cause general signs like fever + enlarged spleen/liver, renal failure, enlarged LN. Most severe, can be fatal.
can occur post-kala-azar cutaneous leishmaniasis even after treatment.
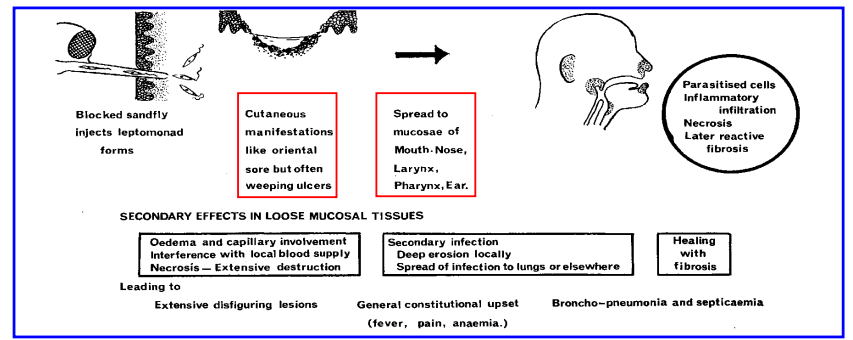
2) Cutaneous form: Affects skin only. Lesions starting at head. Alopecia, nodules/papules, skin sores, pustules. Does not itch. excessive dandruff, “volcano-like” ulcers. Usually painless.
3) Mucocutaenous form: Most destructive/disfiguring. Starts in skin → spread to MM (nose, mouth), signs of necrosis, ulcers, fibrosis/scarring..
hard to confirm as few parasites are in the lesion.
Diagnosis: Direct confirmation by finding amastigotes in skin scrapings, LN, spleen, bone marrow (biopsy).
ELISA, PCR, Cytology - hard due to low number of parasites, western blot.
Treatment: Most cases of cutaneous heal without treatment - immune after.
Other forms are very difficult to treat, long-term and relapses often.
Drugs: Milteforan, pentavalent antimony drugs, allopurinol (norway)
In dogs: can have negative side effects, expensive too.
Eradication of leishmaniosis today is not reachable but control measures can reduce risk → repellents, insecticides (spot-on, collar spray). vaccination (canileish).
Giardiasis and spironucleosis
Phylum: Metamonada, class: Trepomonadea, Order: Diplomonadida, Fam: hexamitidae, Genus: Giardia, spironucleus
Giardia species:
G.duodenalis → mammals, zoonotic.
G.enterica, canis, bovis, cati, simondi
G.muris (rodents, bird), G.ardeae (bird)
Morphology:
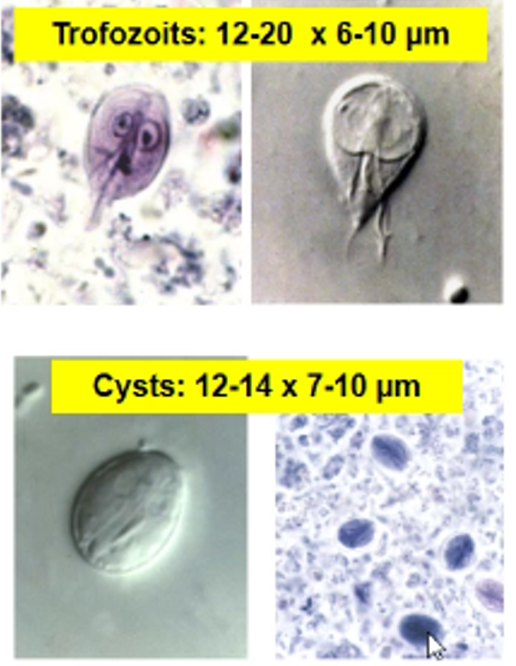
Trophozoite → pear-shape, 2 nuclei, 8stk flagella, adhesive disc, moves like a falling leaf, NO mitochondria/golgi. Replicative stage in SI.
Cyst → oval, 4 nuclei, Each releases 2 trophozoites. Infective in feces.
Location: Small intestine. Worldwide. Peaks around spring/autumn.
Transmission: contaminated food/water, fecal-oral
Life Cycle: Direct. Host eats cyst- → trophozoites released in SI → multiplies by binary fission on villi → forms cysts in colon → shed in feces.
Patho/CS: Trophozoites attach to intestinal villi → damage → villus atrophy, malabsorption, enzyme deficiencies (lactose int.), diarrhea. “Catarrhal gastroenteritis” (esp. young)
Signs range from asymptomatic to severe. Can be acute (diarrhea, vomit, pain) or chronic (weight loss, weak).
Soft diarrhea, foul-smell, mucus, grasy, pale
Not fatal for humans in developed countries, resolve on its own. (General GI signs)
Diagnosis: Flotation with faust sol. Min. 3 fecal exams, PCR.
Treatment: Fenbendazole or febantel (panacur) for 3d. (cat/dog)
cleaning, control of flies, nutrition, carbs supports, protein suppresses.
good prognosis, most recover well. Not good in immunosuppressed.
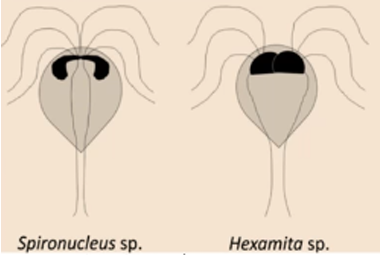
SPIRONUCLEUS (Hexamita/hexamitiasis)
Order: Diplomonidida, Family: Hexamitidae, Genus: Giardia, Spironucleus
Hexamita meleagridis (turkey), Columbae (pigeon), muris (mice/rat), salmonis/truttae (fish).
Morphology:
Trophozoites - no ventral adhesive disk here.
cyst: oval, 8 nuclei, cannot move, 4 trophozoites.
Location: small intestine, cecum, Bursa fabricii, liver/body cavity
Transmits by fecal-oral. Similar LC to giardia.
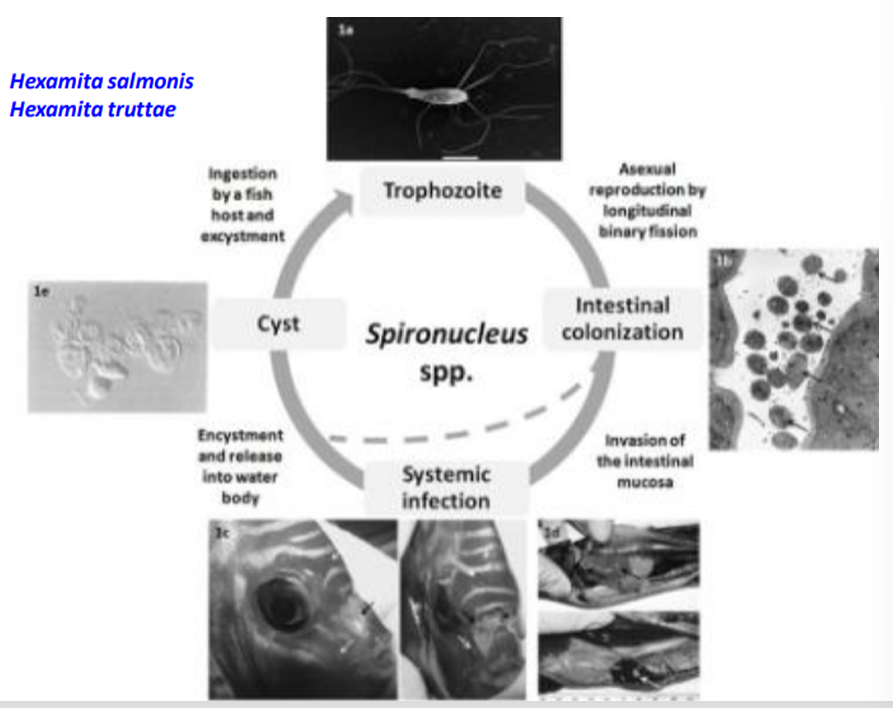
Patho/CS: non-specific - watery yellow diarrhea, weight loss, dehydration, death. enteritis.
ruffled feathers, high mortality in young. Severe - coma, convulsions and death (meleagridis).
Diagnosis: Detect cysts/trophozoites - PM - catarrhal inflammation in SI, fluid in intestine.
No known treatment. Prevent by hygiene, nutrition, separate.
Histomonosis
histomonosis/Histomonas Meleagridis (Blackhead disease)
Phylum: Metamonoda, Class: Trichomonadea, Order: Trichomonadida, Fam: Monocercomonadidae, Genus: Histomonas.
Cause disease in birds, mainly turkeys
Lives in Cecum & Liver
Spread mostly through eggs of the worm Heterakis Gallinarum (Vector - Paratenic host). Worm eggs can survive in soil for over 2 years, eartworms can carry them too. Can spread directly bw. birds by cloacal drinking (birds sit on feces). NO cyst formation.
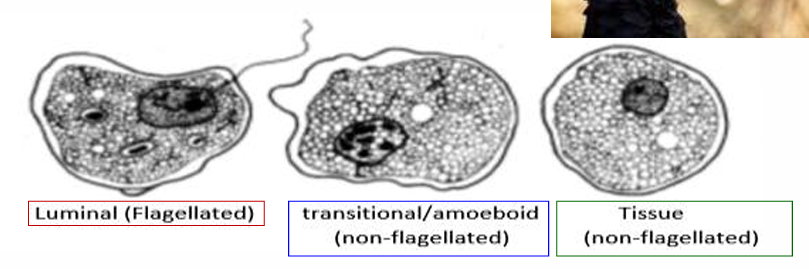
Morphology: Similar to trichomonas. Nucleus & Flagellum. 3 forms:
Flagellated luminal form - in cecum
Amoeboid invasive form - enters tissues/Liver
Resting form - inside heterakis eggs.
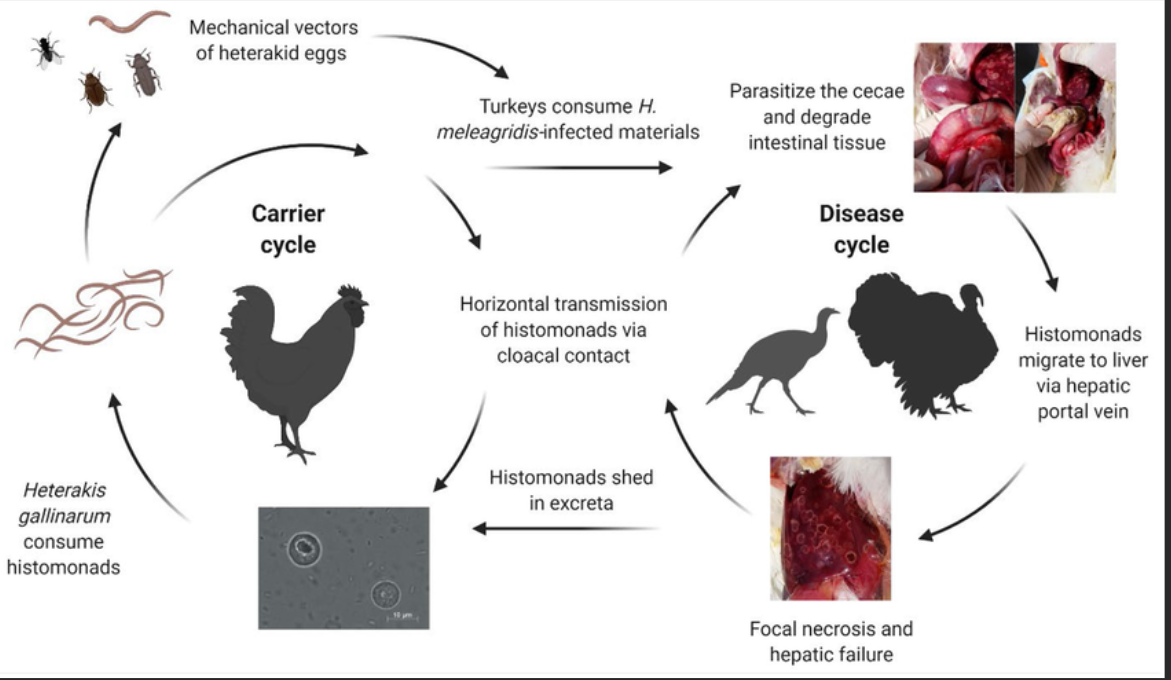
Life Cycle - Indirect route (via Heterakis):
Histomonads are shed in feces of infected birds → the nematode (heterakis gallinarum) eats them → histomonas enters nd survives inside the eggs → passed in feces → survive in soil → turkeys ingest the eggs → invade ceca → travels to liver.
Direct: from bird to bird.
Patho/CS:
Mostly severe in young turkeys (14 weeks old) - recovered birds get scarred cecum/liver.
Depression, ruffled feathers, Sulfur-yellow diarrhea
Dark/cyanotic head = Blackhead! Circulatory issues - death within 1-2w untreated.
Chickens: More resistant. Asymptomatic/mild. Can get liver damage.
Lesions: Necrotic ceca with Hard cheesy Plugs - exudate. Liver lesions/scars.
Diagnosis: PM & PCR/culture detection. Histology.
Diff. dx: consider candida albicans for ex.
Treatment: No approved EU drugs (antihistomonal).
Paramomycin + alternative ATBs can be used in feed at conc. 400ppm.
Prevent by separation, hygiene/dry litter, rotating pasture.
Bovine and Avian trichomonosis
Taxonomy: Phylum: Parabasala, Class: Trichomonadea, Order: Trichomonadida, Family: Trichomonadidae, Genera: Tritrichomonas, Trichomonas, Pentatrichomonas
Bovine trichomonosis → Tritrichomonas foetus B (bovine strain)
Avian Trichomonosis → Trichomonas gallinae. (also have Tetratrichomonas gallinarum/anatis/ansertis
There is also T.foetus - strain C for cats. T.vaginalis, tenax, Pt. homonis.
Phylum - Parabasala - Morphology:
Pear-shaped protozoa with flagella + undulating membrane.
Flagella varies - bovine (3ant. + 1 post.) & Avian (4post.)
Named after parabasal apparatus (Golgi body on nucleus by fibrils)
Has hydrogenosomes instead of mitochondria. (energy - metabolism)
Reproduces by longitudinal fission.
NO true cysts (Some form pseudocysts - T.foetus, muris)
Infective stage = Trophozoite.
Parasites of genital & digestive tract. Anaerobic.
1) Bovine Trichomonosis:
In reproductive tract of cattle. Spread by mating/AI.
Bulls = major carriers (often asymptomatic)
Life Cycle - Direct. Trophozoites are mobile and acts as infective stage.
Mating → once inside host, trophozoites will multiply asexually - binary fission.
Female: colonizes vaginal mucosa → cervix, uterus.
Male: in preputial mucosa.
Patho/CS:
Early abortion, infertility/irregular estrus. Endometritis, pyometra.
Bulls - inflammation of preputial sac, nodules. Under 4y = lifelong carriers, over = temporary infected.
Diagnosis: History & Lab test. Vaginal/preputial washings, microscopy, culture, PCR.
Treatment: WOAH-listed (need to report), Symptomatic.
Nitroimidazoles (not registered for use) (metronidazol, dimetridazol)
resistant to penicillin, streptomycin + many other ATBs.
Prevent: check bulls, hygiene, disinfect AI instruments, TrichGuard Vaccine available.
2) Avian Trichomonosis:
Mainly in pigeons (but also turkeys, parrot), Located in mouth, crop, esophagus.
Spread by crop milk feeding, contaminated water, eating infected birds (hawks)
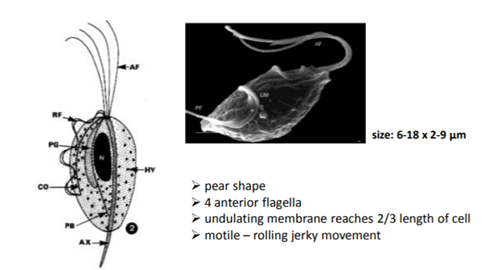
LC: Direct. Same.
Patho/CS: Yellow necrotic lesions in mouth/Throat (Cheesy lesions). Can spread to liver/heart. Difficulty eating/breathing. Often fatal in young pigens.
Nodules in upper resp. tract (beak, sinus, esophagus, throat)
Acute - necrotic masses (creamy) → chronic - enlarged lesions, invades.
Diagnosis: Crop swab microscopy
Treatment: Nitroimidazoles (non-food birds) - like metronidazole
Pathogenic and potential pathogenic Amoebae of animals and humans
Phylum: Amoebozoa, Class: Entamoebidae Lobosea, Order: Entamoebida, Family: Entamoebidae, Genera: Entamoeba, Naegleria & Acanthamoeba
General: varies in shape, moves by pseudopodia, feed by phagocytosis, has nucleus with chromatin granules, can be parasitic or free-living, Many form cysts. Reproduce mainly by binary fission/Mitosis.
Important species:
Entamoeba histolytica (colon/liver) - in humans, dog/cat, pig, rat (Zoonotic) Cosmopolitan.
Naegleria Fowleri (nasal cavity/olfactory nerves/meninges)
Acanthamoeba (skin, resp. urogenital, meninges)

1. Entamoeba histolytica (Pathogenic - causes Amoebiasis)
Disease - causes amoebiasis / Amoebic dysentery
Can cause intestinal / extraintestinal disease
Morphology:
Trophozoites have pseudopodia, ingested RBCs, central karyosome.
2 Forms: Forma minuta → non-invasive/non-pathogenic in lumen & Forma magna → invasive/pathogenic
Cysts have 1-4 nuclei - quadrinucleate cyst (infective stage).
Location: Colon (cecum, sigmoid colon)
can spread to liver, lungs, brain
Typical is forma minuta in colon, dysentery, ulcers. While forma magna is in other places, causing abscess, hepatitis.
Transmission: Fecal-oral. Eating cysts in food/water. Hand-mouth.
Life cycle (Direct): Cyst ingested → excystation in SI → trophozoites in colon → multiply by binary fission → cyst excreted.
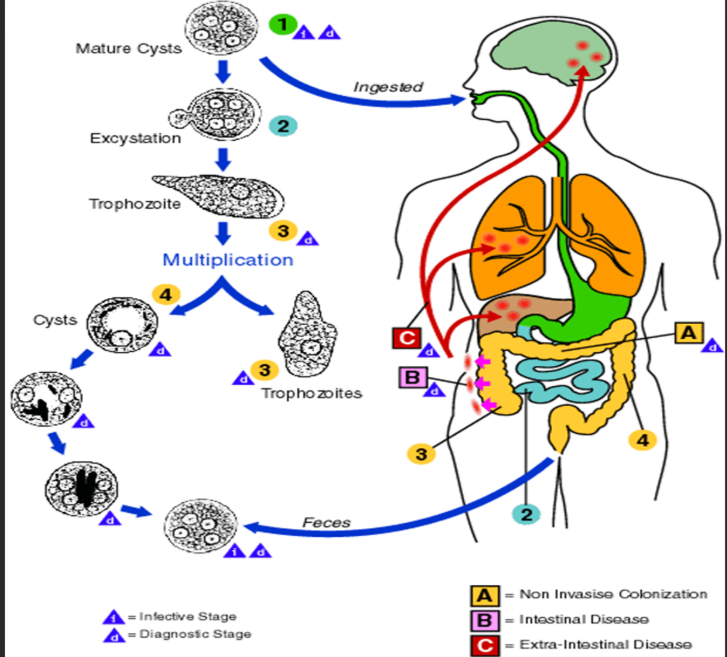
Patho/CS: Mostly asymptomatic, ranging to intestinal.
Intestinal: diarrhea, colitis, ulcers, perforation
Extraintestinal: liver abscess (“anchovy/chocolate paste pus), hepatitis, pulmonary abscess
Diagnosis
Fecal exam (cysts/trophozoites - but common to have nothing)
Serology - ELISA
CT/imaging for abscess
Treatment: Nitroimidazoles (ex. metronidazole)
2. Naegleria fowleri (“Brain-eating amoeba”, free-living)
Morphology: 3 forms - trophozoite (infective), flagellate and cyst.
Causes PAM = Primary Amoebic Meningoencephalitis - intense headache, fever, nasal blockage, vomit, coma and death.
Usually affects healthy young people, almost always fatal (Humans).
warm freshwater entering nose, amoeba migrates from nose → olfactory nerve → brain.
Dg: CT/Biopsy. Treatment - mostly fatal. poor prognosis.
3. Acanthamoeba spp.
Opportunistic, mainly in immunocompromised. Humans.
Causes: GAE = Granulomatous Amoebic encephalitis & keratitis (eye infection) → headache, encephalitis, blindness, skin ulcers/papules.
Form: Trophozoite (mitosis repl.) & cysts.
Transmission by broken skin, resp. & eyes. Found in pools, hot tubs/tap water.
Dg: eye scraping, CT, biopsy for GAE. FATAL - poor prognosis.
8.Eimeriosis of cattle, sheep, and goats.
Taxonomy: Phylum: Alveolata, Subphylum: Apicomplexa, Class: Coccidea, Order: Eimeriida, Family: Eimeriidae, Genus: Eimeria
Obligatory intracellular, single-celled. no cilia/Flagella.
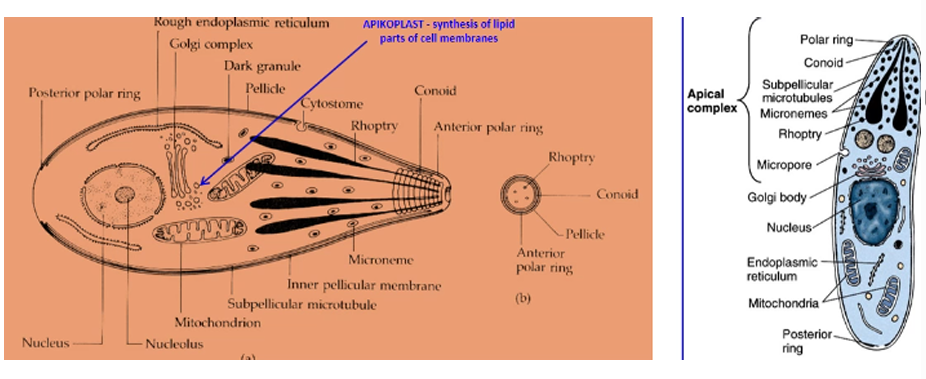
Apical complex for entering host cells - at end of sporozoite/merozoite
Consist of: polar rings, conoid, micronemes, Rhoptries, dense granules (release enzymes to help go into host)
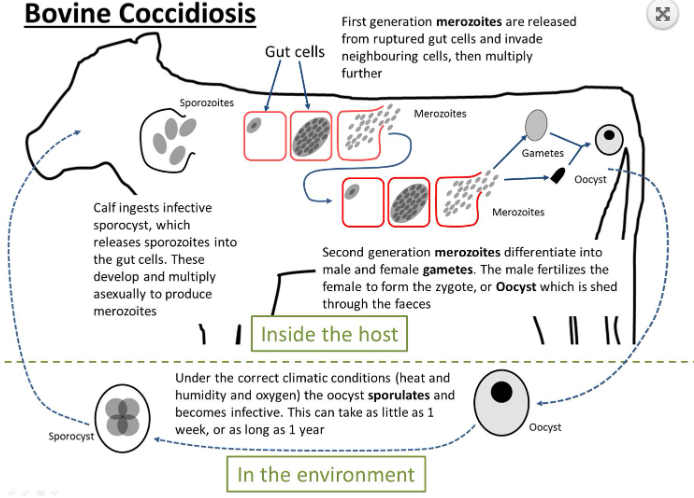
LC: Direct. (same for eimeria, cryptosporidium, cystospora)
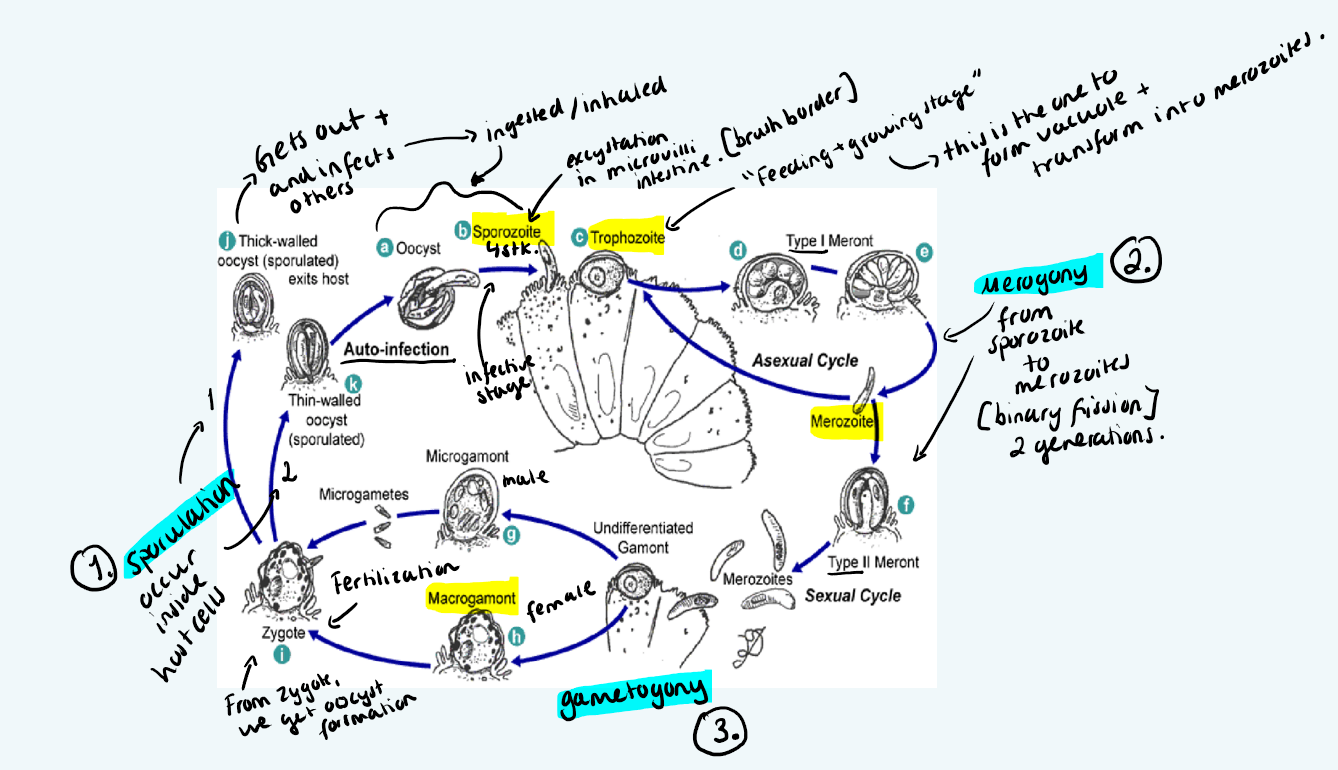
Merogony (asexual): host eats sporulated oocyst → sporozoites released → trophozites → meronts → inside meront, nuclei reproduce asexually → merozoites from ruptured gut cells, then invade other cells.
Gametogony (sexual): Merozoites → gametocytes - macro & microgamete → fusion → zygote → unsporulated oocyst excreted in feces.
Sporogony (environment): outside host, oocyst sporulates → infective sporulated oocyst forms.
Morphology: Oocyst - oval, 2 shells, 4 sporocysts each with 2 sporozoites. Infective stage is sporulated oocyst. micropyle can be present.
Eimeria destroys the intestinal epithelium, reduces absorption of nutrients → diarrhea, dehydration, severe hemorrhage.
Bovine Eimeriosis, worldwide in young cattle.
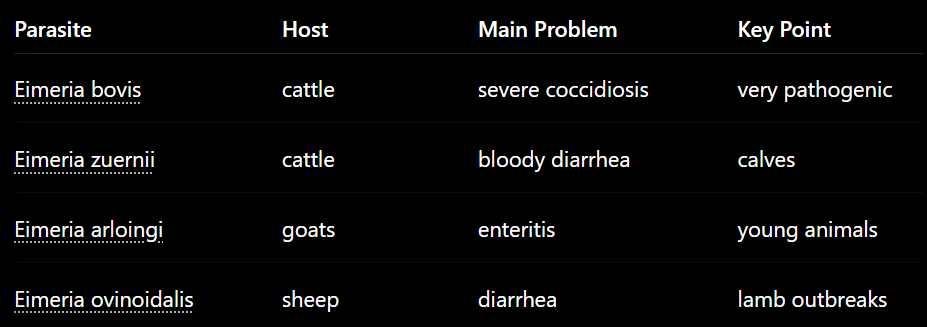
Species: E.bovis (Very patho), E.zuernii
In SI, Fecal-oral route.
Bovis: death within 4w. Severe diarrhea, tenesmus, fever, hemorrhage, edema.
Zuernii: bloody diarrhea. Death after 7d of symptoms.
Diagnosis: Flotation method. Treatment: Amprolium, Sulfamethazine. Prophylaxis of coccidiosis in cattle - monensin rumensin.
Eimeriosis of sheep and goat, worldwide.
Sheep: E.ovina, intricata (SI) + E.parva (non/low patho).
Goat: E.arloingi (crypts), E.christenseni (SI)
Mostly affects young, adults usually immune. Stress/crowding = predispose. Host specific.
CS: dirty tail, diarrhea, poor growth, anorexia, bloody diarrhea in severe, sudden death possible.
Dg: Fecal flotation - oocyst are common in healthy ones too, PM intestinal scrapings. Treatment: sulfonamides, monensin prophylaxis.
9.Eimeriosis of rabbits and hares.
Eimeriosis in rabbits
Main species:
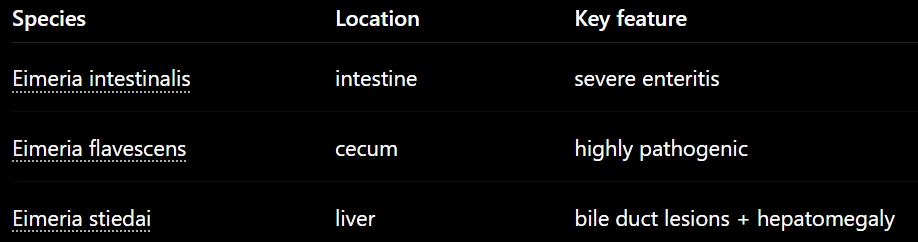
E. intestinalis – Intestinal. Highly pathogenic!
E. flavescens – Highly pathogenic! Cecum
E. magna, Media, piriformis (LI), steidai (liver).
Common during weaning, high risk in crowded farms, young.
Life cycle:
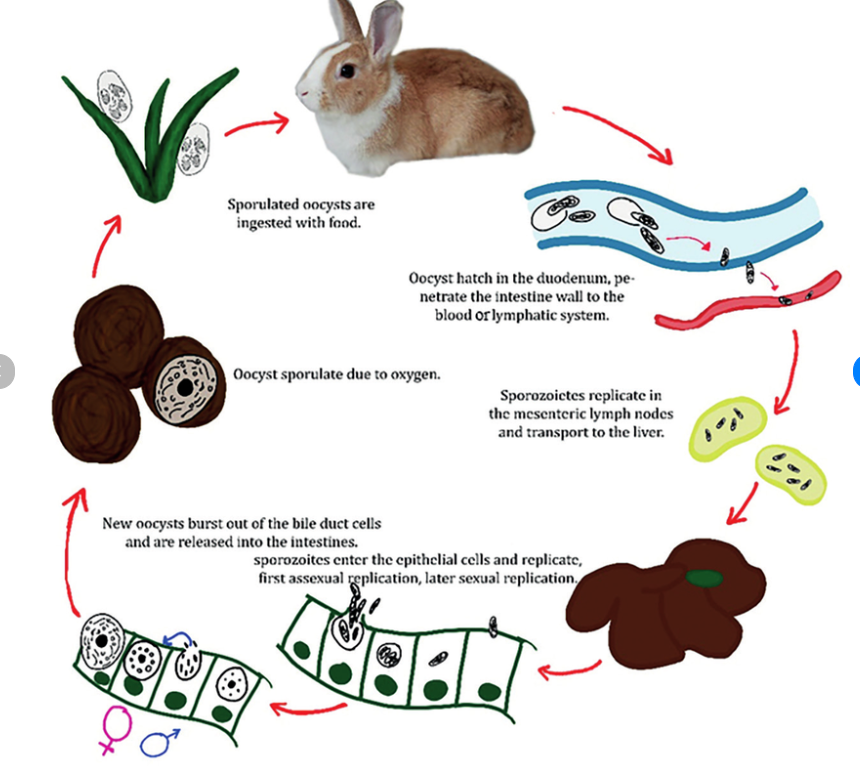
Clinical signs:
Intestinal: enteritis, watery diarrhea, dehydration, weight loss
High mortality in young. In severe cases → convulsions/paralysis.
Many intestinal infections are mild/subclinical.
Stiedai: Intestinal mucosa → blood → liver → bile ducts → hepatitis, jaundice, enlarged bile ducts + bloackage, hepatomegaly, white nodules.
Diagnosis: Fecal flotation, PCR, PM
Treatment: Monensin, Toltrazuril, Sulphonamids.
Hare: E. townsendi & E. leporis
Disease uncommon in wild hares. Mostly in young during wet season (oocyst survives better).
10.Describe Eimeriosis in poultry.
Main species - Intestine, Worldwide
Most common:
E.tenella (chicken - cecum, highly pathogenic, causing bleeding → anemia). Typically in fattening chicks. death in under a week.
E.Acervulina (SI, lesions - white spots, duodenum)
E.Maxima (chicken, SI - jeju/ileum, has the largest oocysts)
E.necatrix, E.praecox, E.brunetti
E.danilovi (duck)
Turkey: E.truncata (kidney), E.meleagrimitis
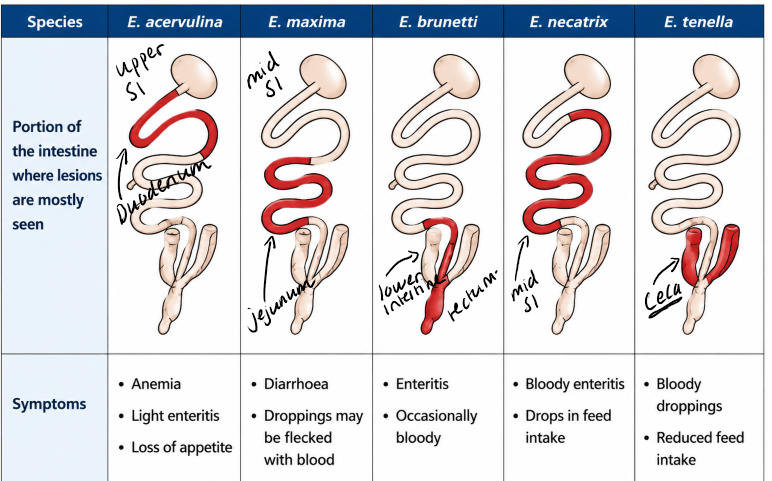
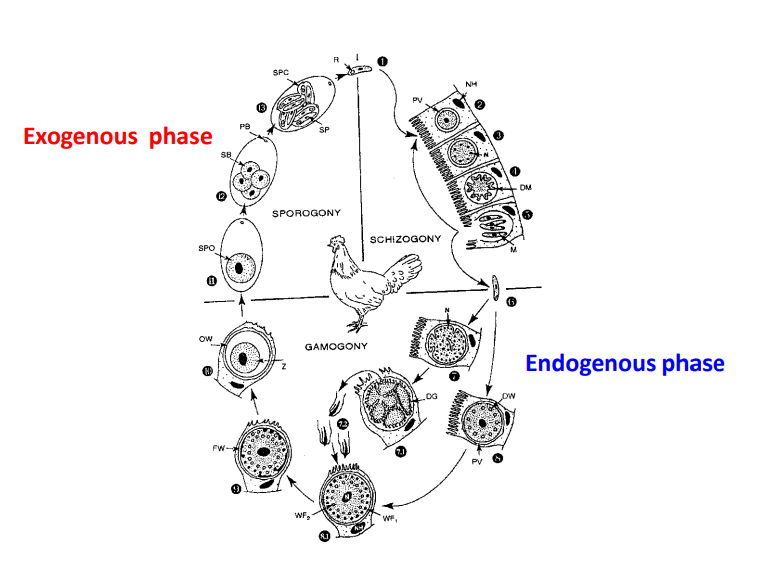
Chicken Eimeriosis:
Affects young chicks, kept in high conc. in small areas. Cause economic loss in industry due to less weight gain. Occurs through fecal-oral route. Oocyst are highly resistant and survive well in litter.
Life cycle: General apicomplexan scheme.
Merogony (3 generations of merozoites) → gametogony → sporogony.
E.tenella: 1st gen: 900 merozoites, 2nd: 300, 3rd is gamogony.
Patho/CS:
Characterized as catarrhal-hemorrhagic enteric disease
General intestinal signs of diarrhea, anemia decr. growth + nodules, sometimes CNS disorders.
Pathology due to the destruction of intestinal cells, each sp. has tissue site specificity
E. tenella - quick, bleeding into lumen of cecum, whitish mucosa, cecal cores of clotted blood.
Diagnosis:
Intra vitam: coprological examination - flotation method - find oocysts
PM: Look for scratches of the epithelial layers of the intestine, histological sections, we can see villus atrophy, crypt hyperplasia and incr. WBC infiltration.
Treatment: Anticoccidals (amprolium, sulphonamides, totrazuril)
ATBs - tylosin, amoxicillin, Supportive care. Management of environment, cleaning, livacox gel vaccine.
11.Eimeriosis and Cystoisosporosis of pigs
Pig coccidia - Parasites of intestinal epithelium. Mainly affect young piglets. Fecal oral transmission. Cause enteritis & diarrhea.
LC: merogony (from sporocyts → sporozoites → trophozoite → meront → merozoites released) → gamogony (gametes → oocyst) → sporogony (environment - sporulates into sporulated oocysts)
1) Pig Eimeriosis:
In SI, worldwide
Main species: E. scabra, E. suis
E. debliecki, E. neodebliecki, E. spinosa, E. poliata
LC = Monoxenous, entire cycle in ONE host.
Patho/CS: Usually affects lower SI.
Acute enteritis, poor growth/loss of condition, perianal staining.
yellow fibronecrotic pseudomembrane + bloody diarrhea
Diagnosis: Fecal flotation, examine intestine (8-10d)
Treatment: Anticoccidials.
2) Pig Cystoisospora (aka pig isosporosis)
Order: Eimeridia, Fam: sarcocystidae, Genus: cystoisospora, sarcocystis
Species: Cystoisospora suis
In SI, Mostly affects piglets. Adults usually asymptomatic. Worldwide (EU, america).
Morphology: Oocyst with 2 sporocysts, each with 4 sporozoites.
Small, oval, 2 thin shell, unsporulated, transparent. No stieda body.
Very common due to short life cycle, piglets highly susceptible, oocyst resistant to disinfectants, rodents can be paratenic host.
Life cycle: Monoxenous in pigs (direct cycle) but can also use paratenic host like rodents.
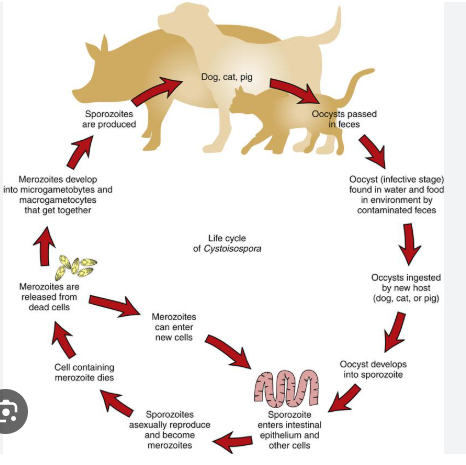
Patho/CS:
Merogony + gametogony in enterocytes of SI → villous atrophy/fusion, necrotic enteritis, crypt hyperplasia → leads to poor absorption and secondary bacterial infections.
Pasty/watery diarrhea (NO blood), weight loss/poor growth in piglets.
Diagnosis: Fecal smear/flotation, McMaster count, autofluorescence microscopy, histology/intestine scraping at necropsy.
Treatment: Toltrazuril, sulphadimidine
ATBs usually ineffective in piglets
Treat with oral baycox once in first week after birth.
12.Cystoisosporosis of carnivores.
Phylum: Alveolata, Subphylum: Apicomplexa, Class: Coccidea, Order: Eimeridae, Family: Sarcocystidae, Genus: Cystoisospora
Infects epithelium of SI, worldwide, fecal-oral transmission.
Main species:
Dog: C. canis, C. ohioensis, C. burrowsi, C. neorivolta
Cat: C. felis & C. rivolta (larger oocyst than felis)
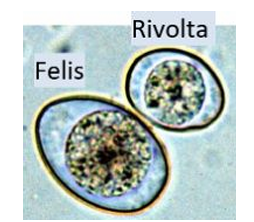
Morphology: Oocyst is small, oval, has 2 sporocysts, each containing 4 sporozoites. 2 thin shells, transparent when unsporulated.
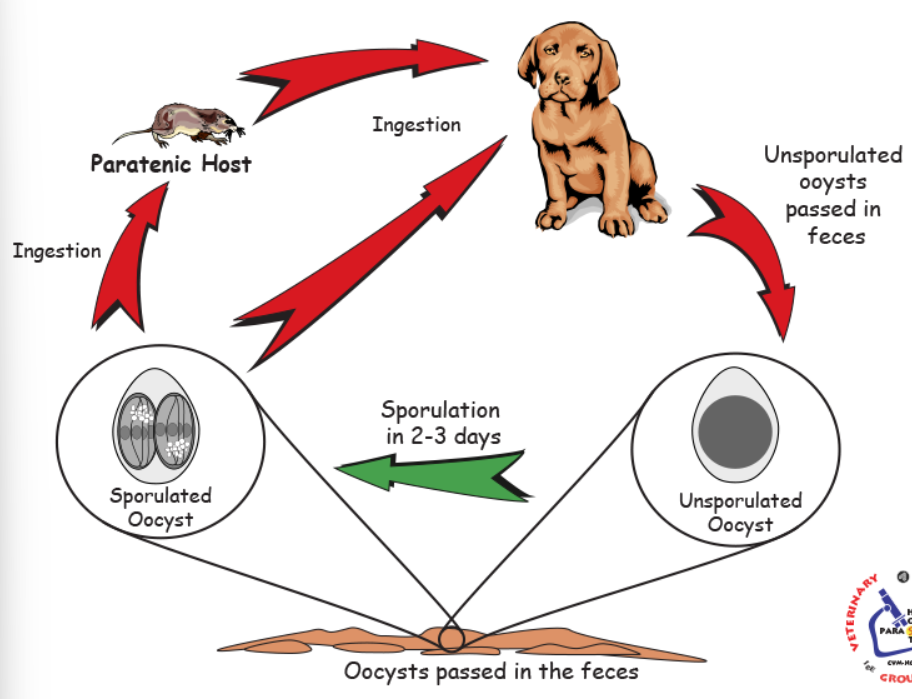
FH - dog, cat. Host specific. Rodents can be paratenic host.
Life cycle: Same basic coccidian. In one host or with paratenic host.
Sporogony (environment) → into hostm excyst in SI, into epithelial cells (merogony, binary fission) → gamogony → unsporulated oocyst into feces.
Inside paratenic host → parasite forms a dormant cyst (hypnozoite). NO disease. But FH is infected if it eats the rodent.
Patho/CS:
Mild infection: often asymptomatic
Severe infection (esp. C. canis): in puppies 3-4 weeks - 4m. old. gets GI signs, diarrhea (bloody), vomit, abd. pain, hemorrhagic enteritis. Anorexia.
Systemic: dehydration, fever, anemia, poor growth
Inc. period: 1week
immunity in cats: 2-5months (temporary)
Diagnosis: Coproscopy (fecal exam) detection of unsporulated oocyst. Flotation method with FAUST.
Treatment: toltrazuril, ponazuril. Hygiene, remove feces, disinfection of breeding areas, not feeding of raw meat.
13.Cryptosporidiosis of mammals and birds.
Phylum: Alveolata, subphyl: Apicomplexa, Class: Coccidea, Order: Cryptosporida, Fam: cryptosporiididae, genus: cryptosporidium
General: Infects microvilli of GI tract, worldwide, fecal-oral transmission, waterborne/foodborne zoonosis, imp. in calves, young animals, immunocompromised, cause watery diarrhea.
Species: C.hominis (humans), parvum (ru, man,rodent), bovis, andersoni (ru), suis, canis/felis, muris.
Birds: C.baileyi (resp. tract, intestine), C.galli (stomach), C.meleagridis (turkey)
In Genus cryptosporidium we have:
Small oocysts - infects intestinal enterocytes = C.parvum, homonis, canis/felis, bovis & suis
Large oocysts - infects stomach glands = C.muris, galli
Morhology: Oocysts are very small (5-7um), round, pink, 2 shells, sporulated (4 free sporozoites). NO sporocysts. Microgametes lack flagella.
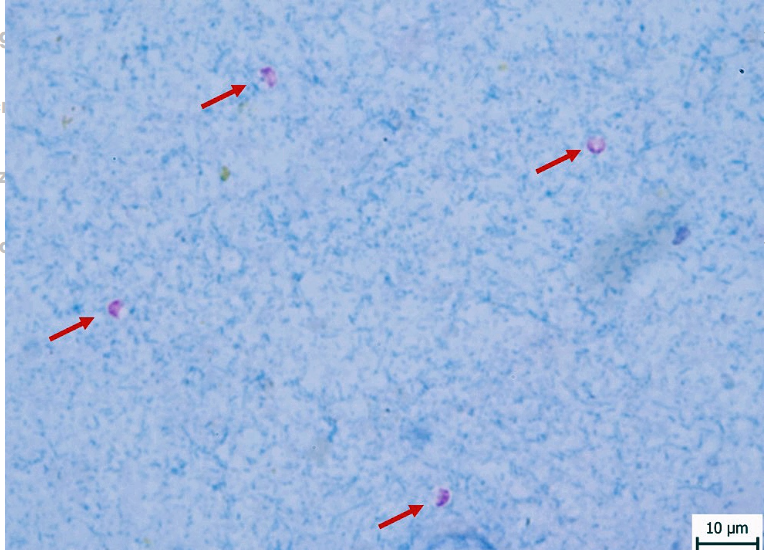
Location: Intestinal microvilli, gastric glands, sometimes resp. tract
Intracellular but extracytoplasmic - Parasite (sporozoite) sits in membrane pocket - inside host cell membrane but outside cytoplasm.
Life Cycle: entire cycle inside host - sporulated oocyst eaten → invade microvilli → merogony → merozoites → gamogony → gametes fuse into oocyst → sporogony → sporulated oocysts shed in feces (already infective).
Pathogenesis: Villus shortening, crypt cell hypertrophy, inflammation of lamina propria, incr. water secretion → diarrhea + malabsorption.
CS in calves: Profuse watery diarrhea (mucus/fibrin), dehydrated, anorexia, wek, fever, foul-smelling yellow feces.
CS in humans: severe in young, elderly & pregnant, immunocompromised. Watery diarrhea, abd. cramps, weight loss, mild fever. Can be fatal in immunodeficient.
Diagnosis: Microscopy - fecal flotation & acid-fast stains. Stain with kinyoun stain or ziehl neelsen. Findings - red/pink oocysts. Other tests ELISA/Fluorescent Ab tests.
Treatment/therapy:
Animals - halofuginone (halocur) for calves.
Humans - nitazoxanide
dog/cat - pyrvinium pamoate
Suportive care, no fully curative therapy. Prevention!
14.Neosporosis
Classification: Phylum: Alveolata, Subphyl: Apicomplexa, Class: Coccidea, order: Eimeridae, Fam: sarcocystidae, genus: Neospora.
Species: Neospora caninum - cause bovine neosporosis
Very similar to Toxoplasma gondii in morphology/LC.
Transmission: Fecal-oral, transplacental, infection often lifelong. Main disease is abortion in cattle, neurologic disease in dogs/calves.
Hosts: Dog, coyote, wolf. IH: cattle (mainly), eq, sheep
Morphology:
Oocysts: round/oval, 2 sporocysts, each with 4 sporozoites.
Tachyzoites: intracellular, inside vacuoles in cytoplasm of cells
Tissue cysts: thick walled
Location: Intestinal enterocytes (multiplies). Cysts in muscle/neural tissue
Geo: First found in Norway in dogs, but exists worldwide. In cattle, occurrence is in cosmopolitan.
Life cycle: Indirect. Merogony + Gametogony in FH, Sporogony in environment. IH - tachyzoites spread through body, tissue cysts form.
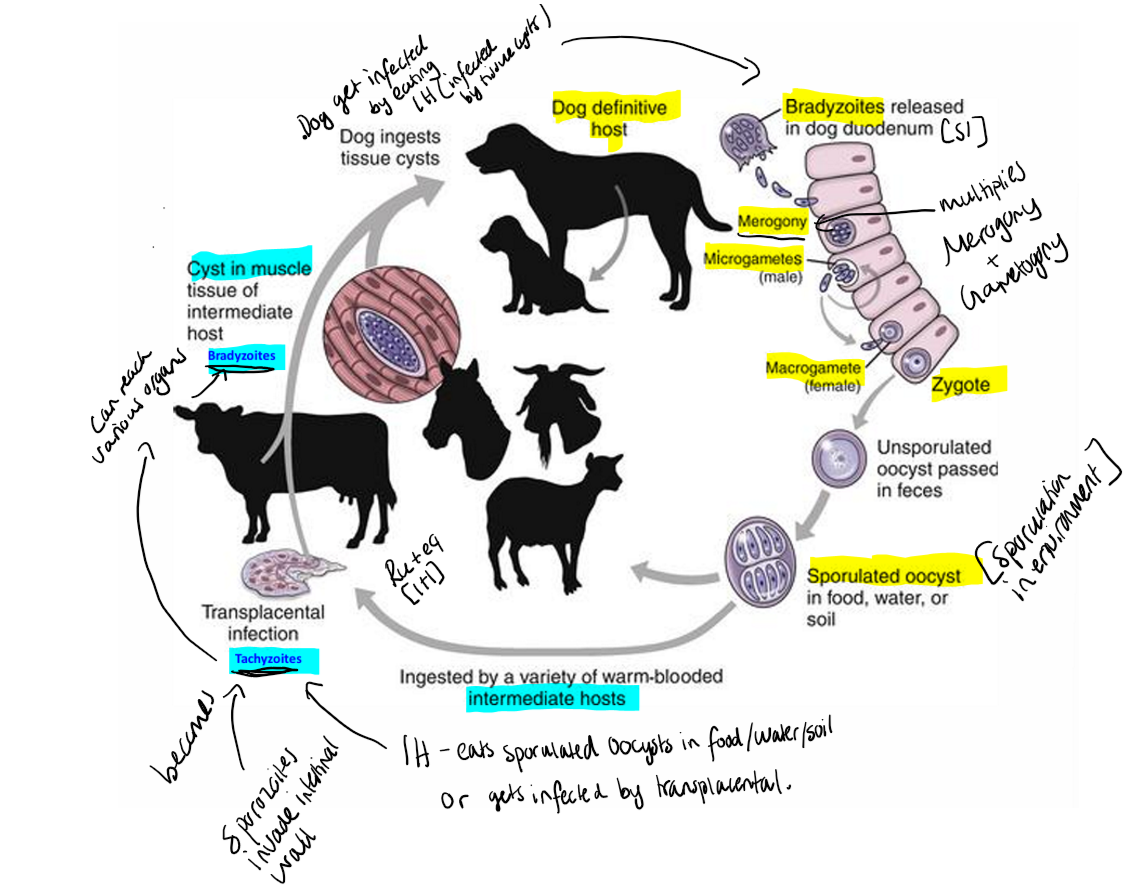
Pathogenesis and clinical signs:
Adult cow: Abortion, can occur from 3 months gestation → term. Possible fetal outcomes are fetal death, mummification, stillbirth, autolysis, weak calf/chronically infected. Seropositve abort more often.
Calves: under 2months, neurologic signs - ataxia, weak reflexes, abnormal limb flexion, not standing, exopthalmosis - asymmetric eyes.
Dogs: Neuromuscular disease, muscular atrophy, paresis, paralysis, muscle pain/rigid, torticollis. Skin ulcers
Congenital infection: from mother to puppies, leading to necrosis in brain/spinal cord/nerves → severe neuroligc disease.
Diagnosis: Serology → detects specific Ab to identify between neospora and T. gondii. Flotation method.
CS (a lot of abortions in the herd lately), PCR, Histopathology, examination of fetus for definitive diagnosis.
Treatment: Dogs - toltrazuril.
No effective treatment for tissue cysts. No effective prevention of transplacental transmission in cattle
Control - feed, contact with abortion/placenta tissues.
15. Sarcocystosis of mammals
phylum: Alveolata, Subphyl: Apicomplexa, class: Coccidea, Order: Eimeridae, fam: Sarcocystidae, genus: Sarcocystis
Main species:
Zoonotic species: S.bovihominis (cattle → man) & S.suihominis
S. cruzi/bovicanis – cattle (IH) → canids (FH)
S. bovifelis – cattle → cats
S. suicanis – pigs → dogs
S. tenella (ovicanis) – sheep → dogs
S. gigantica (ovifelis) – sheep → cats
S. suifelis, S.equicanis, S.capricanis, S.neurona
1st name is IH, then FH.
Morphology/location: Intracellular parasite, forms cysts in striated muscles of IH. Some species form macrocysts visible to naked eye. Oocysts contain 2 sporocysts with 4 sporozoites each.
Worldwide, Tissue mainly in skeletal & cardiac muscle
Transmission: Predator-Prey LC. Fecal-oral transmission. Ingestion of sporocysts/tissue cysts. Dogs/cats contaminate feed with feces.
Life cycle: 2 hosts (heteroxenous).
Asexual reproduction - sporocysts eaten → parasite migrate via bv → Merogony → meronts develop in endothelium → metrocysts → bradyzoites
In FH - sexual reprod. - gametogony occur in intestine & sporogony within host. Sporocysts shed in feces.
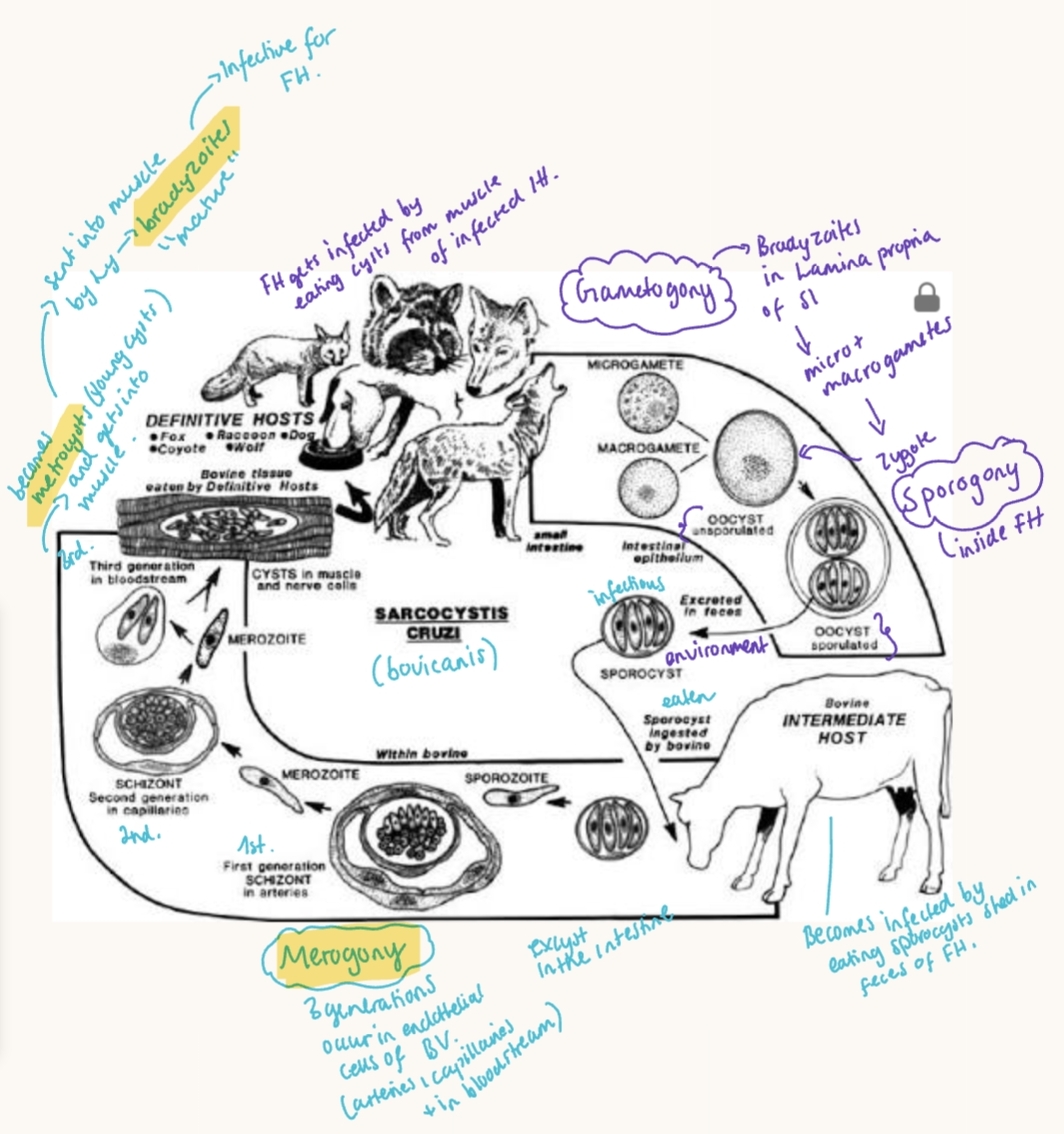
Patho/CS:
FH - usually asymptomatic, sometimes mild diarrhea. (Dog/cat)
intermediate host:
Acute/subacute infection (rare): occur after eating many sporocysts. Gets general like fever, anorexia, weight loss, decr. milk prod. abortion.
Chronic: asymptomatic
Lesions: edema, hemorrhage, necrosis (after 20-40d of infection)
Microscopical - myositis (inflammation of muscle), granulomas around cysts, mononuclear infiltration
Diagnosis:
FH: Fecal flotation (faust), detect oocyst/sporocyst. Species cannot be distinguished morphologically.
IH: muscle cyst at meat inspection, digestive method for microcyst, serology
Treatment: No therapy for IH.
FH: toltrazuril & diclazuril
Do not feed dogs raw meat
16. Equine protozoal myeloencephalitis (S. neurona)
EPM – equine protozoan myeloencephalitis (worldwide, common in USA)
Caused by Sarcocystic Neurona
Infection of CNS
FH: possum (opossum)
IH: Small mammals. Horse = aberrant host (parasite can infect and partially develop but cannot complete normal life cycle)
Location: cysts in striated muscles. In horses → brain, spinal cord.
Transmission: Horses get infected when ingesting possum feces.
Life cycle:
FH (opossum) - gametogony + sporulation. IH: eats sporocyst from environment, sarcocysts develops → which is eaten by FH.
Horse: eats the sporulated oocysts → merogony occurs (no cyst formation), meronts develop CNS.
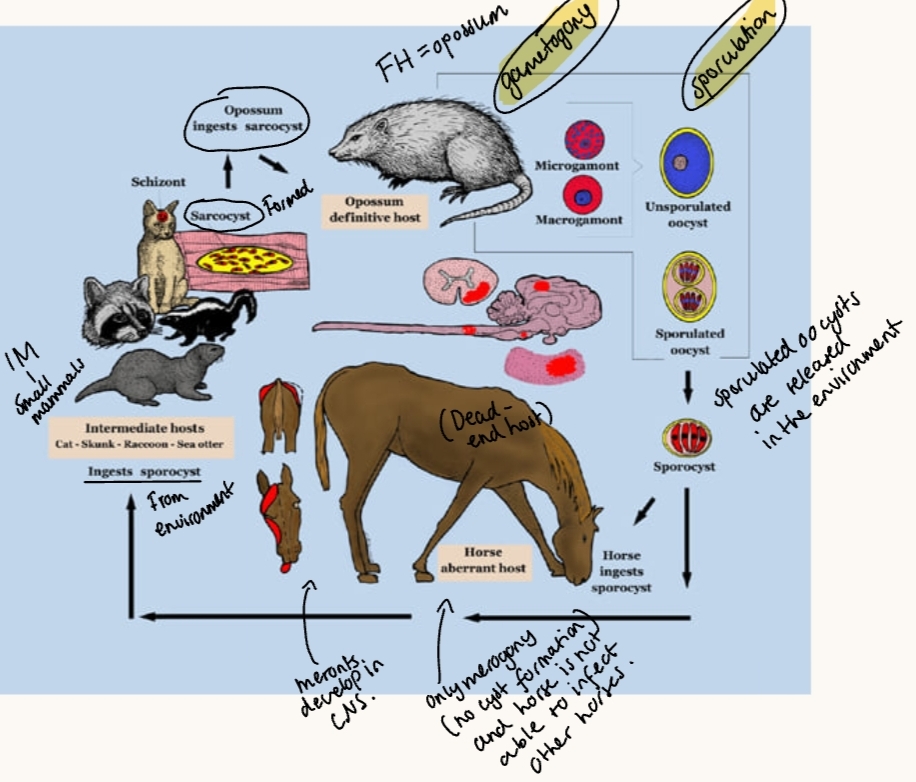
Patho/CS:
Meronts migrate to brain & spinal cord in horse → focal hemorrhage, necrosis + myeloencephalitis (inflammation of brain and cord).
CS: NS, incoordination, gait issues, asymmetric ataxia, weak, lameness, acute recumbency. Cranial signs - head tilt, facial nerve paralysis etc.
Diagnosis:
based on CS, necropsy findings, serology, PCR
Western blot on cerebrospinal fluid (CSF)
It has high seroprevalence, 50%. Many healthy horses are seropositive without clinical disease.
Treatment:
Main therapy: Trimethoprim/sulfadiazine, pyrimethamine for 4-12w.
Side effect from therapy can be leukopenia, thus give Vitamin B complex.
We may also give toltrazuril, diclazuril
Supportive - anti-inflammatory drugs, vitamin E
17. Toxoplasmosis of animals and humans
phylum: Alveolata, subphyl: apicomplexa, class: coccidea, order: eimeridae, fam: sarcocystidea, genus: Toxoplasma
Main species: T. gondii - 3 clonal strains (I,II, III). Worldwide.
Zoonotic, infects most warm-blooded animals. FH = cats/felines.
IH: mammals, birds, humans (aberrant host).
Morphology: 3 forms
Sporozoite - infective stage from oocysts
Tachyzoite - rapidly multiplying form, acute/early infection mark. Common in brain, skeletal & cardiac muscle.
Bradyzoite - slow growing dormant form, inside tissue cysts. Chronic infection mark. Located also in brain, skeletal & cardiac muscle. Resistant to low ph & Digestive enzymes. In IH.
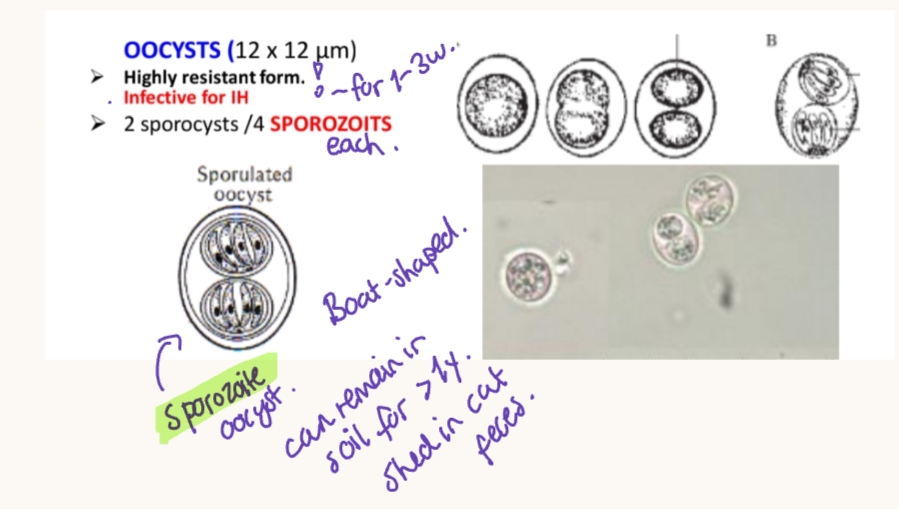
Location: Intracellular. Infect nucleated cells, not erythrocytes.
Transmission:
Broad IH range, narrow FH (cats only).
Oocysts become infective after shedding, survives in warm most soil for more than 1y. Fecal-oral route, humans get it by cat feces/litter, gardening/soil, raw veggies/fruit, undercooked meat, congenital transmission (rare in cat).
Previously infected (seropositive) cats are usually not shedding oocyst. Immunosuppressed cats may shed again, but freshly shed oocysts are not immediate infective. Direct contact with cats alone does not usually cause infection.
Life cycle: Indirect or direct (rare). Predator-prey LC.
Sylvatic (wild felids + wild IH) & domestic (bw. domestic animals)
cats: gets bradyzote → tachyzoite → merogony + gametogony
Sporulation in environment
IH - eats oocyst → sporozoites → merogony → tachyzoites → bradyzoite.
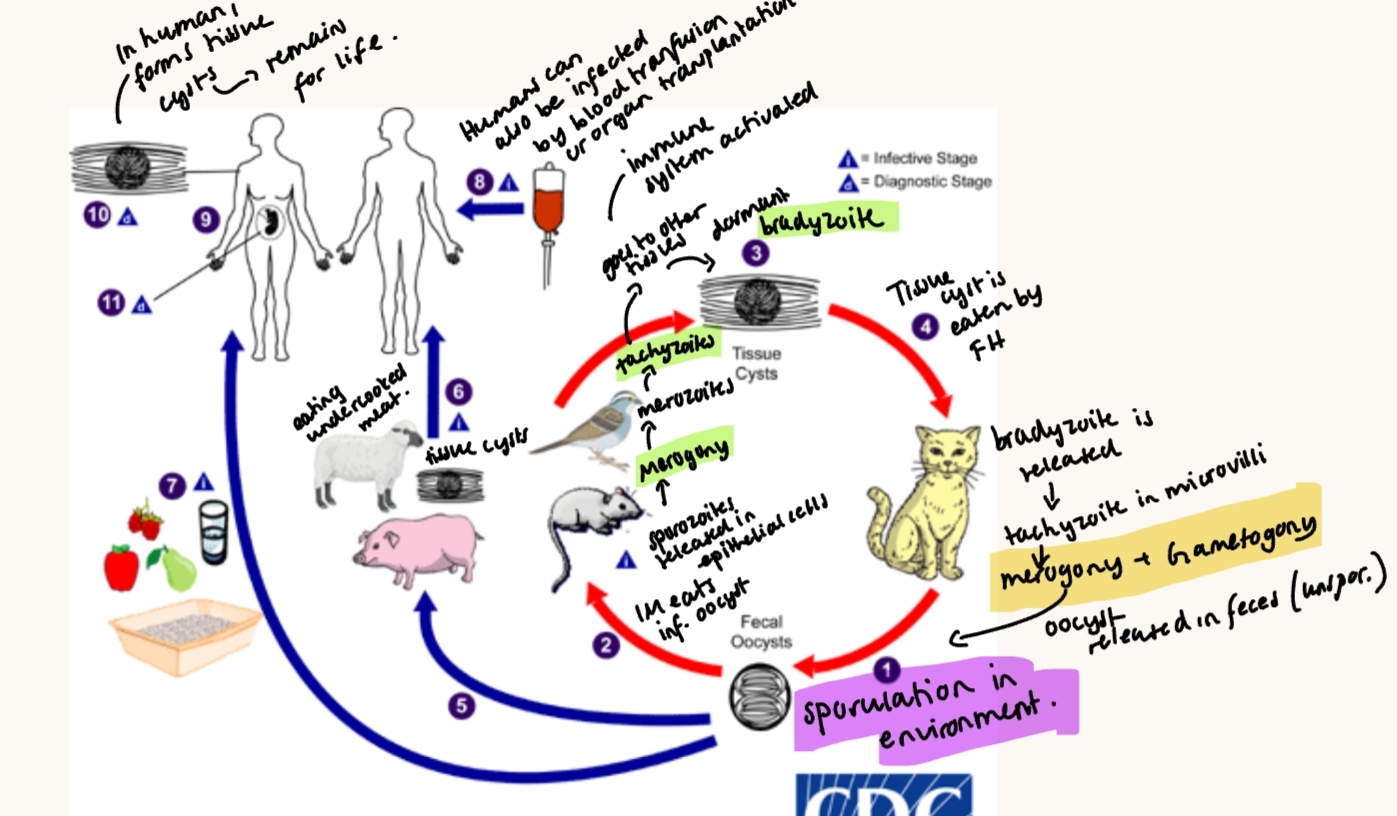
Patho/CS:
Humans: usually asymptomatic, mild flu-like illness.
Congenital toxoplasmosis: abortion/stillbirth, hydrocephalus, microcephaly, brain damage.
Post-natal infection: mononucleosis-like illness, local hypersensitivity.
Cats: usually asymptomatic.
More severe in young & immunocompromised - can be fatal.
Signs of diarrhea/enteritis, lymphadenopathy, pneumonia & dyspnea, encephalitis, weight loss, fever, retinitis.
Dogs & pigs are similar signs - acute (fever, diarrhea, pneumonia) & chronic (hepatitis, myocarditis + CNS signs like paralysis)
Large ru: Abortions, Small ru: abortions, CNS disorders
Rest - asymptomatic usually.
Diagnosis: In cats - serology - blood tests & biopsy/body fluids.
Human: multiple test + Ab monitoring. Rising Ab titer = active infection.
Treatment:
Cats: Toltrazuril, dogs = sulfonamids.
Human: Spiramycin (protect fetus in pregnant). sulfadiazine.
18.Plasmodiidosis of humans and animals.
Phylum: Alveolata, subphyl: apicomplexa, Class: Haematozoea, order: Haemosporida, fam: plasmodidae, genus: Plasmodium / Malaria
Main species:
Humans: P. vivax (most common), Falciparum (most severe), malariae, ovale, knowlesi (zoonotic)
Monkeys: P. brasilianum, central/south america
Chimpanzees: P. Scwetzi, africa
Morphology: Apicomplexa phylum - has simple nucleus, lacks flagella/cilia.
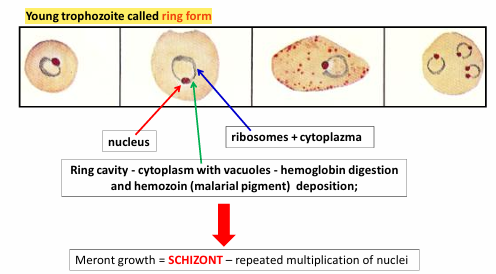
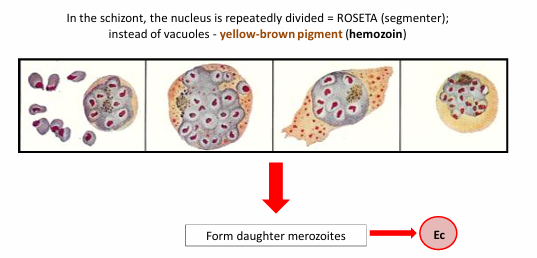
Young trophozoite = ring form. Looks like a ring inside the RBC. with nucleus, cytoplasm and ribosomes. The central clear space (vacuole) creates the ring look. It will feed on Hb → eat RBC content → produces hemozoin pigment (malarial pigment) - yellow/brown/dark as a waste product.
Location: Intracellular in RBC, liver cells and MF.
FH - mosquitoes - anopheles (in mammals)
IH: reptile, bird, rodent, monkey + man.
Geo: Worldwide, but has endemic zones in tropical areas.
Transmission: causes Malaria. Specific of host and vector. Transmitted by insect vector - anopheles mosquito (mammalian).
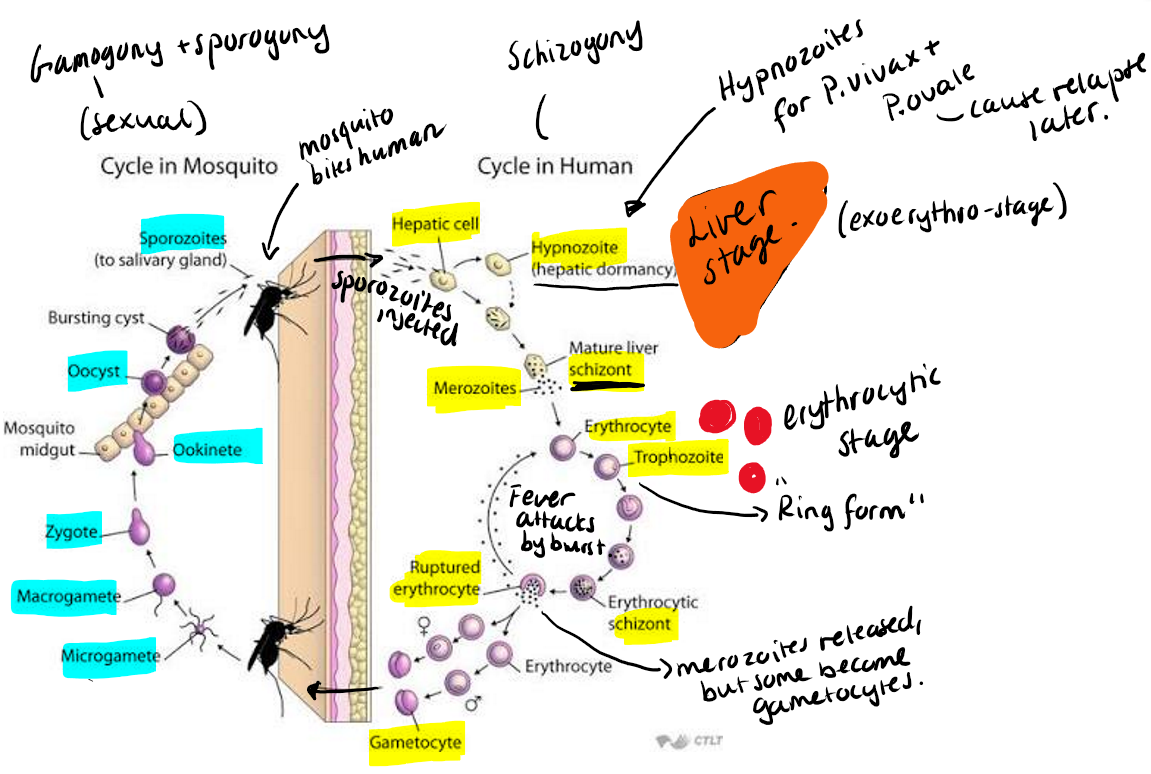
Life cycle: 2 hosts. Sexual stage in mosquito, asexual in vertebrate.
Note: when RBC ruptures → merozoite released → fever attacks “malaria attack”
46h cycle in falciparum, 72h in malariae.
microgamont → makes 8 flagellated microgametes (sperm-like cells).
No sporocyst structure, the sporozoites develop inside oocysts
Process: in mosquito → micro + macro gamete - gametogony → oocyst → sporozoites inside the oocyst → inject into human → merogony -hypnozoites for P.vivax + ovale → merozoites → trophozoites (ring form) → gametocytes formed.
Patho/CS:
Early signs - general flu-signs, fever, headache, nausea, diarrhea.
Classic malaria signs: periodic fever attacks, chills/sweating, anemia, thrombocytopenia & hepatosplenomegaly.
Severe: renal dysfunction, mental status changes, cerebral malaria (P.falciparum)
Monkeys - asymptomatic or mild
Immunity to malaria develops gradually after infections, can be transferred from mother to child (3-6 months protection).
Diagnosis: Stained blood smear, PCR, ELISA, IFAT
Treatment: Doxycycline, Malarone (also as prophylaxis)
19.Avian malaria (Hemoproteus, Leucocytozoon, Plasmodium).
class: haematozoea, order: Haemosporida
Families:
Plasmodidae → plasmodium
Haemoproteidae → haemoproteus
Leucocytozoidae → leucocytozoon
All are blood coccidia, apicomplexan parasites. Intracellular - blood cells, mainly infects birds, vector borne + heteroxenous cycle (2 hosts). Similar to Human Malaria.
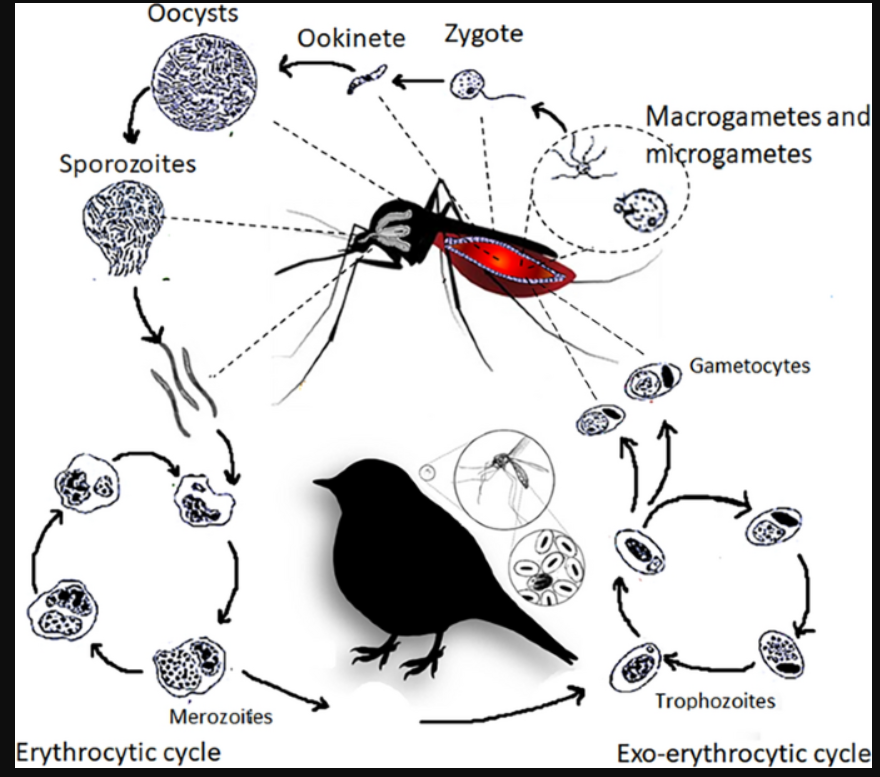
LC (similar to malaria)
vector: gametogony + sporogony, injects sporozoites in host
Host: Merogony → trophozoites + gametocytes → then the vector gets them.
Haemoproteus - NO merogony in RBC, only gametocytes.
Leucocytozoon - meront damage liver/tissues, megaloschizonts rupture → hemorrhage.
Clinical signs shared: often asymptomatic, anemic, weak/depressed, organ enlargement + cause death in severe infections.
Diagnosis: Blood smear microscopy, finding gametocytes in blood cells.
Transmission: biting insects (as DH/vector)

1) Plasmodium
Species:
P. relictum → passerine/pigeons
P.gallinaeceum → chickens
P.cathemerium → passerine birds
Many live in birds (their specific host) without making them sick, but cause disease in non-native birds.
Patho/CS:
Replication inside RBC → rupture → anemia + hypoxia
Signs: intermittent fever, splenomegaly, anemia
2) Haemoproteus
Species:
H. meleagridis – Turkey
H. nettionis – water poultry
H. columbae – pigeon (pigeon malaria)
Patho/CS: Endophlebitis (capillary inflammation), RBC destruction (anemia), Hemozoin pigment accumulation → chocolate colored organs.
Diagnosis: Finding only gametocytes inside RBC.
Treatment/control: Chloroquine
3) Leucocytozoon
L. smithi (turkey)
L. simondi (duck, geese)
L. caulleryi (chicken)
Patho/CS: schizonts damage liver/tissues. Megaloschizonts rupture → hemorrhage. Usually asymptomatic but severe disease cause paralysis/inability to fly and sudden death in young.
Granulomas in lung, heart, brain, nerves
DG: Blood smear - large gametocytes, no pigment, distorted RBC, WBC.
Histopathology - megaschizonts
Treatment: poor response. We can give pyrimethamine, clopidol.
20.Babesiosis in ruminants and horses.
Phylum: Alveolata, Subphylum: Apicomplexa, Class: Haematozoea, Order: Piroplasmida, Family: Babesiidae, Genus: Babesia
Host/IH: Cattle, eq, small Ru, dog/humans for some.
Vector/FH: Ticks - Boophilus anulatus
Main species - worldwide.
Cattle: B.bovis (severe - Texas Cattle fever, small), B.bigemina (milder, large)
B.major (large), B.divergens (small)
Equids: B.equi (=now Theileria equi), B.caballi
Sheep/goat: B.ovis & B.motasi
Zoonotic species: B.divergens, B.bigemina, equi, microti.
OIE-listed diseases: bovine babesiosis, equine piroplasmosis - B.equi.
Morphology: Pear-shape, round/oval forms. Mainly inside RBCs, no sporocyst stage, free sporozoites present.
Location: RBCs, some in Ly, histiocytes + erythroblasts
Transmission: Vectors - ticks. Transtadial & Transovarial in vector.
Life cycle: Vector/Tick: Gametogony & Sporogony. IH: Merogony/binary fission inside RBCs.
Patho/CS: Parasites invade and multiply inside RBCs → destruction (hemolysis) → hemolytic anemia. General: high fever, hemoglobinuria (red urine), weak, pale MM, tachycardia, dyspnea, anorexia.
B.bigemina (milder): Hemoglobinuria + Hemoglobinemia, stands isolated from herd, arched back.
B.Bovis (Severe - cerebral form): CNS disease, Parasites in RBCs - blocks brain capillaries → thrombi/emboli → brain ischemia. Typical signs are ataxia, incoordination, teeth grinding, mania, collapse, paddling. Sudden death. Pink/red brain (packed with infected RBC).
B. equi: Dies within 24-48h. Severe anemia, jaundice, rapid.
Severity: much worse in adult cattle - death within a week of infection in B.bigemina. Cattle that recover is immune for life.
Humans: B.divergens - not many cases 50% fatal. B.microti - with mice, vector ixodes.
Diagnosis: Giemsa-stained blood smear, PPM brain lesions, serology.
Humans: blood smear, IFA, molecular methods.
Treatment: Diminazine, regular dipping, vaccines for animals.
Humans: clindamycin + quinone
21.dog piroplasmosis (Babesia, Hepatozoon).
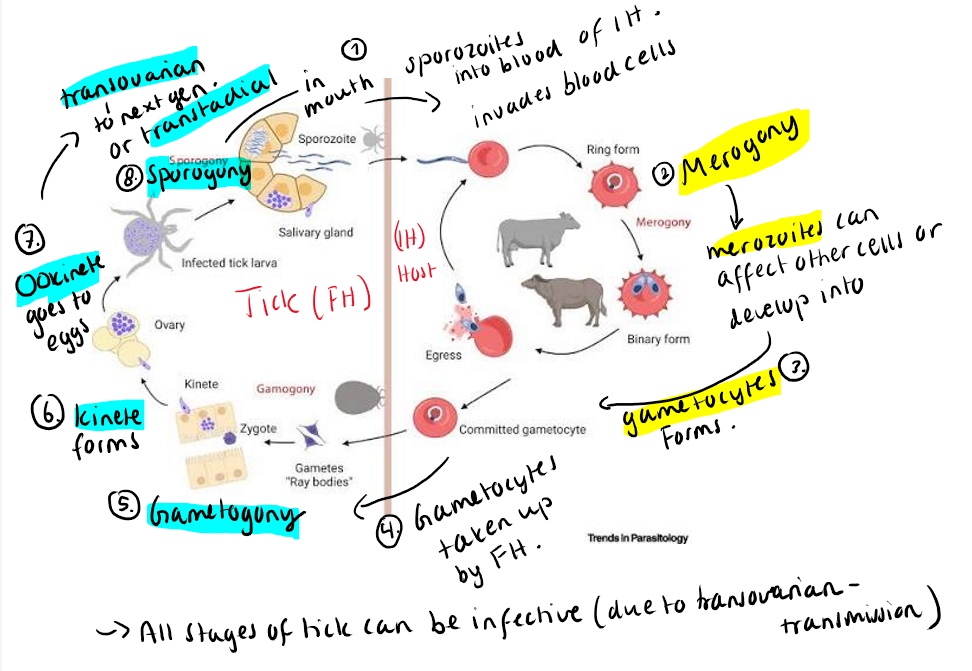
Both belong to Apicomplexa subphylum, under Alveolata. TIck-related LC.
LC: Vertebrate host (Merogony). Tick Vector (Gametogony & Sporogony)
Dg: blood smear (microscopy), Serology/PCR confirmation.
1) BABESIA
class: haematozoea, order: piroplasmida, fam: babesiidae (babesia).
Blood parasite of IH, some in leukocytes or other blood cells. Binary fission - reproduction in vertebrate host.
Vertebrate host: cattle, domestic animals, sometimes human.
Main species:
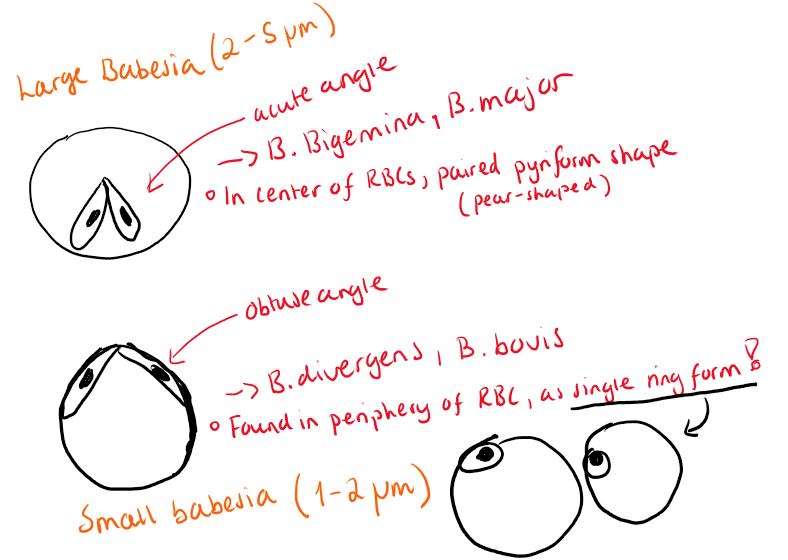
Species + vector ticks of large babesia (2-5um, pyriform/pear-form, paired in RBC)
B. canis canis - dermacentor reticulatus (europe, asia)
B. canis vogeli - Rhipicephalus sanguineus - often without CS, but fatal in puppies (USA, tropical + subtrop. areas)
B. canis rossi - haemophysalis elliptica (formerly leachi) (most patho! south africa). Causes severe anemia, shock, high mortality
Species and vector ticks of small babesia (1-2um, single ring form)
B. canis gibsoni - rhipicephalus/haemaphysalis (Asia, Europe, USA)
B.microti-like isolates (also known as B.vulpes and T.annae).
Geographic: Worldwide, but Europe has the highest of cases for dogs (rare in human cases).
LC:
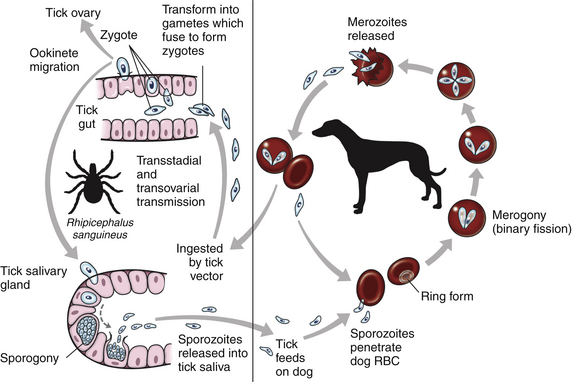
Patho/CS:
Invades RBC → multiples → rupture → intravascular hemolysis, immune-mediated destruction, oxidative damage.
Hemolytic anemia, fever, weak, anorexia, pale MM, jaundice, hemoglobinuria, splenomegaly, sometyimes neurologic signs.
Acute = severe, rapid, Chronic = mild, intermittent signs.
Diagnosis: Blood smear (Giemsa/quick stains) - low sensitivity but fast. Serology (ELISA/IFAT) - exposure not active disease. PCR - most accurate.
Treatment: Imidocarb, diminazene aceturate, trypan blue. Supportive, tick control.
2) HEPATOZOON
class: coccidea (new: conoidasida), order: Adeleida, fam: hepatozoidae, genus: Hepatozoon
Main species: Hepatozoon canis (old world), H. Americanum (new)
Morphology: more elongated.
Location: Gametocytes in circulating leukocytes and tissue “cysts” in muscle.
FH/Vector: Rhipicephalus sanguineus (old world), Amlyoma maculatum (new world)
IM: Dogs
Transmission: Ingestion of Infected tick. NOT by bite.
Life cycle: Dog eats tick → parasite released → merogony → gamonts in leukocytes → taken up by tick again. Gamonts in WBC for H.canis.
H.americanum → muscle cysts. by eating tick, cysts forms in skeletal muscle, merogony within → merozoites released, pyogranuloma.
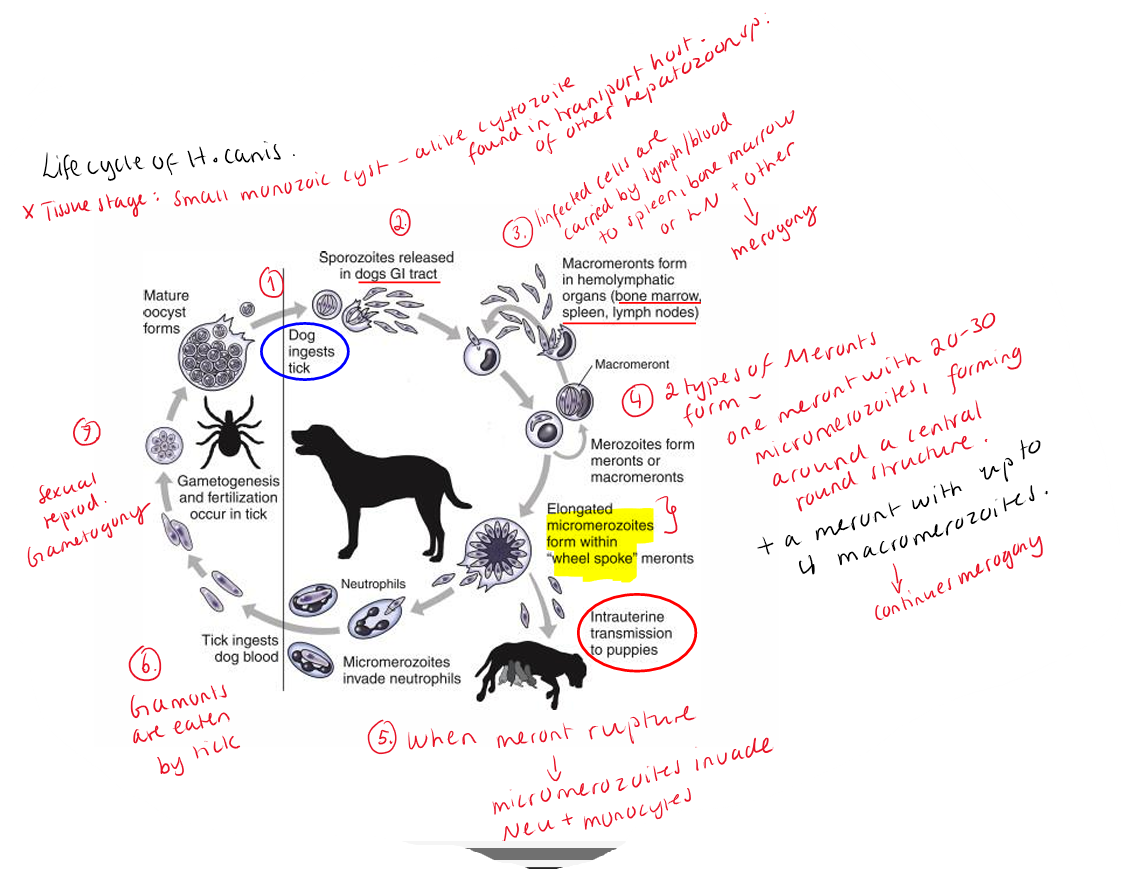
Patho/CS: Dogs are often asymptomatic, unless infected with more than one pathogen at same time, immunosuppressed or younger than 4months.
Severe cases - muscle pain/stiffness.
Fever, weight loss, lethargy, marked leukocytosis (high WBC).
Severe - H.americanum - onion skin muscle lesions
Diagnosis: blood smear - detecting micromerozoites in neu. Gamonts in leukocytes. Muscle biopsy (cysts), PCR (confirmation).
Treatment: doxycycline, toltrazuril.
22.Theileriidosis of mammals.
alveolata, subphyl: apicomplexa, class: haematozoea/Aconoidasida, order: piroplasmida, fam: theileriidae, genus: theileria.
Cattle species:
T.parva → east coast fever (Africa, very severe)
T.parva parva (bw. cattle, less patho)
T.parva Lawrencei (buffalo→cattle, most patho)
T.parva bovis (milder)
T.annulata → tropical theileriosis (Asia, Africa)
Small ru spp:
T.lestoquardi (very high mortality)
T.ovis (usually non patho)
Others: T.equi, wild ruminant theileria ex. mutans
Location: Intracellular, first leukocytes (Ly, MF), later RBCs
Vector/FH: Ticks - Rhipicephalus appendiculatus (T.parva), Hyalomma (T.annulata/T.lestoquardi)
Transmission: Tick bite + Transstadial transmission by ticks (bw generations)
Life cycle: Transformation of leukocytes = main idea.
Tick injects sporozoites into host → infects leukocytes → develop into meronts (Merogony) → cause host cell transformation & uncontrolled proliferation → merozoites released → invade RBCs → gamonts → ticks suck blood → gamogony → sporogony → sporozoites in salivary gland of tick.
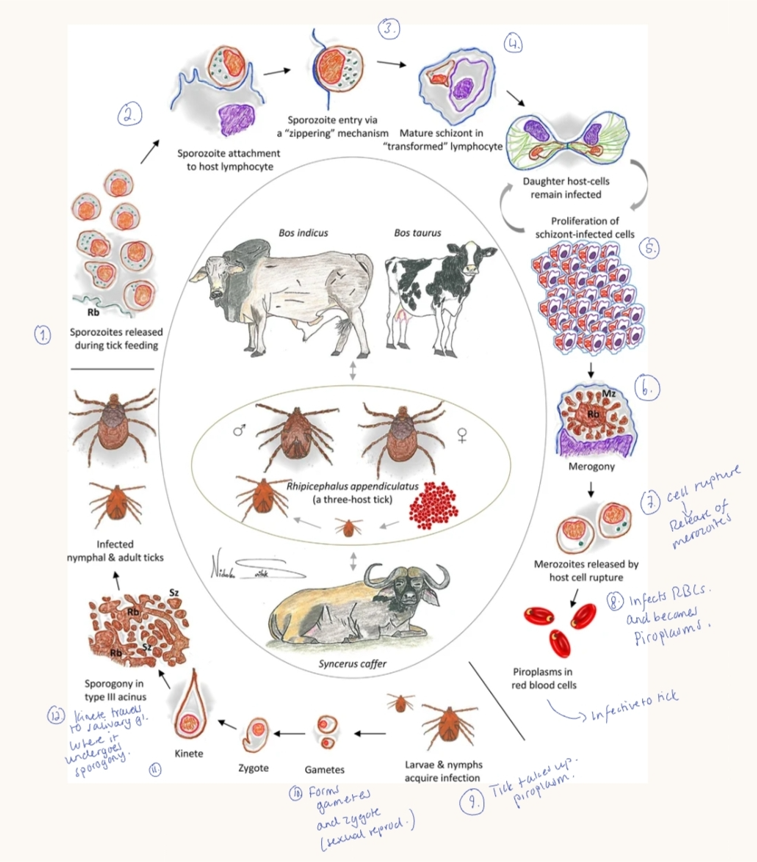
Patho/CS: infection in wild animals - mostly asymptomatic
Transforms Ly (cancer-like proliferation of infected cells).
Main effects: Massive LN enlargement, fever, systemic inflammation, immune suppression + tissue damage.
T.Parva: early high fever, enlarged hemorrhagic LN, severe resp. signs (lung edema), weight loss, terminal drop in temp. before death - dies in 20d.
T.annulata: hemoglobinuria, high mortality
PM: severe lung edema and intersitital pneumonia, splenic infarcts, thrombosis, hyperemia.
Diagnosis:
Blood smear (giemsa) → RBC piroplasms, LN smear → schizonts, PCR, Serology (IFA). problem - difficult to distinguish species microscopically.
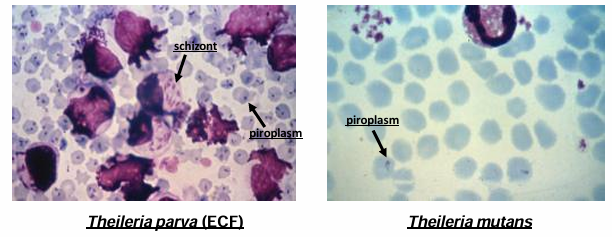
Treatment: T.parva - Halofuginone. Tick control.
23.Balantidiosis of pigs.
phyl: ciliophora, class: litostomatea, order: vestibulferida, fam: balantidiidae (only one genus), genus: balantidium
Main species: Balantidium coli (Zoonotic ciliate protozoan)
Found in intestinal tract of arthropods (ex. bee, ant), Some vertebrates, pathogen of human, pigs, monkeys.
Morphology:
nuclear dimorphism = 2 nuclei
micronucleus → genetic functions, elongated, kidney-shape
Macronucleus → somatic functions, spherical
2 development stages:
Cyst (non-replicating form, infective)
trophozoite (active, pathogenic form) - repr. by transverse division.
Location: LI, lives in the opportunistic protozoa.
FH: Pigs, wild boar. Accidental host = humans, primates. Pigs are usually asymptomatic carriers.
Humans - rare infection, less than 1% but potentially severe.
Most often in tropical regions.
Transmission: Fecal-oral, zoonotic.
Life cycle: Direct - one host needed.
Asexual reprod. by transverse division. Sexual by conjugation can occur.
Eats cyst → excystation in intestine → trophozoites colonize LI → multiplies by binary fission → encystation in colon → cysts excreted in feces → survive months in environment.
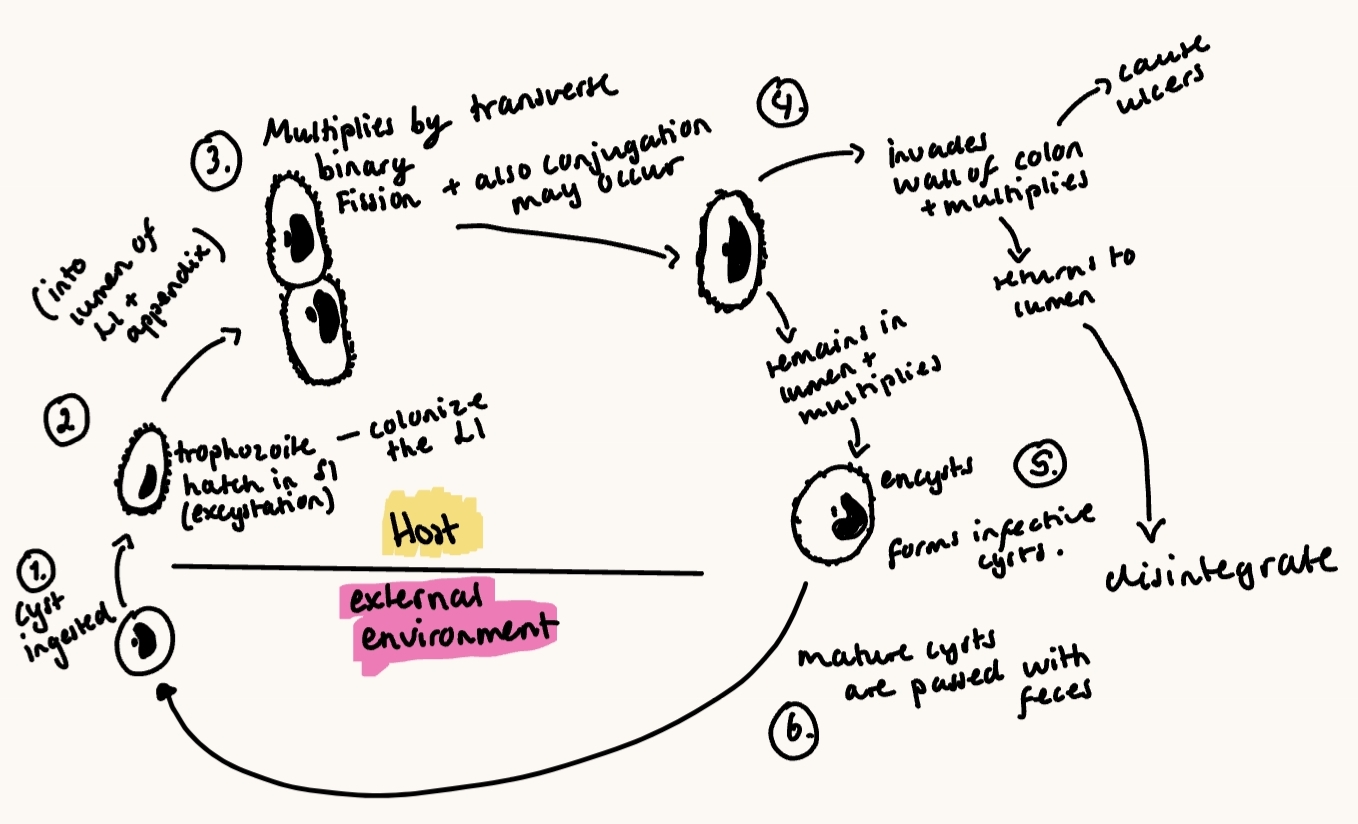
Patho/CS: Usually asymtpomatic (esp. for pigs).
when pathogenic - invades colon mucosa → colitis, can lead to hemorrhage.
severe - diarrhea, dysentery (blood + mucus) → can die. can spread to liver, lungs.
chronic carriers after infection.
Diagnosis: microscopy of feces (cyst - flotation, trophozoites in acute diarrhea - wet smear), tissue biopsy - colon, staining - lugols, PM
Treatment: Metronidazole. Effective ATB therapy in human/animals.
24.Encephalitozoonosis and Nosematosis
Both diseases are caused by Microsporidia (phylum).
intracellular, spore-forming, small size (difficult to detect), lacks mitochondria, infect host cells using a polar tubule (polar filament) to inject infective material.
Same LC: infection starts with spore ingestion → germinates → polar tube injects sporoplasm into host cell → asexual multiplication (merogony) → sporogony → mature spores → host cell ruptures, spores released → new infections.
Transmission: fecal-oral, environmental contamination with sproes, direct spread (no IH).
Intracellular replication → cell destruction, chronic, often worse in immunocompromised host, can involve multiple organs, depending on species.
Dg: microscopy (stains), PCR, TEM (gold standard). Difficult species ID without molecular tools. Treatment options limited but include albendazole/fenbendazole or fumagillin for nosematosis.

1) Encephalitozoonosis
Fungi - Phylum: Microsporidia, fam: unikaryonida (encephalitozoonidae before), genus: encephalitozoon.
Main species:
Encephalitozoon cuniculi (zoonotic)
E.hellem, E.intestinalis (wide host range)
Geo: Cosmopolitan extension cases on all five continents. In free living wildlife on all continents - asia, america, africa Eu.
E. cuniculi - encephalitozoonosis of rabbit:
Infects rabbits, humans. Zoonotic potential.
Cause systemic infection - kidneys (renal failure), CNS, eyes (uveitis, cataracts). Cause granulomas & pseudocysts.
signs: NS (head tilt, ataxia), kidney disease, eye lesions, weight loss. chronic/fatal in rabbit/immunocompromised.
Transmission: transplacental in rabbits, by urine, oral.
Life cycle of E.cuniculi:
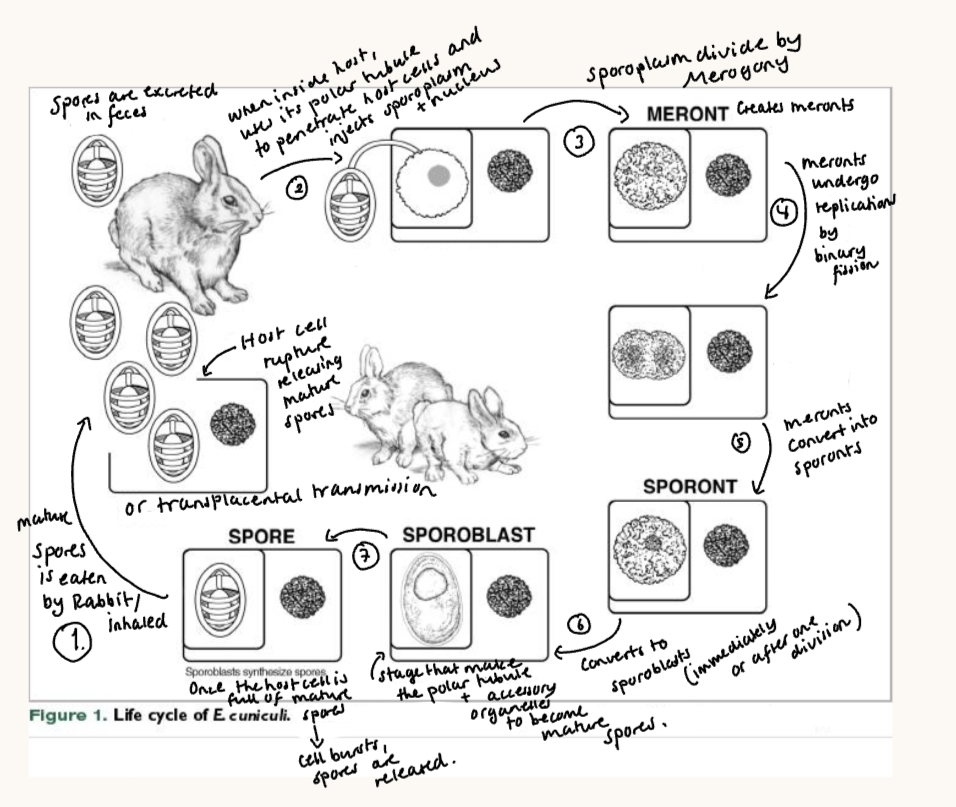
Encephalitozoonosis of dogs:
opportunistic, usually only cause disease when the dog`s immune system is weak. puppies can get it from their mother (transplacental).
Diagnosis - difficult, Ab can be found in even healthy dogs. Spores can be found in urine. PM - granulomas in tissue (histology).
in other animals - often cause no symptoms.
2) Nosematosis (in honey bees)
Fungi - microsporida, fam: nosematidae, genus: Vairimorpha (syn: nosema)
V. ceraneae (no obvious signs) & V. apis (dysentery). In Europe, Asia, America.
restricted to midgut epithelium → digestive dysfunction, reduced lifespan, colony collapse effects in bees. It cause change in sugar metabolism & hormone levels.
CS: bee colony-level - weak growth, dysentery (V.apis), reduced foraging & behavior change, queen laying reduction.
Dg: bee gut microscopy, PCR, LAMP, DNA testing.