General Chem Things
1/30
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
31 Terms


Gas Laws (not on sheet)
(P1)(V1) = (P2)(V2) - Boyles
(V1)(T2) = (V2)(T1) - Charles
(P1)(T2) = (P2)(T1) - Gay Lussac
(V1)(n2) = (V2)(n1) - Avogadro
Percent Composition
(part/whole)x100
Empirical formula
% to grams
grams to moles
divide by small
multiply until whole
Percent Yield
(actual / theoretical) x 100
Solubility Rules
SNAP
Compounds containing sodium, nitrate, ammonium, potassium are ALWAYS soluble
Formal Charge
Valence - lone pairs (EACH ELECTRON COUNTS AS ONE (1 lone pair = 2!!!!!)) - bond (each bond counts as their own (ex. double bond = 1))
Valence - (Dots + Lines)
Coulombs Law
Small radius and big charge = strong attraction
Henderson Hasselbalch
pH = pKa + log ([A-]/[HA])
[A-] = conjugate base
[HA] = weak acid
Faraday’s Law
q = I * t
(remember as quit)
Strong Acids
So I Brought No Clean Clothes
H2SO4
HI
HBr
HNO3
HClO3
HClO4
Delta G equations
Standard conditions: Delta G = Delta H - T*Delta S
Nonstandard conditions: Delta G = Delta G + RTlnQ
Thermo R = 8.314
Equilibrium: Delta G = -RTlnK
Electrochemistry: Delta G = -nFE
n = moles
F = Faraday’s constant (96485)
E = cell potential
MAKE SURE DELTA S, H, G ALL HAVE MATCHING UNITS!!!!!!!!!!
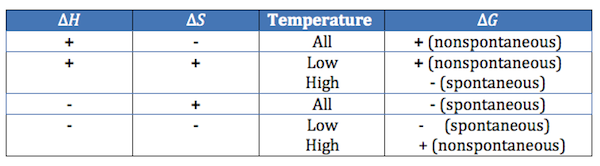
Spontaneous Conditions
Think: Both positive H and S = HIGH temp and both negative = LOW temp
positive = high
negative = low
Rate Law
rate = k [A]n[B]m
Electrochemistry
Ecell = Ecathode - Eanode
Electrolytic Cell
HIGHER E VALUE IS FLIPPED!!!!!
(lower E value is flipped in Galvanic)
Enthalpy change (delta H)
Delta H = reactants - products
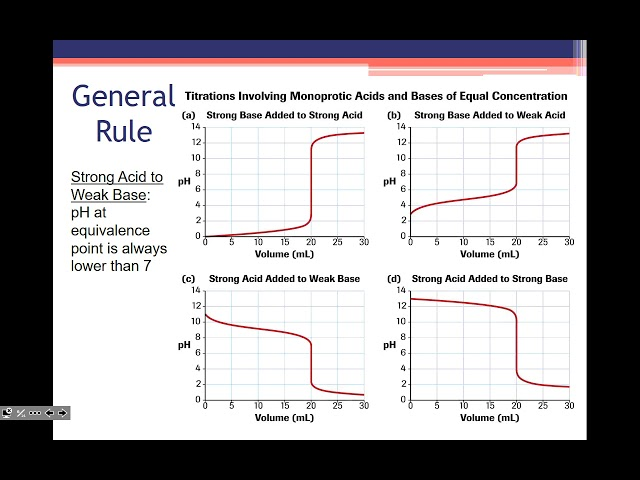
Acid/Base Titration Curves
Strong Acid + Strong Base
S shaped
pH=7 @ equiv point
Low starting pH
Strong Acid + Weak Base
pH<7 @ equiv point
Higher starting pH
Weak Acid + Strong Base
S shaped
pH>7 @ equiv point
higher starting pH
Weak Acid + Weak Base
gradual
Linear
2 electron pairs
0 lone pairs
linear electron geometry
180 bond angle
sp hybridization
Trigonal planar
3 electron pairs
0 lone pairs
trigonal planar electron geometry
120 bond angle
sp2 hybridization
Bent
3 electron pairs
1 lone pair
trigonal planar electron geometry
119 bond angle
sp2 hybridization
Tetrahedral
4 electron pairs
0 lone pairs
tetrahedral electron geometry
109.5 bond angle
sp3 hybridization
Pyramidal
4 electron pairs
1 lone pair
tetrahedral electron geometry
107.5 bond angle
sp3 hybridization
Bent
4 electron pairs
2 lone pairs
tetrahedral electron geometry
105.5 bond angle
sp3 hybridization
Trigonal bipyramidal
5 electron pairs
0 lone pairs
trigonal planar electron geometry
120, 90 bond angle
sp3d hybridization
See-saw
5 electron pairs
1 lone pair
trigonal planar electron geometry
119, 90 bond angle
sp3d hybridization
T-shaped
5 electron pairs
2 lone pairs
trigonal planar electron geometry
90 bond angle
sp3d hybridization
Linear
5 electron pairs
3 lone pairs
trigonal planar electron geometry
180 bond angle
sp3d hybridization
Octahedral
6 electron pairs
0 lone pairs
octahedral electron geometry
90 bond angles
sp3d2 hybridization
Square pyramidal
6 electron pairs
1 lone pair
octahedral electron geometry
90 bond angle
sp3d2 hybridization
Square planar
6 electron pairs
2 lone pairs
octahedral electron geometry
90 bond angle
sp3d2 hybridization