Immunology New Material for Final Exam (Dr. Ekesi)
1/127
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
128 Terms
TCR can recognize antigens but cannot signal on its own, why?
short cytoplasmic tails on the alpha and beta chainsv
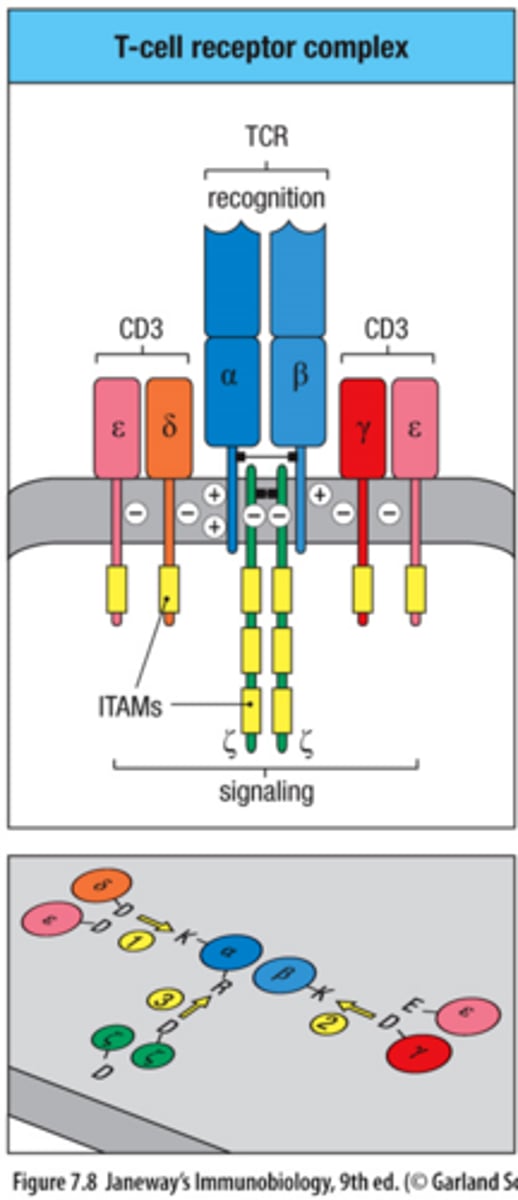
TCR signaling is mediated by what?
CD3 complex (γε dimer, δε dimer, ζζ dimer)
CD3 chains contain __________?
ITAM motifs; ITAMs become phosphorylated during activation; ITAMs recruit intracellular signaling proteins
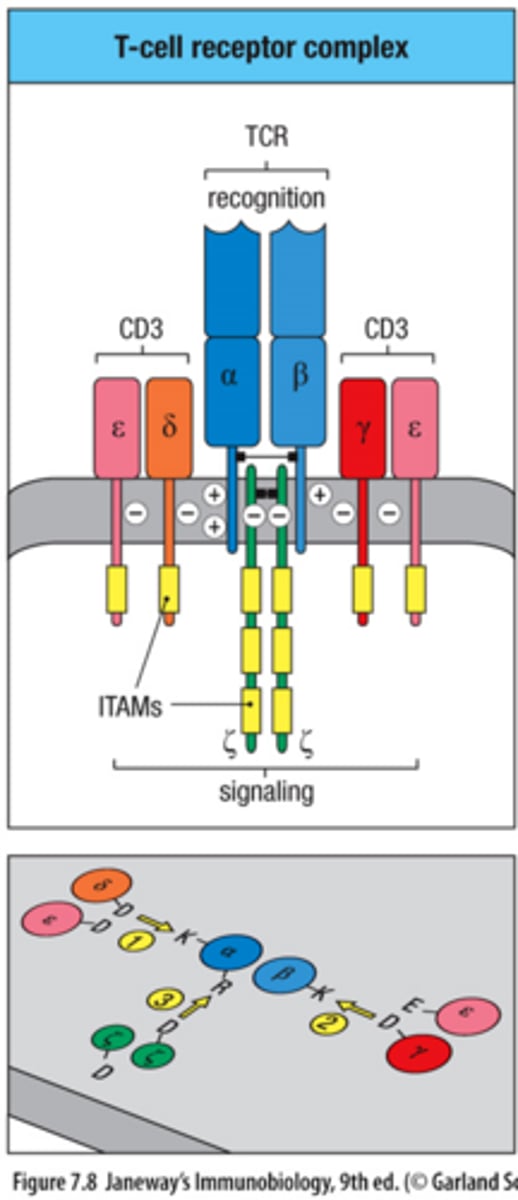
CD4 and CD8 function?
Co-receptors; CD4 binds MHC II and CD8 binds MHC 1
What is recruited by CD4/CD8 that phosphorylates CD3 ITAMs?
Lck kinase; phosphorylates CD3 ITAMs for signaling
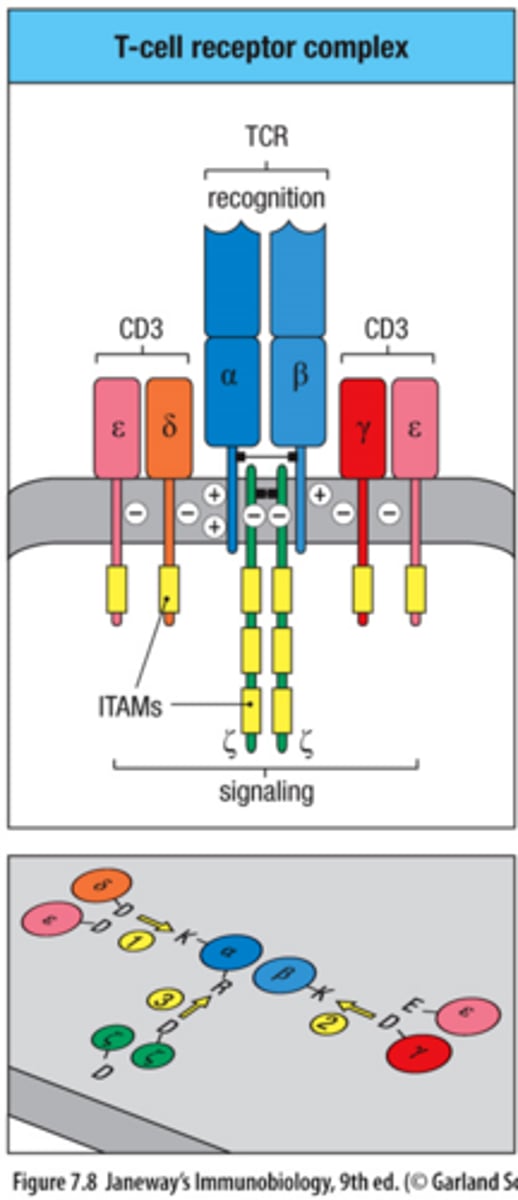
Activation of Naive T cells requires 3 signals:
1. TCR signaling
2. Costimulatory sinaling
3. Cytokine signaling (IL-2)
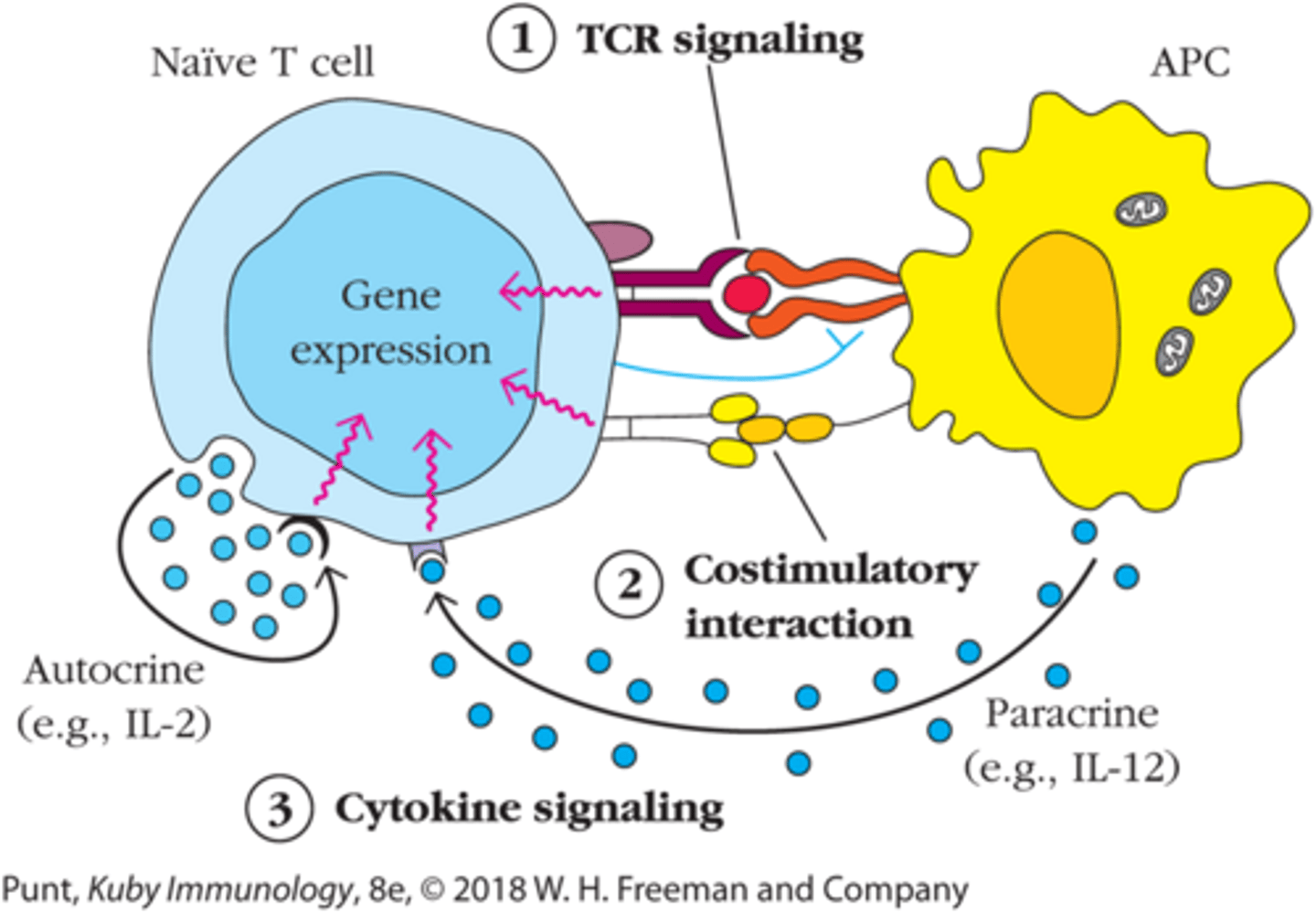
TCR signaling alone is insufficient for full activation, but what happens if only TCR signaling occurs?
leads to anergy or deletion (depending on context)
Co-stimulatory signal molecule?
CD28 (t cells) and B7 (dendritic cells); signal = CD28 + B7-1 or C7-2 (CD80 or CD86)
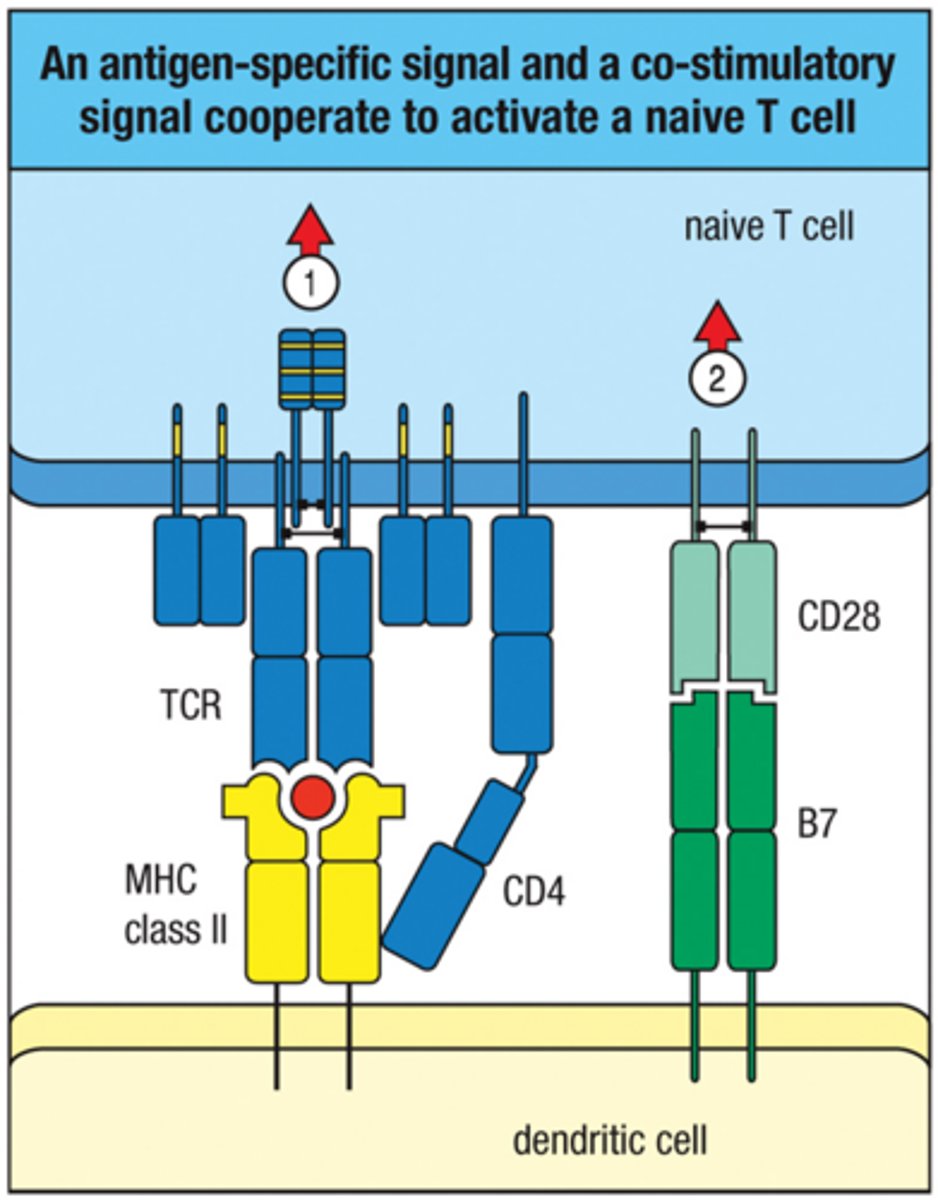
What induces co-stimulatory molecules?
PRR recognition of PAMPs and DAMPs
Absence of signal 2 (Costimulatory signaling) lead to? (T cell activation)
anergy and peripheral tolerance
Co-stimulatory signaling ensures T cell activation occurs only in context of _________ or _________
infection or danger
APCs most effective for T cell activation?
Dendritic Cells
Dendritic cells perform antigen uptake through?
phagocytosis and macropinocytosis
How does Dendritic cell maturation happen?
PRR activation
Dendritic cell maturation causes what?
increased MHC I and MHC II expression, increased CD80/CD86 expression, and decreased antigen uptake
What receptor do Dendritic cells use to migrate to the lymph nodes and what chemokines does the receptor responds to?
CCR7; CCL19 and CCL21
Transcription Factors that turn on expression of IL-2
NFAT, NFkB, and AP-1
T-Cell - APC Immunological Synape
almost forms a pestle-mortar structure so that products expressed do not diffuse out aimlessly
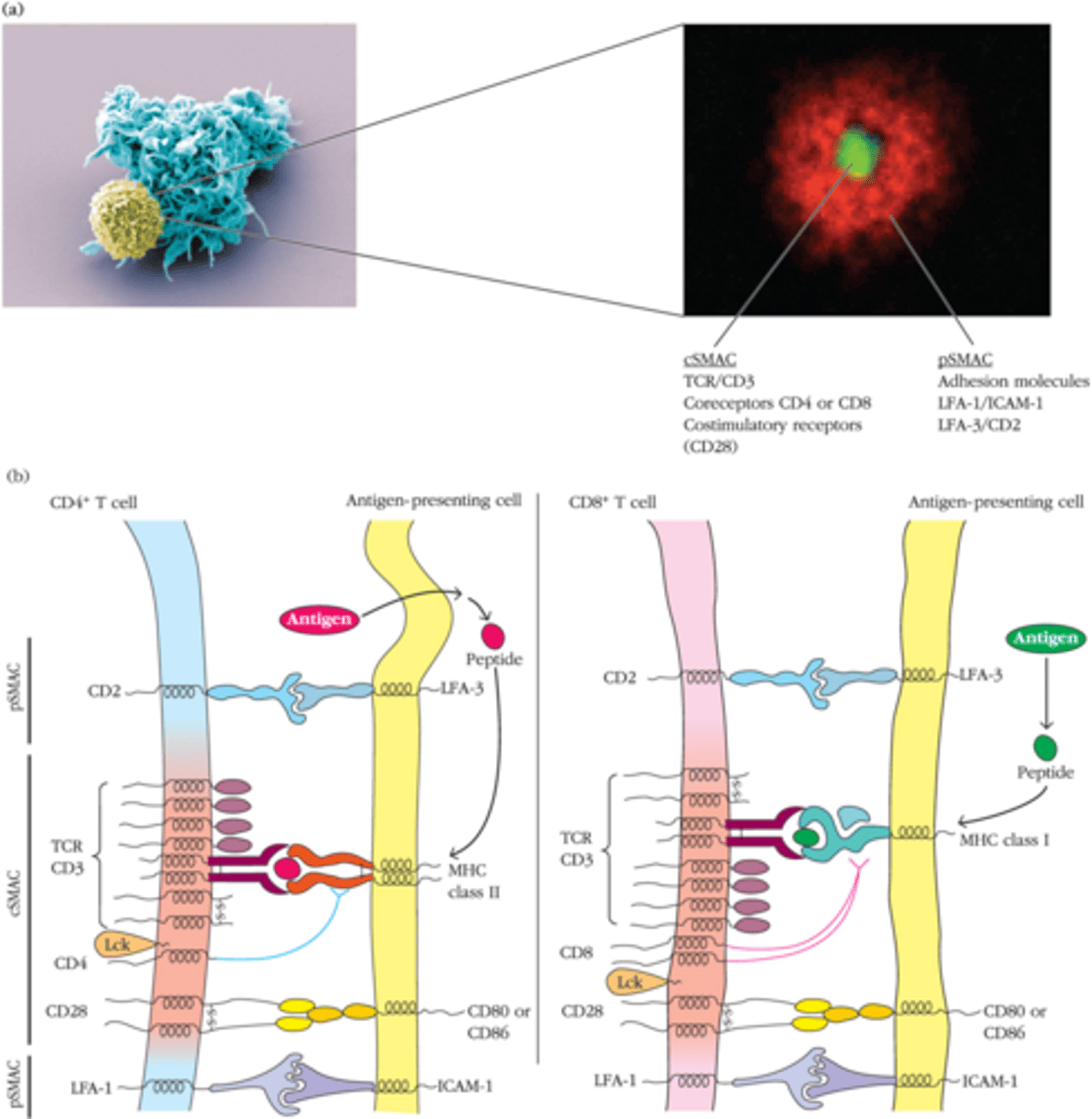
Immunological synapse stabalizes T cell-APC interactions and enhances
signaling efficency
cSMAC (central supramolecular activating complex)
TCR, CD3, CD28, and signaling molecules
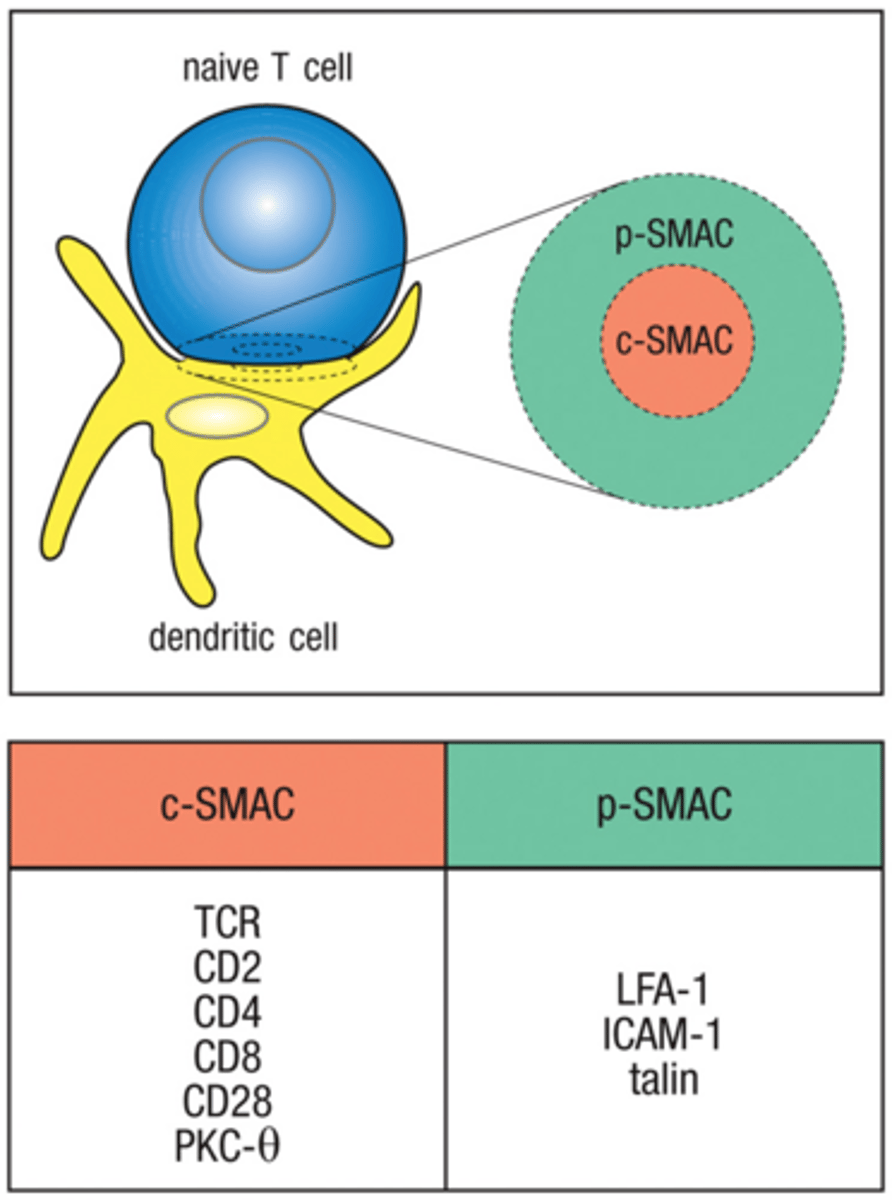
pSMAC (peripheral supramolecular activating complex)
LFA-1 and ICAM
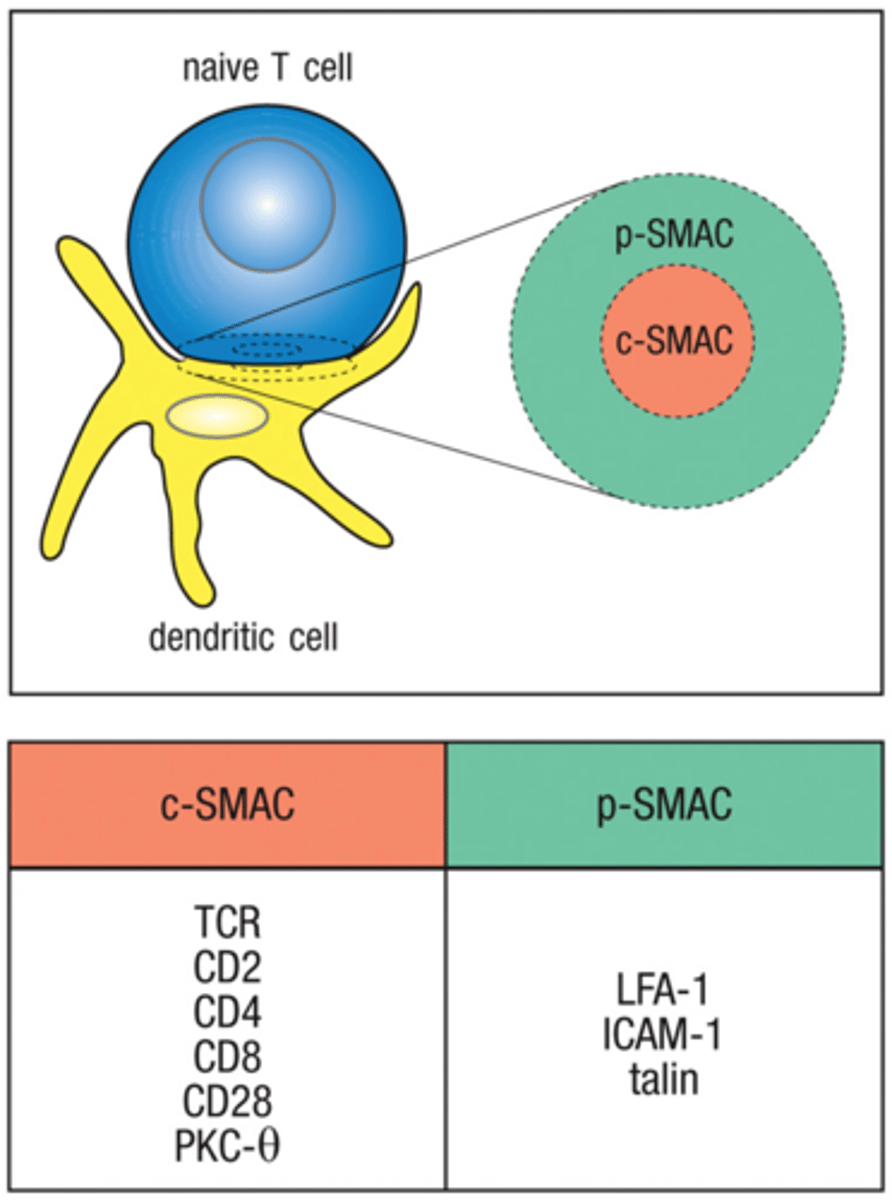
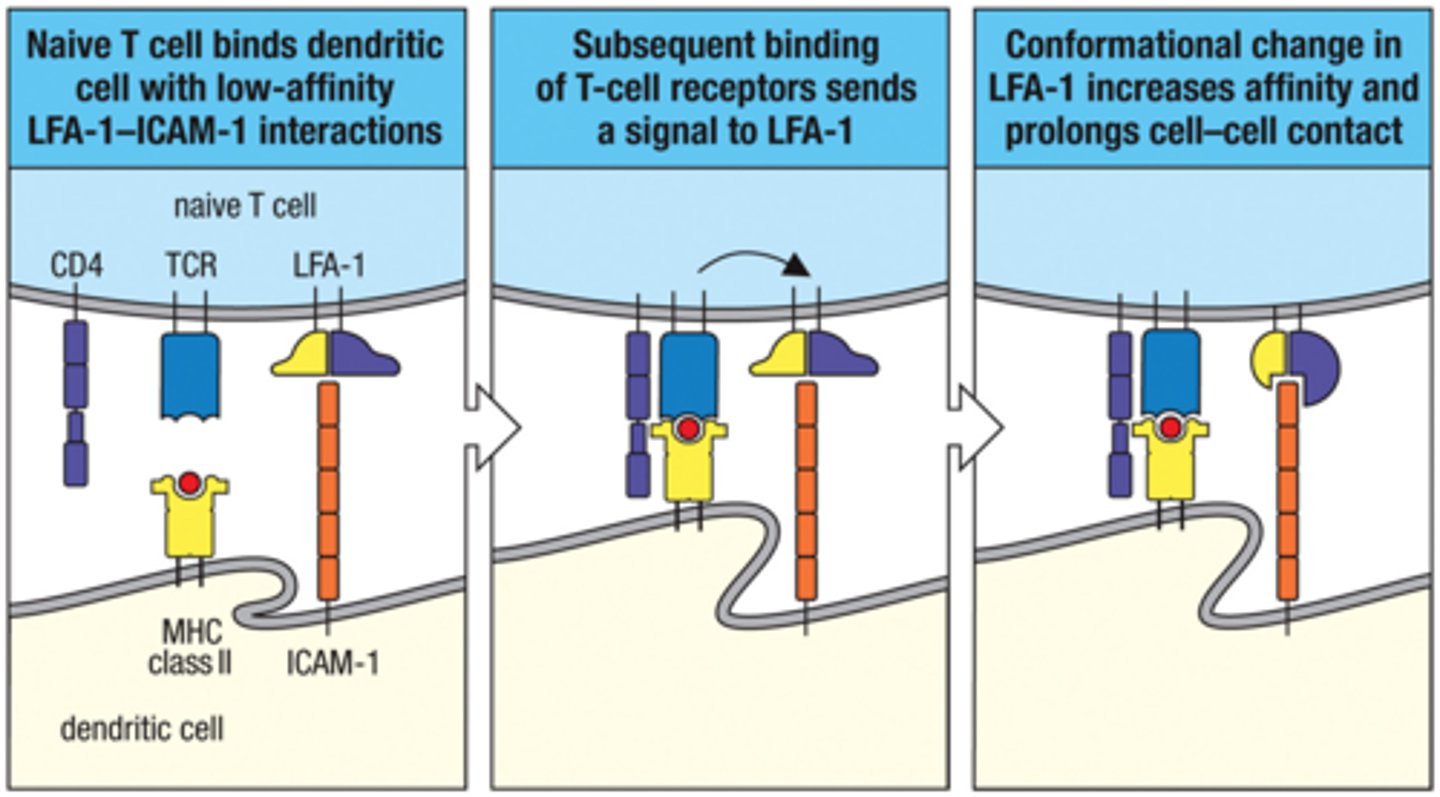
How does Immunological synapse form?
1. naive T cell bindd dendritic cell with low-affinity LFA-1-ICAM interaction
2. Subsequent binding of T-cell receptors send a signal to LFA-1
3. Conformational change in LFA-1 increases affinity and prolongs cell-cell contact
Once Lck phosphorylates ITAMs, _____ draws close and binds the Zeta Chain; ______ becomes phosphorylates
ZAP70; ZAP70
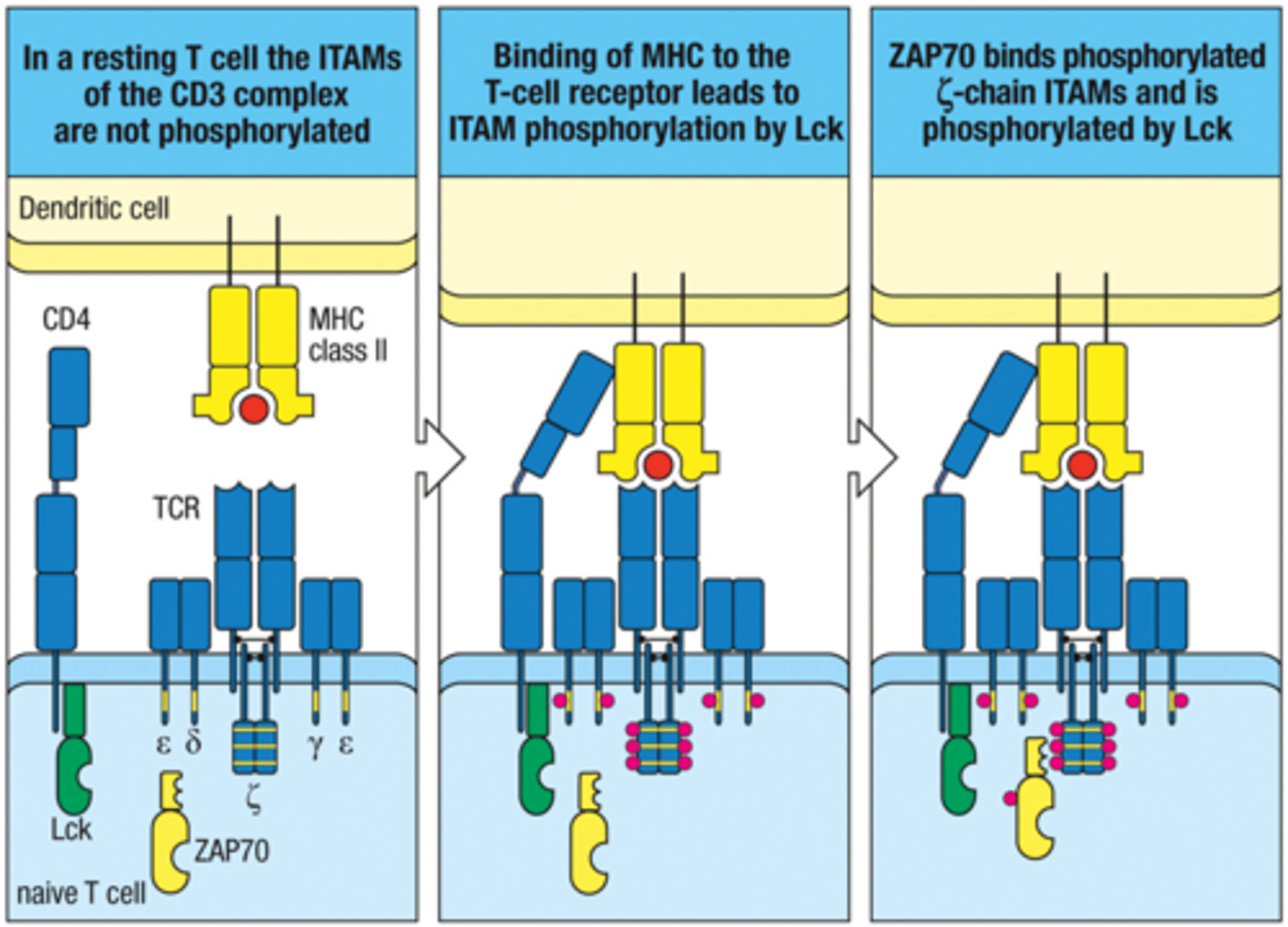
What phosphorylates CD3 ITAMs after TCR engagement? (Early TCR Signaling Cascade)
Lck
Phosphorylated ITAMs recruit ______ (Early TCR Signaling Cascade)
ZAP70
Once ZAP70 becomes activated it phosphorylates what? (Early TCR Signaling Cascade)
adaptor proteins LAT and SLP-76
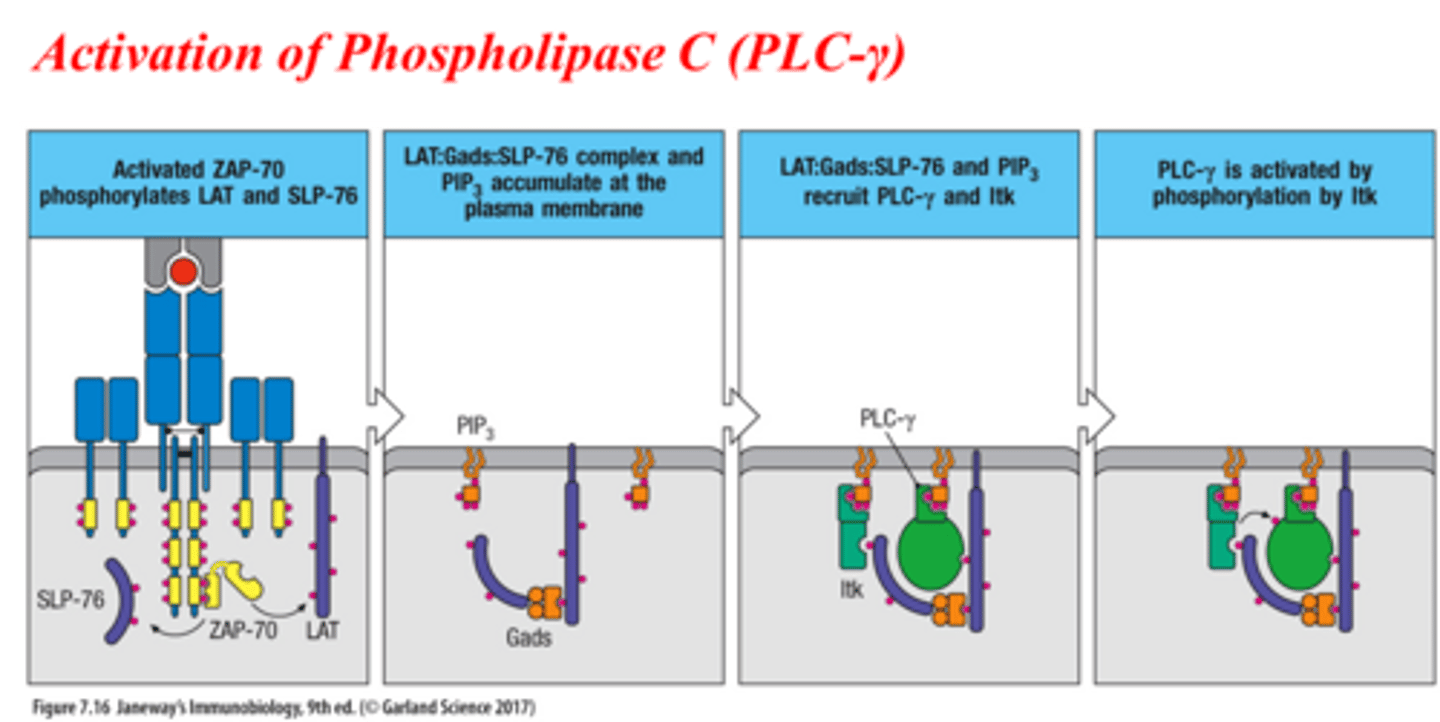
Activation of Phopholipase C (PLC-γ)
1. Activated ZAP-70 phosphorylates LAT and SLP-76
2. LAT:Gads:SLP-76 complex and PIP3 accumulate at the plasma membrane
3. LAT:Gads:SLP-76 and PIP3 recruit PLC-γ and Itk
4. PLC-γ is activated by phophorylation by Itk
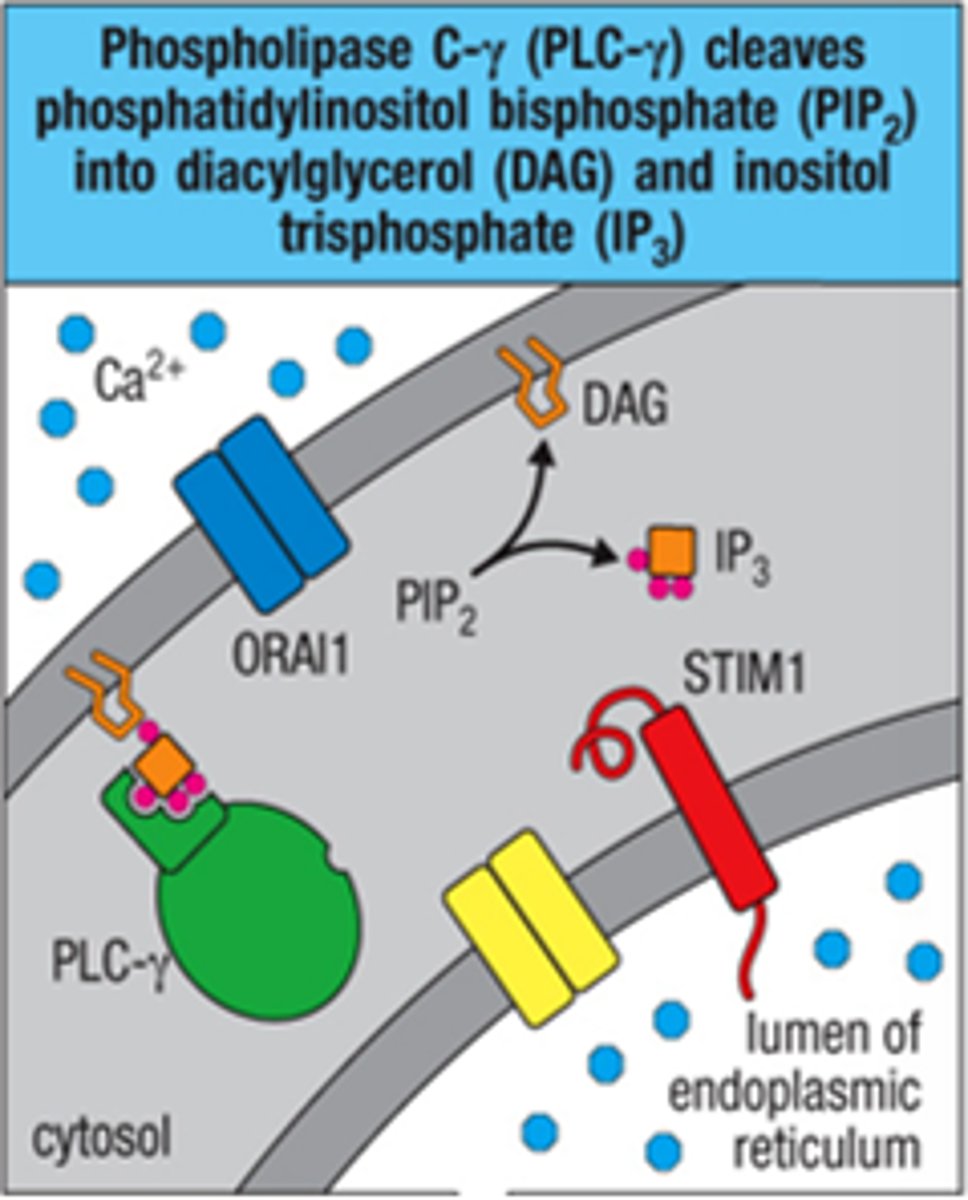
PLC-γ cleaves PIP2 (Phosphatidylinositol Biphosphate) into 2nd messengers:
1. diacylglycerol (DAG)
2. inositol triphophate (IP3)
DAG feeds into 2 pathways:
Kinase C-θ pathway or Ras-MAPK pathway
IP3 uses seperate pathway to release:
Ca2+ from endoplasmic reticulum (ER)
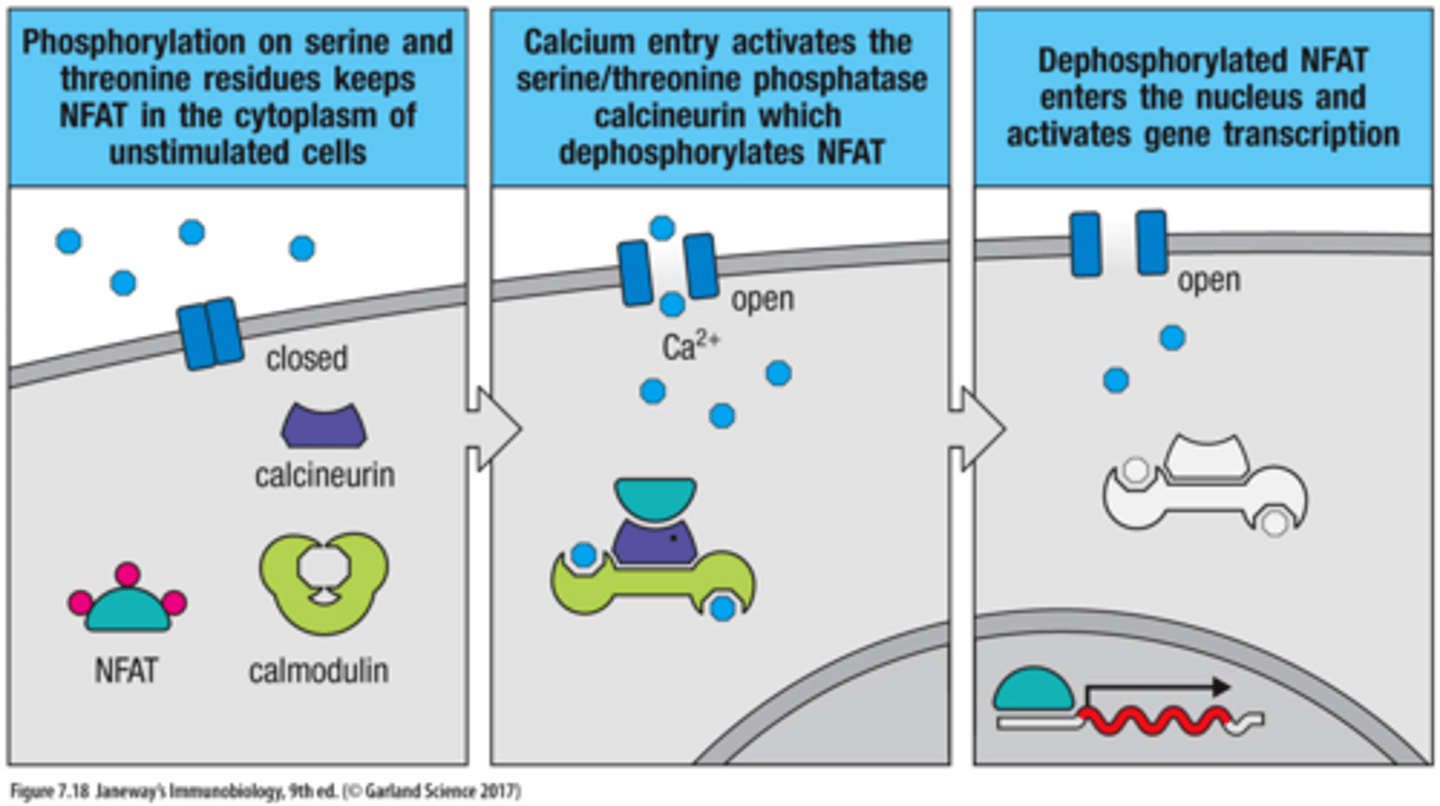
IP3 Pathway Steps
1. phosphorylation on serine and threonine residues keeps NFAT in the cytoplasm of unstimulated cells
2. Calcium entry ativates the serine/threonine phosphotase calcineurin which dephosphorylates NFAT
3. Desphosphorylated NFAT enters the nucleus and activates gene transcription
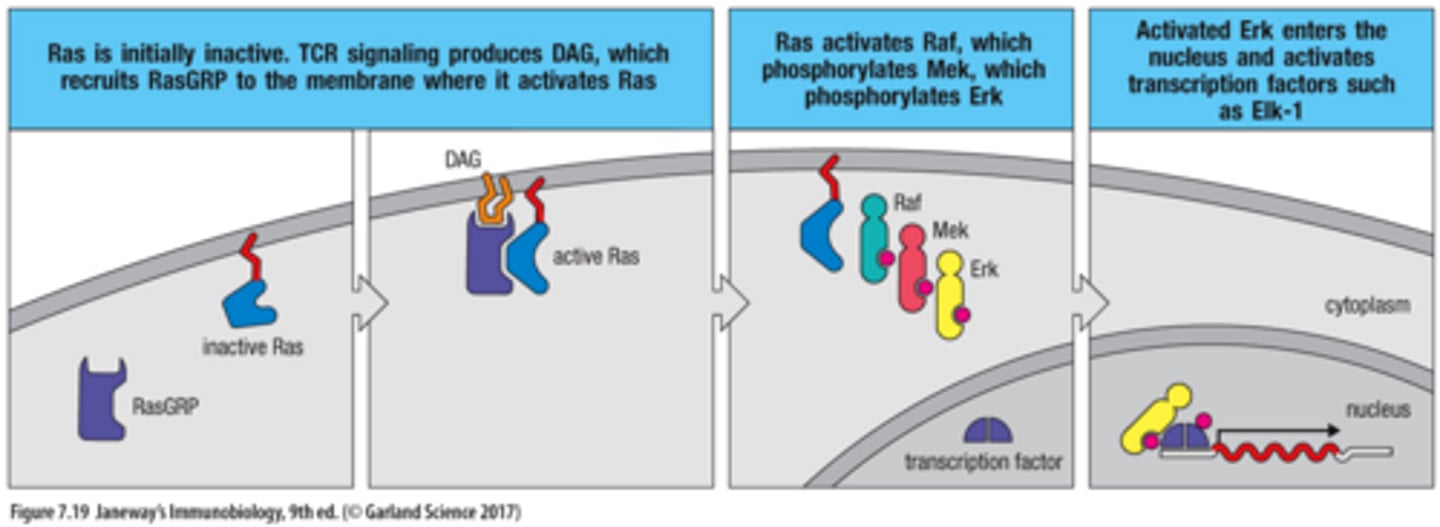
DAG Ras-MAPK pathway of Transcription Activation
1. Ras in initially inactive. TCR signaling produces DAG, which recruits RasGRP to the membrane where it activates Ras
2. Ras activates Raf, which phosphorylates Mek, which phosphorylates Erk
3. Activated Erk enters the nucleus and activaes transcription factors such as Elk-1
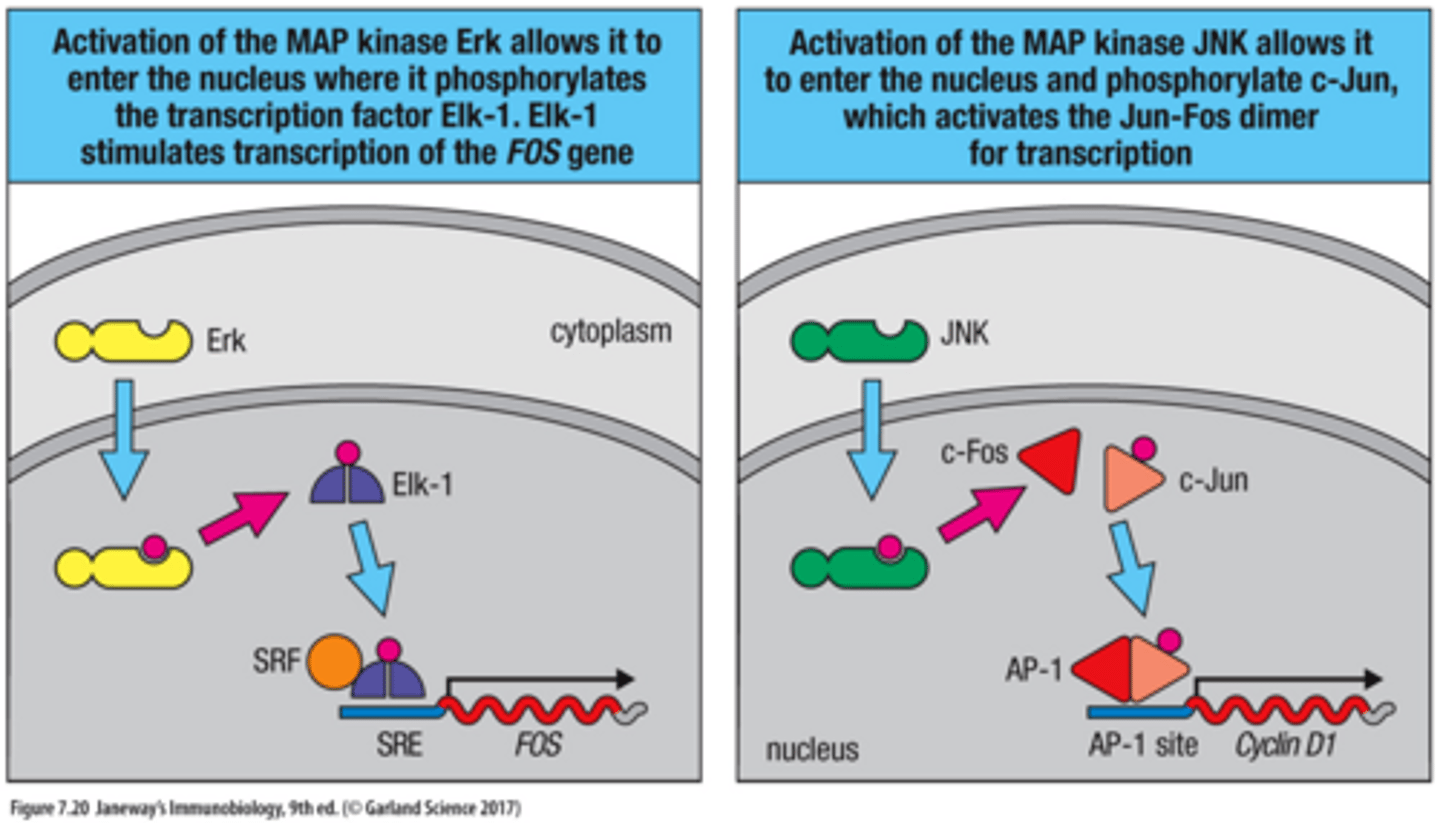
Activation of MAP Kinase Erk (DAG Ras-MAPK pathway)
Activation of the MAP kinase Erk allows it to enter the nucleus where it phosphorylates the transcription factor Elk-1. Elk-1 stimulates transcription of the FOS gene
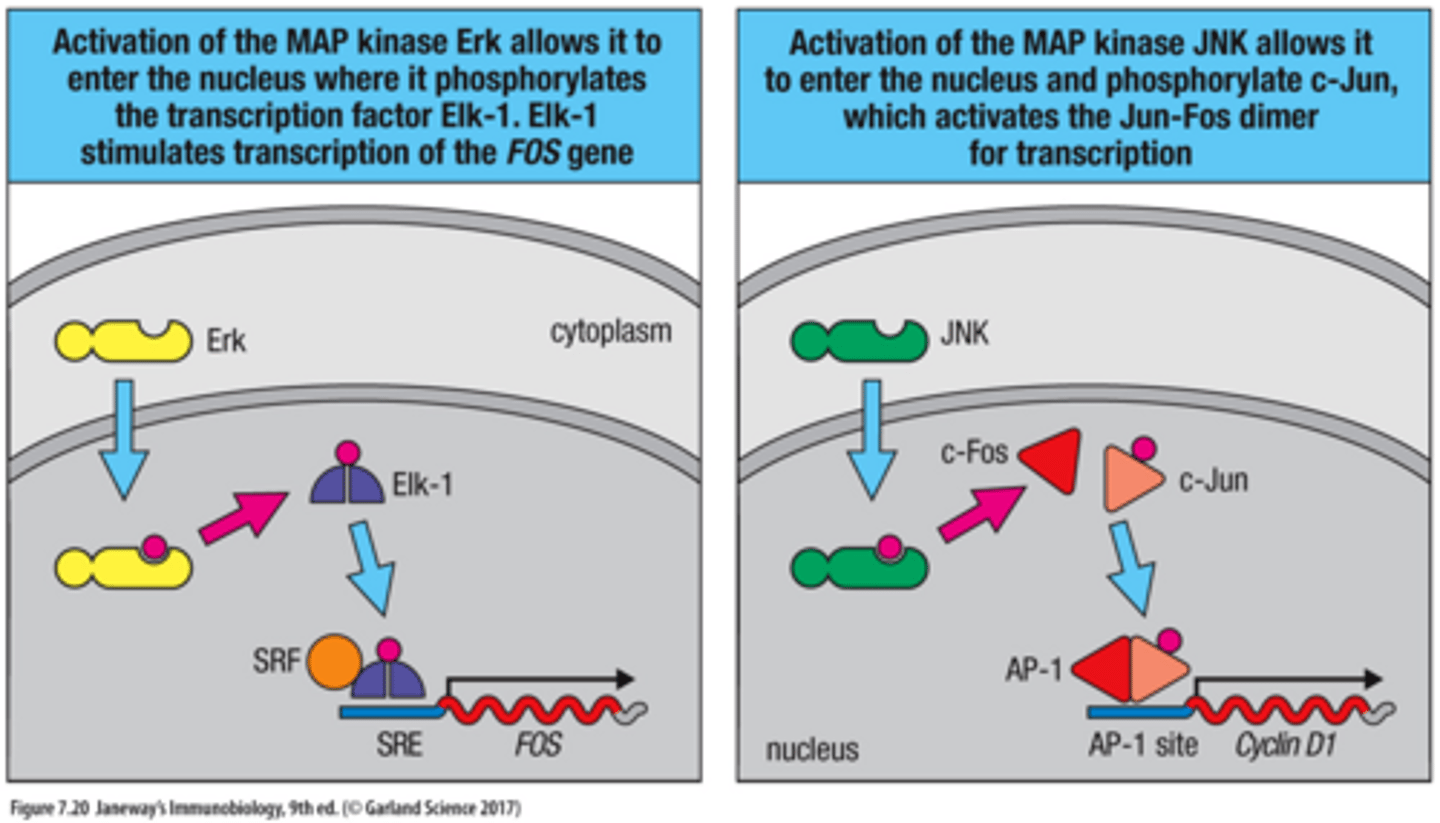
Activation of MAP Kinase JNK (DAG Ras-MAPK pathway)
Activation of the MAP kinase JNK allows it to enter the nucleus and phosphorylate c-Jun, which activates AP-1 (Jun-Fos dimer) for transcription
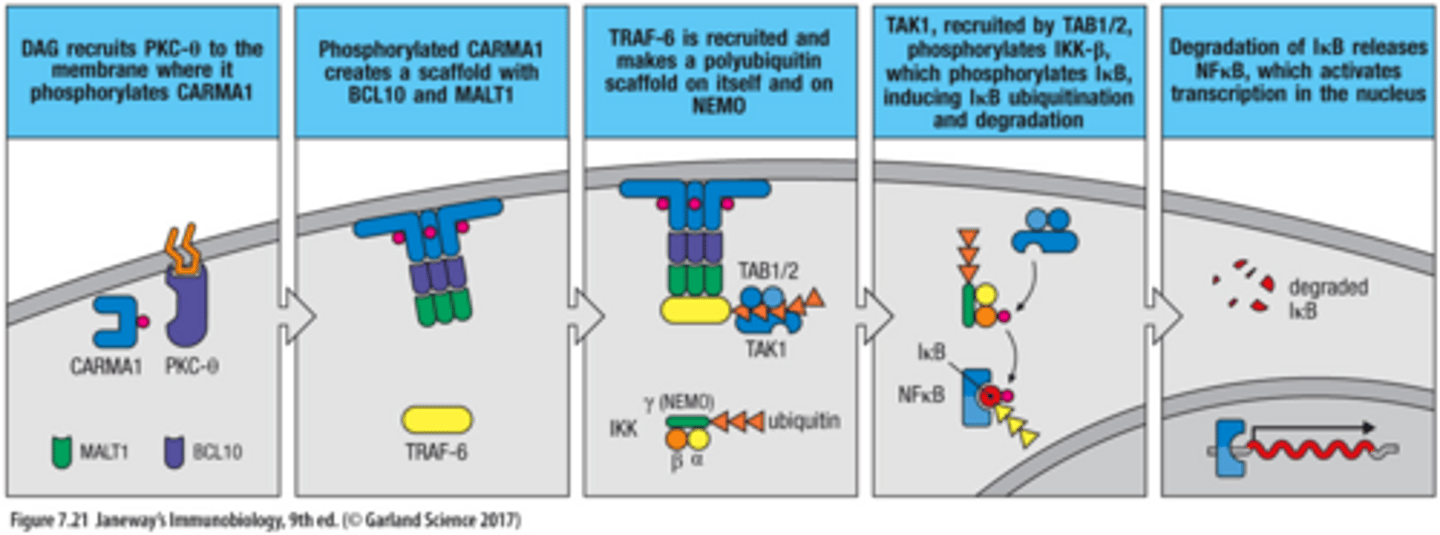
DAG Kinase C-θ Pathway
1. DAG ecruits PKC-θ to the membrane where it phosphorylates CARMA1
2. Phosphorylated CARMA1 creates a scaffold with BCL10 and MALT1
3. TRAF-6 is recruited and makes a polyubiquitin scaffold on itself and on NEMO
3. TAK1, recruited by TAB1/2, phosphorylates IKK-𝛽, which phosphorylates Iκ𝛽, inducing Iκ𝛽 ubiquitination and degradation
4. Degradation of Iκ𝛽 releases NFκ𝛽, which activates transcription in the nucleus
TCR signaling intitates __ pathways that produce Tx factors to express IL-2.
3 (AP-1, NFAT, and NFκ𝛽)
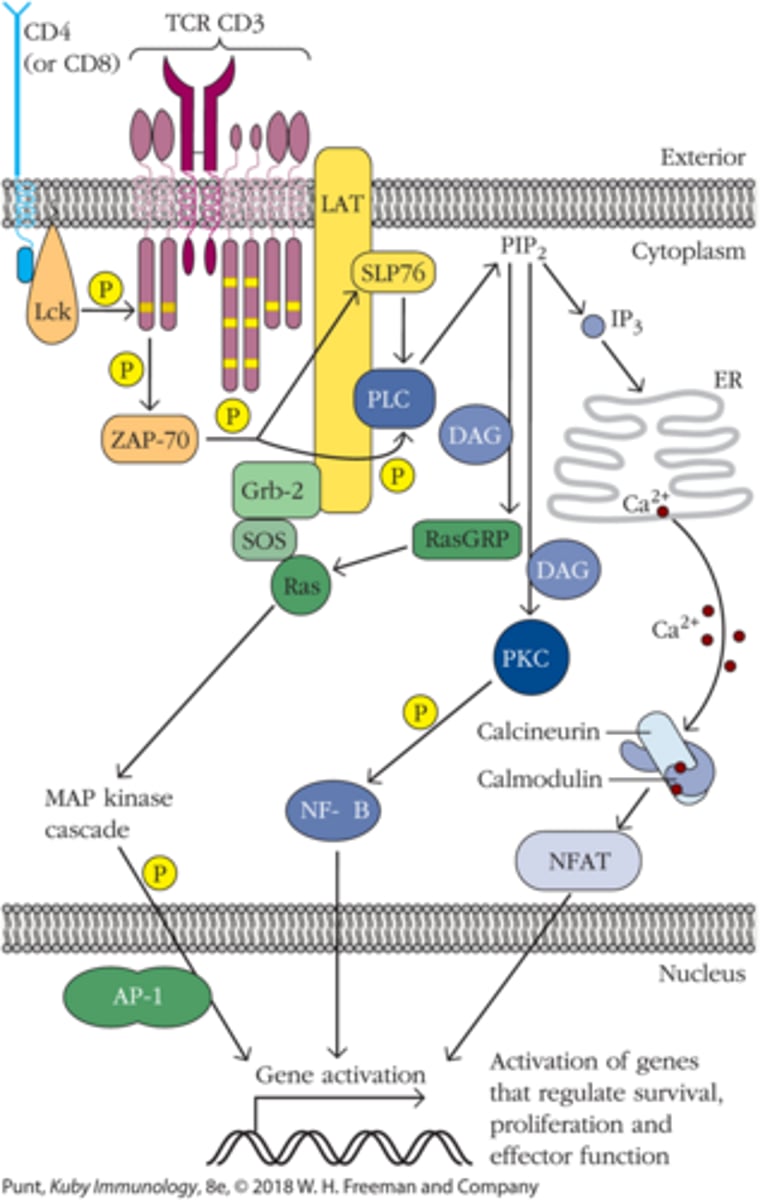
IL-2 expression only occurs when ____ _____ pathways are active
all three (AP-1, NFAT, and NFκ𝛽)
Dendritic Cells (DCs) form the ______ between the innate and adaptive immune responses.
bridge
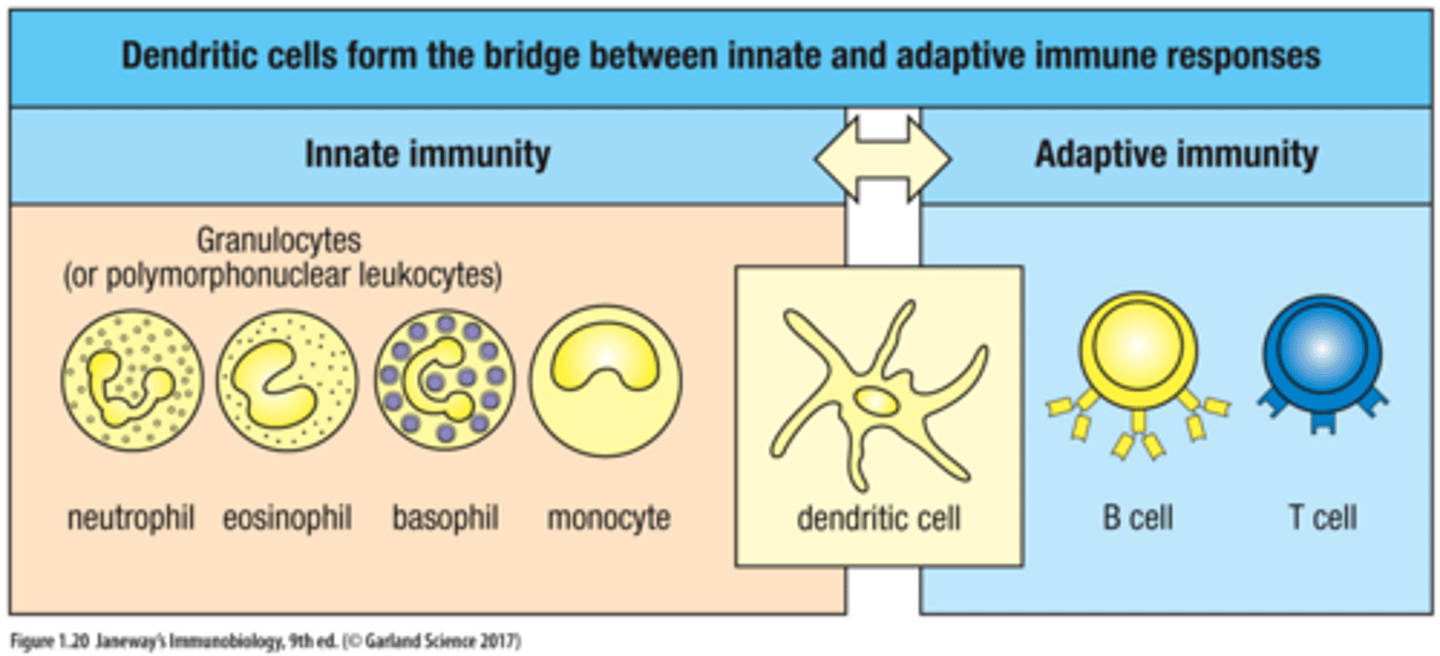
When immature DC meets pathogen/stimulated by PRRs
mature by changing their structure, change the molecules made, and may increase or decrese levels of phagocytosis
IL-2 performs either
autocrine signaling on producing T cells or paracrine signaling to neighboring T cells.
IL-2 binds IL2-R on T cells and give signal for:
survival, proliferation, and differentiation
IL-2 is in which cytokine family?
Class 1 (hematopoietin) cytokine family
IL-2 receptor (IL-2R) with only alpha domain has ____ binding affinity for IL-2.
low
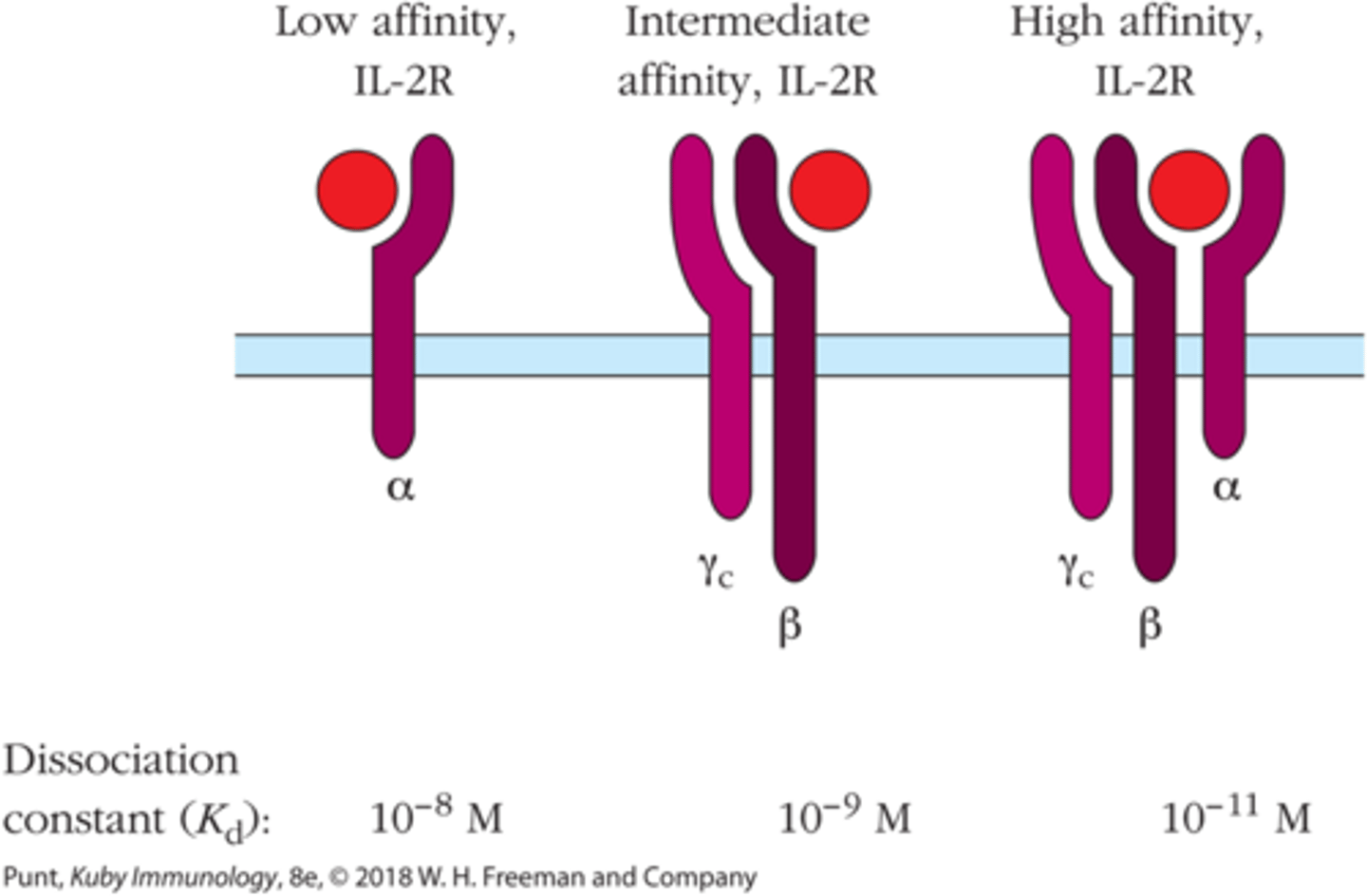
IL-2 receptor (IL-2R) with βγ domains has ________ binding affinity for IL-2.
intermediate
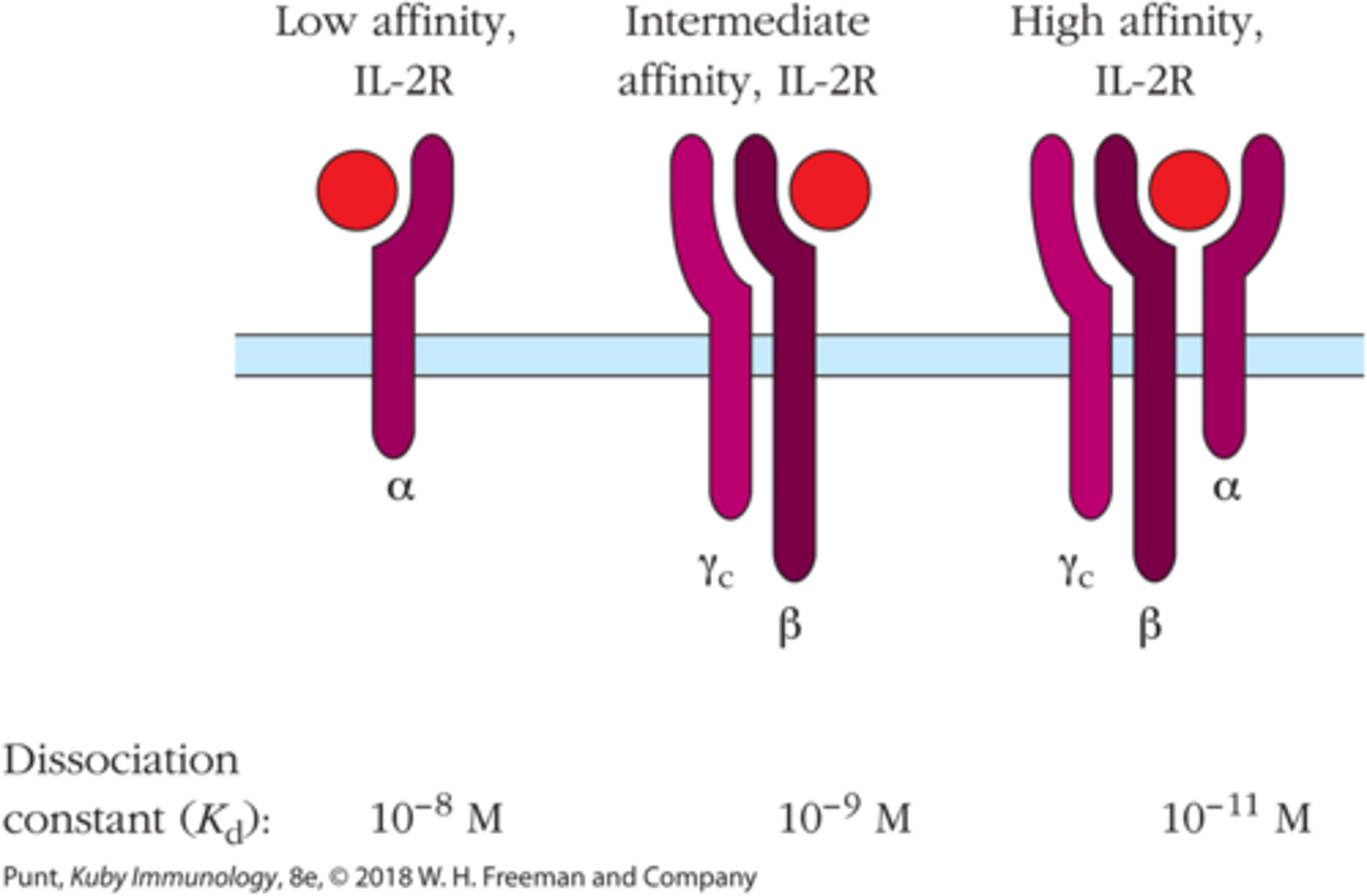
To have high binding affinity for IL-2, IL-2R must have all 3 _______ domains
αβγ
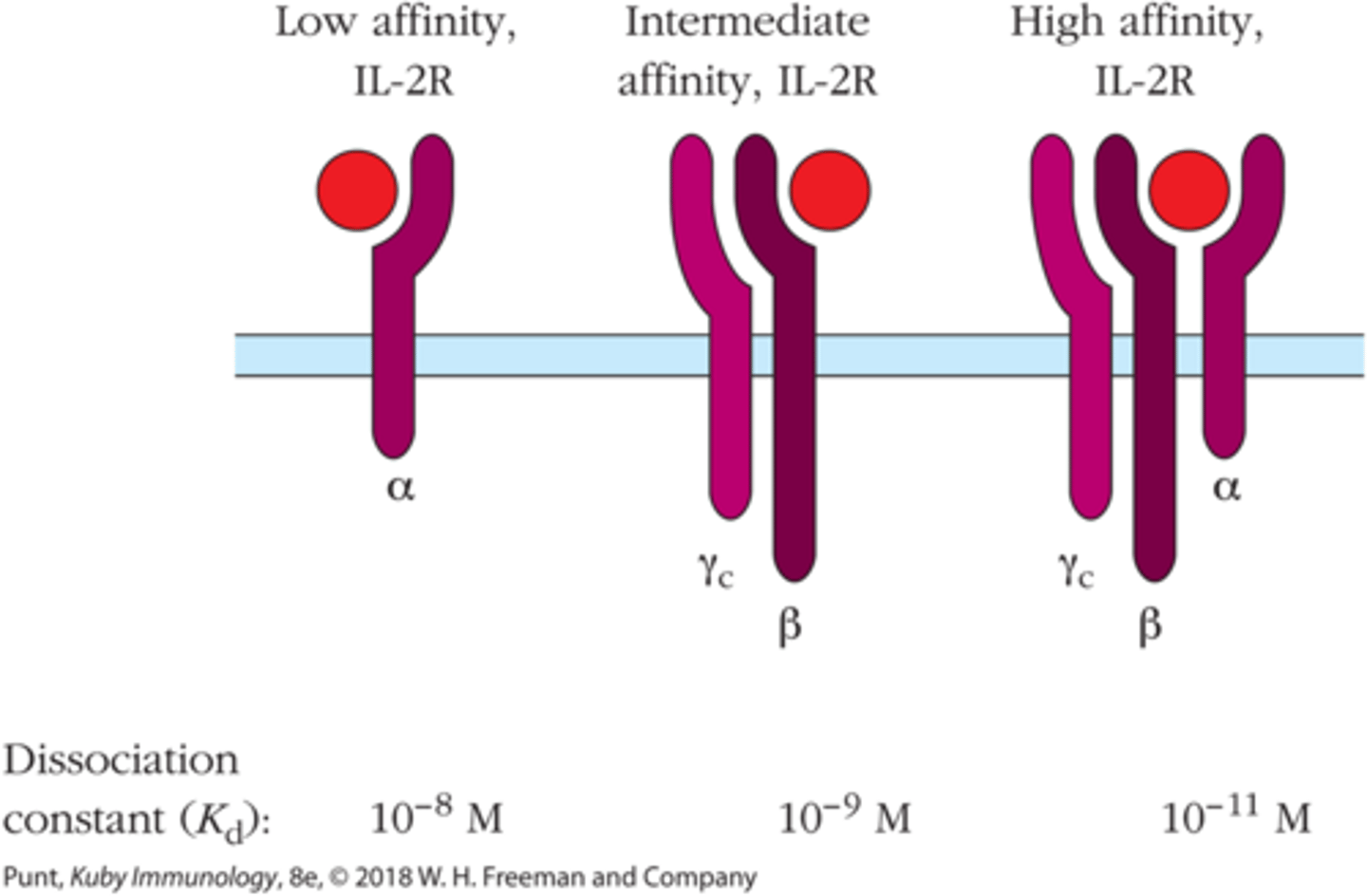
Naive T cells express low affinity IL-2R. Once activated they make _____ and start expressing ____ affinity IL-2R
IL-2; high
Signal 3 is mainly _______ mediated especially IL-2
cytokine
Activated T cells differentiate/proliferate into:
1. Stem Memory T cell
2. Central Memory T cell
3. Effectroy Memory T cell
4. Effector T cell
Stem Memory T cell
self-renewing, long-lasting reservoir
Central Memory T cell
resides in lymphoid tissues (eg lymph nodes; quick proliferation upon re-exposure
Effector Memory T cell
located in peripheral tissues (skin, mucosa); rapid response upon re-infection
Effector T cells
immediate descendants of activated T cells; CD4+ aids immune coordination; CD8+ directly kills infected cells
Regulation of T cells expasion:
Fas/FasL mechanism or CTLA-4 mechanism
Fas/FasL mechanism (Regulation of T cell expansion)
triggers apoptosis; Fas is receptor for Fas Ligand (cell thats going to die express Fas on surface)
Fas + FasL = Apoptosis
Mutation in Fas/FasL means
T cell cannot undergo Activation of Induced cell death; People with the mutation end up with combined and multiple recurring autoimmune diseases (ALPs)
Patients with ALPs (Autoimmune Lymphoproliferative Syndrome) have:
swollen lymph nodes
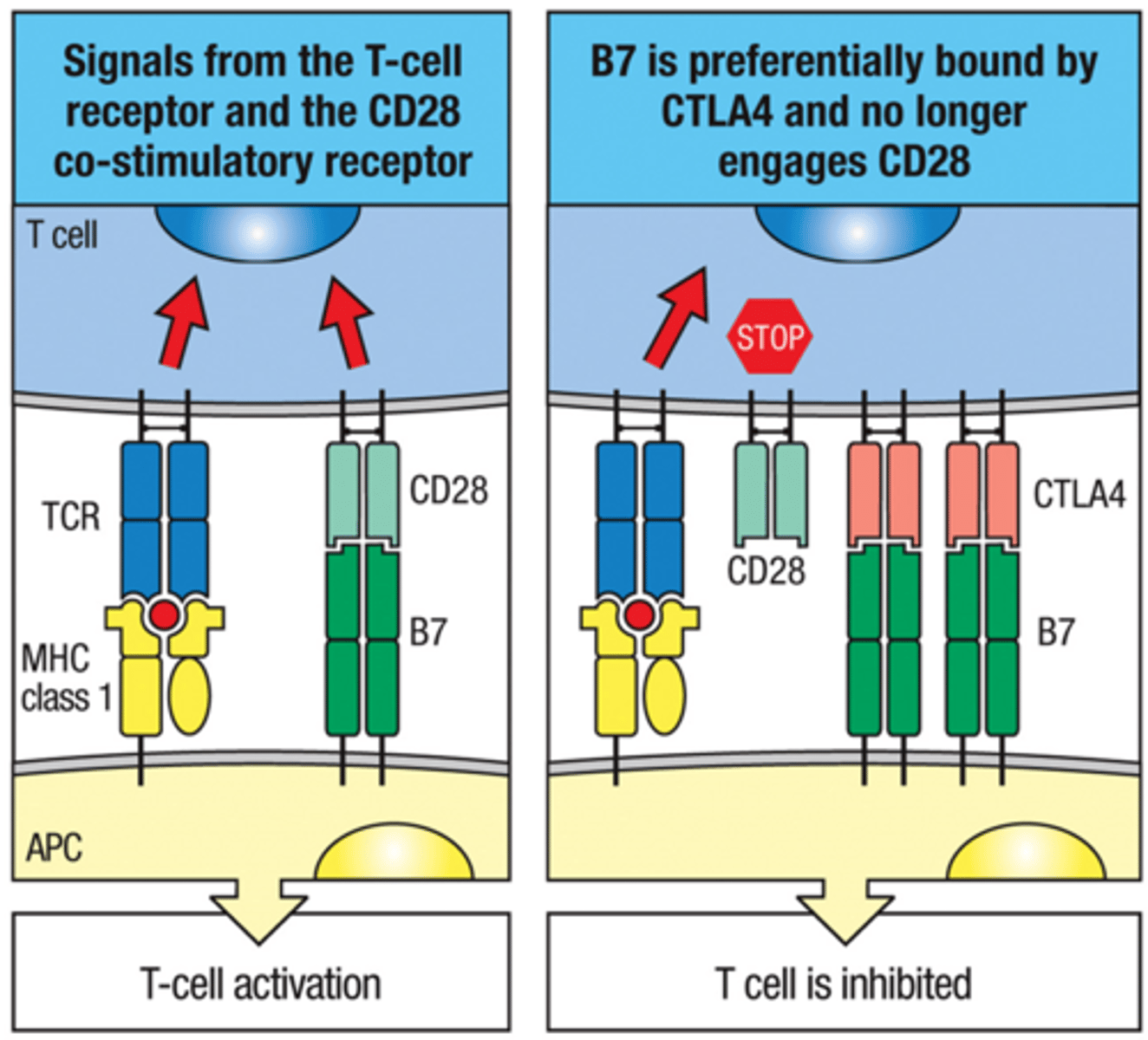
CTLA-4 (co-inhibitory molecule) mechanism for Homeostasis
activated T cell expresses CTLA-4 to be used to be used to turn off T cell responses down the line
CTLA-4 Steps of T cell Inhibition
1. CTLA-4 binds B7 (with higher binding affinity)
2. CTLA-4 gives inhibitory signals; while CD28 gives activation signals
3. CTLA-4 is expressed upon T cell activation
Main T cell classes include:
CD4+ and CD8+
After T cell activation and differentiation CD8+ cells become:
Cytotoxic T cells or CTLs
After T cell activation and differentiation CD4+ cells become different types of Helper T cells:
Peripheral Treg Cell, TH17 cell, TH2 Cell, TH9 Cell, TFH cell, TH1 Cell, TH22 Cell
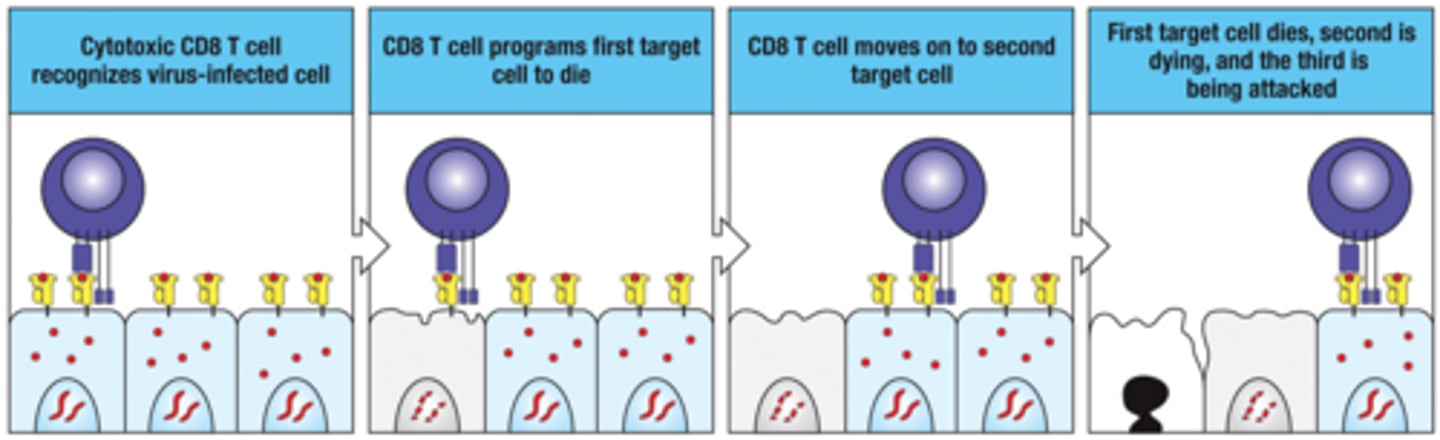
Cytotoxic CD8 T cells can kill many infected target cells in succesion
1. Cytotoxic CD8 T cell recognizes virus-infected cell
2. CD8 T cell programs first target cell to die
3. CD8 T cell moves on to seond target cell
4. First target cell dies, second is dying, and the third is being attacked
Naive CD8+ T cells are turned on in 2 ways:
1. Direct Activation of naive CD8+ T cell
2. CD4+-IL-2 Nudging/support
Direct activation of Naive CD8+ T cell
activation by virus-infected DC when there's sufficient signal
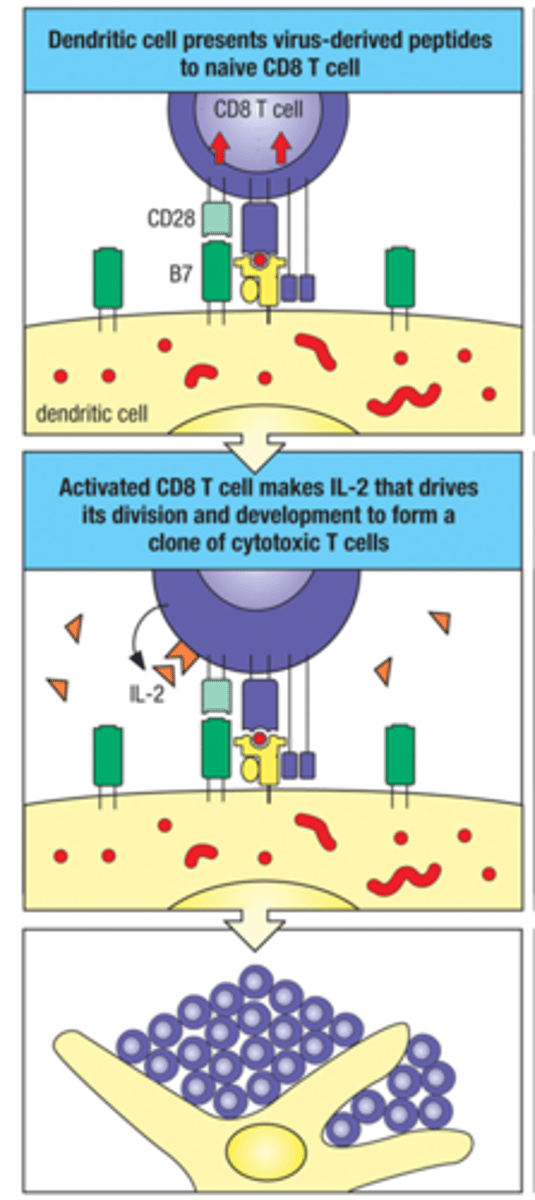
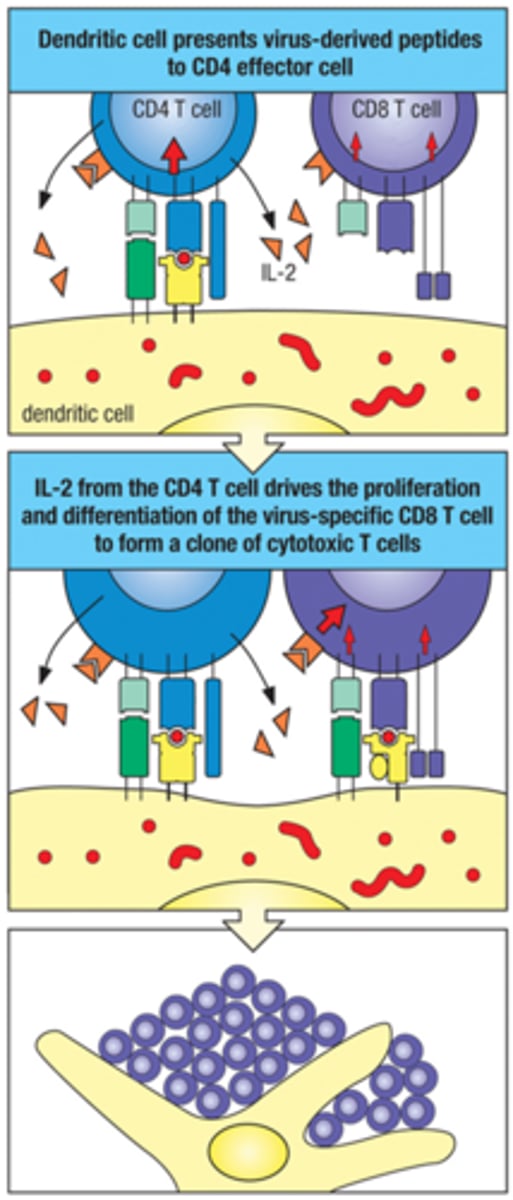
CD4+-IL-2 Nudging/support CD8+ activation
CD4+ T cell comes across cell that is not responding well to peptide on MHC II, and pushes NAIve CD8+ T cells to have a better recognition by making and directing IL-2 to the CD8+ T cells
Cytotoxic T cells mainly kill their target cells directly by:
1. Targeted secretion of lytic cytotoxins
2. Fas-induced apoptosis
3. Secretion of cytokines :TNF-α which promotes apoptosis
4. Or IFN-γ that puts cell in antiviral state and enhances the presentation of specific antigen for deleterious immune response
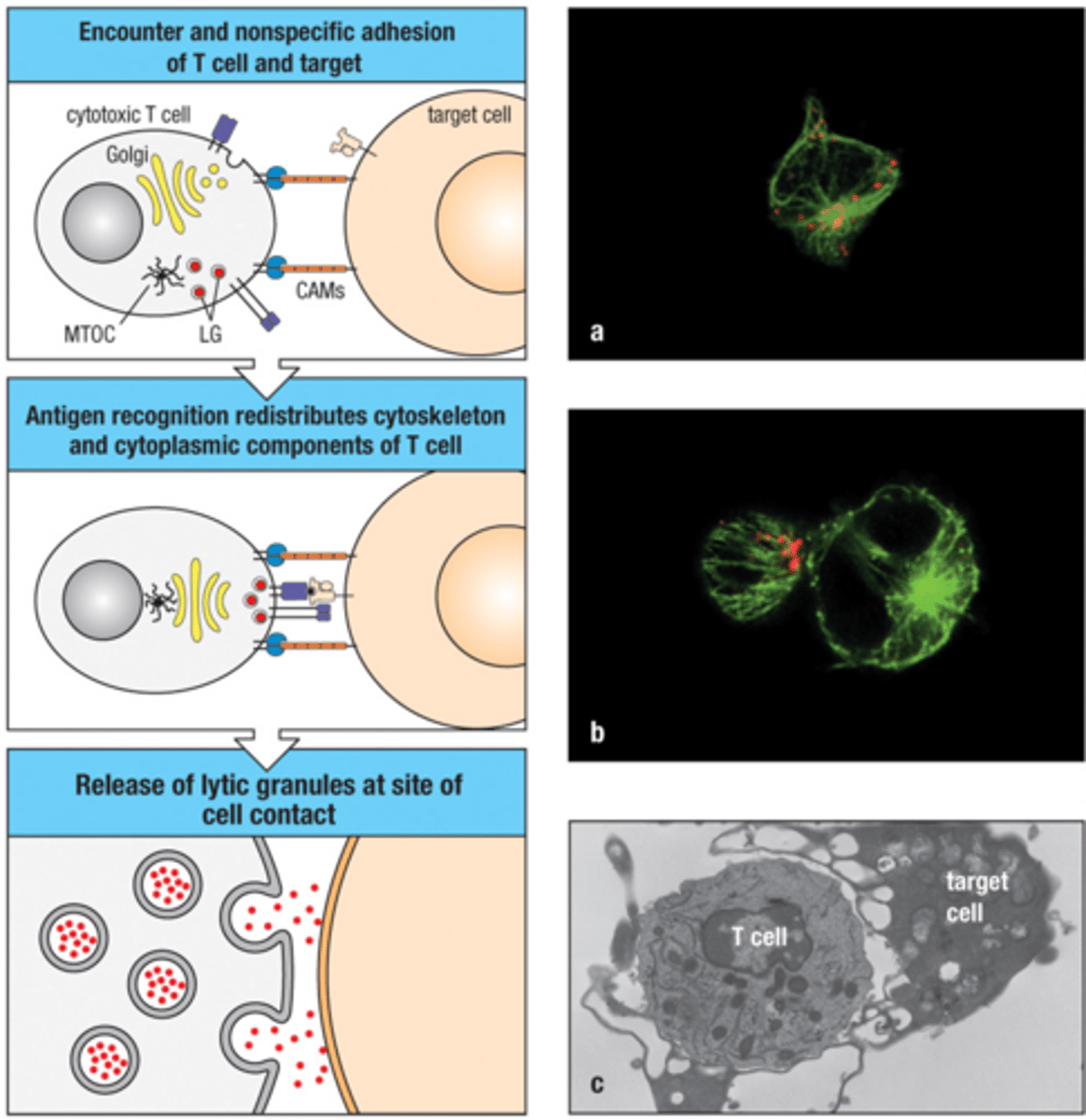
Secretion of Lytic Cytotoxins (Cytotoxic T cell methods of killing)
secretion is aimed - the immunological synapse formed by APC-T cells contains and directs the cytotoxins
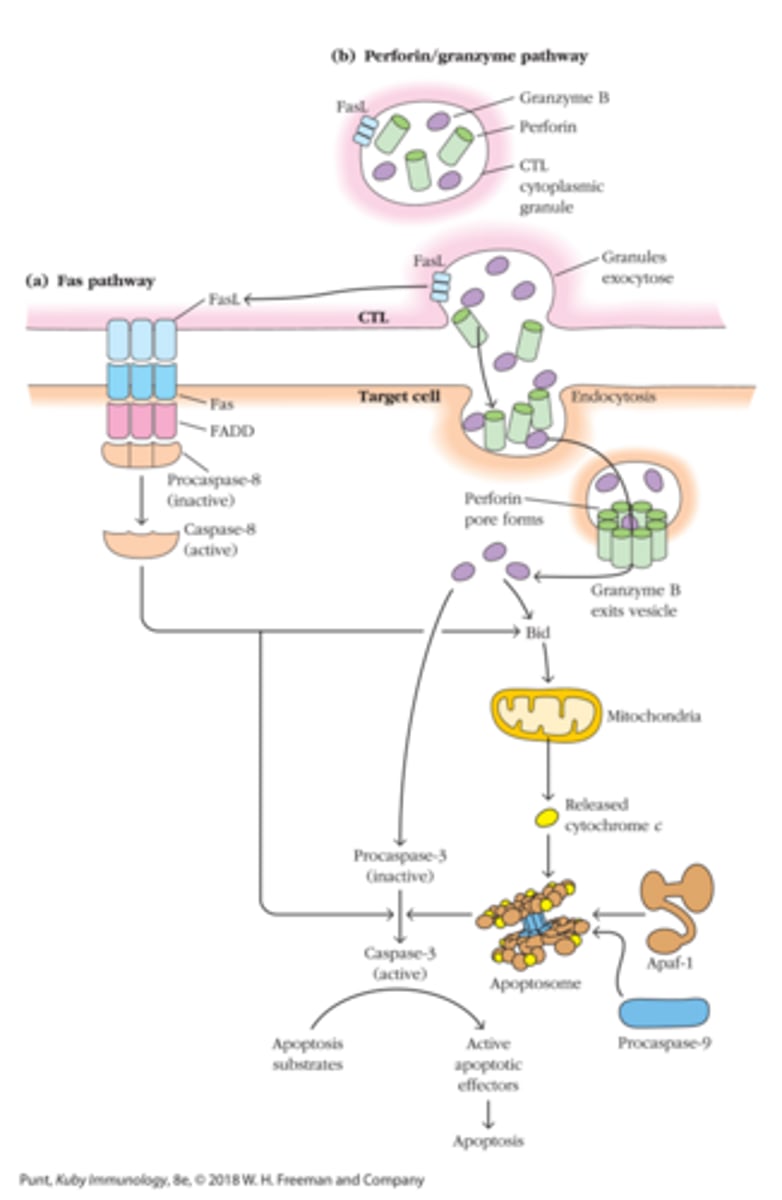
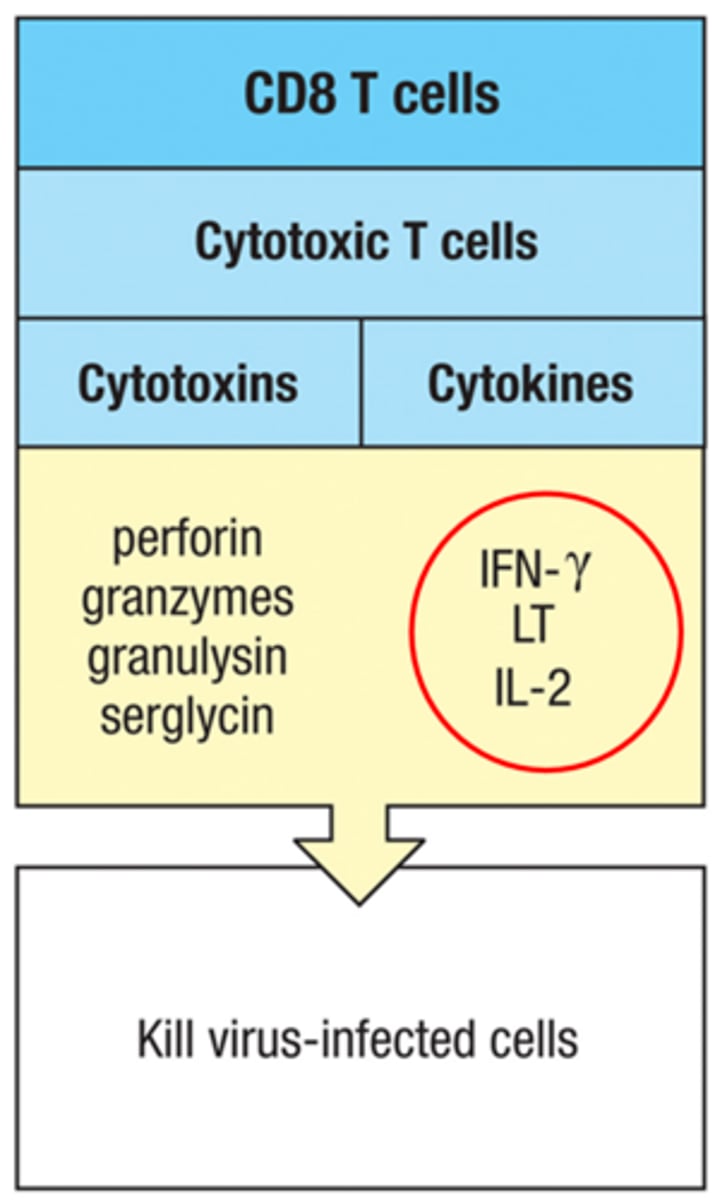
Components of CTL's granular cytotoxin:
1. Perforin: 65 kDa monomers protein that forms pores (Like MAC)
2. Granzyme B: made of several serine proteases
3. Granylysin
4. Serglycin
Granzyme or Perforing mediated Cytolysis
Granzyme comes in via receptor mediated endocytosis after binding mannose 6-phosphate (M6P) receptor on target cell surface; Perforin allows Granzyme B to get out of vesicles
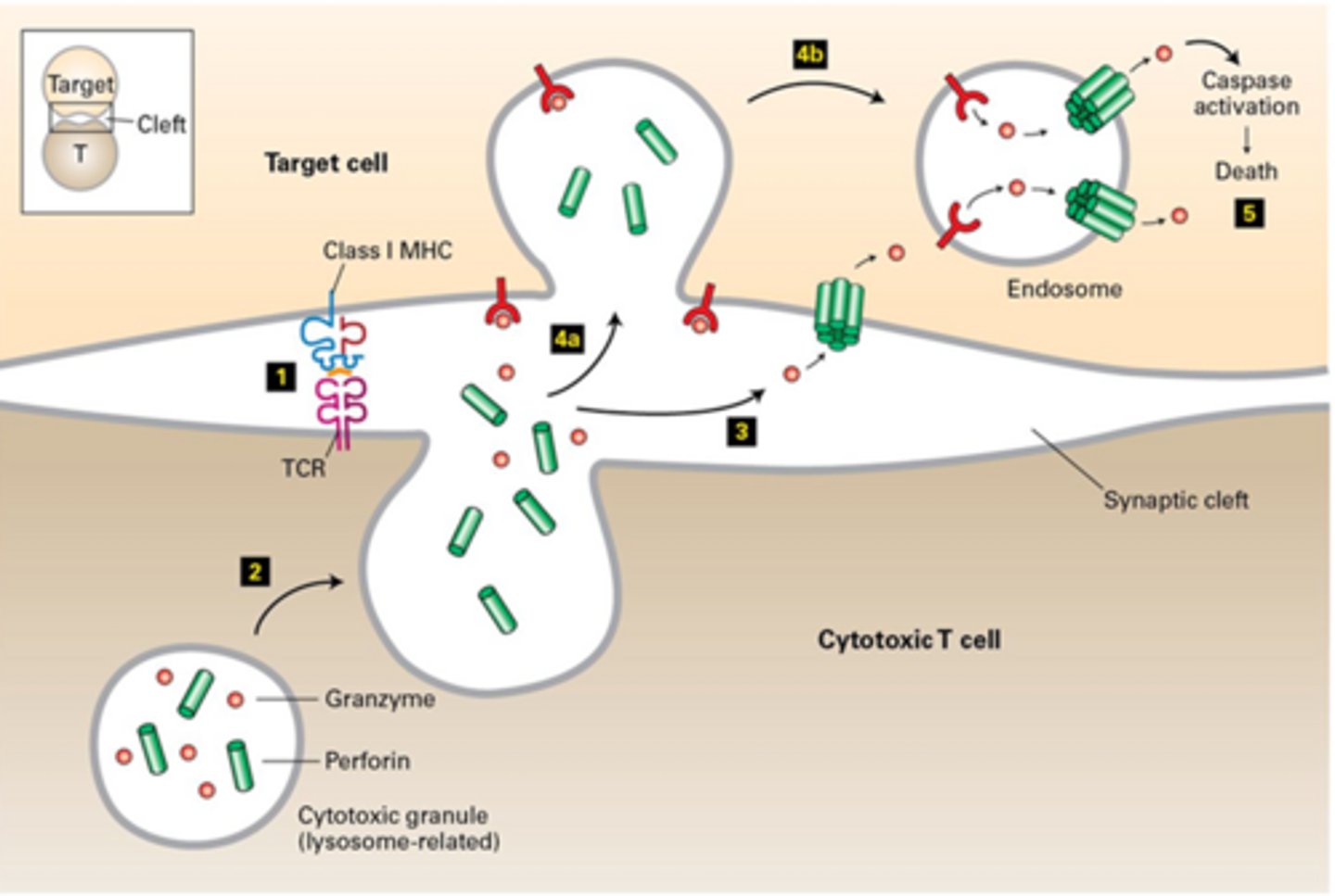
Perforin/Granzyme Effect on Target Cells:
Granzyme B and other granzymes the enter cytoplasm initiate a cascade of reactions that result in apoptosis (fragmentation of target cell DNA into oligomers of 200 BP)
Fas/FasL alternative cytotoxic pathway (Cytotoxic T cell method of killing)
1. CTLs express FasL
2. FasL binding to Fas induces apoptosis
In the lab, CTL activity may be estimated by:
Interferon γ (IFN-γ) Levels
All helper T cell functions are mediated by __________ secretion
cytokine
CD4+ helper t cells differentiation depends on?
Cytokine environement during activation (APC derived cytokines determine T helper lineage)
Polarizing cytokines
cytokines needed for activation of CD4+ T cells into different subtypes of T helper cells
Effector Cytokines
cytokines expressed by each T helper cell subtypes after activation (CD4+ functions by expressing cytokines)
TH1 Cell Differentiation
is driven by IL-12; T-bet master transcription factor of TH1 cells
TH1 Cell Effector function
produce IFN-γ and CD40L; IFN-γ activates macrophages and CD40L enhances macrophage activation
TH1 Cell Function
TH1 responses are important for intracellular pathogens
TH2 Cell Differentiation
driven by IL-4; GATA-3 is the master transcription factor of TH2 cells
TH2 Cell Effector cytokines
TH2 produce IL-4, IL-5, and IL-13
TH2 Cell Functions
IL-4 drives IgE and IgG1 class switching; IL-5 activates eosinophils; IL-4 and IL-13 increase mucus production and peristalsis
TH2 Cells Clinical Relevance
Th2 responses are associated with allergy and helminth infections
TH1 Pathway (TH1 and TH2 Cross-regulation)
IL-12 induces T-bet and TH1 differentiation; IFN-γ inhibits TH2 responses
TH2 Pathway (TH1 and TH2 Cross-regulation)
IL-4 induces GATA-3 and TH2 differentiation; IL-4 inhibits TH1 responses
Functional Implication of Cross Regulation (TH1 and TH2)
respones are polarized depending on pathogen type; incorrect polarization leads to pathology or ineffective clearance
TH17 Cell Differentiation
driven by IL-6 or IL-23; master transcription factor is RORγT
TH17 Effector Cytokines
IL-17A, IL-17F, IL-22
TH17 Cell Effector Functions
protect against extracellular bacteria and fungi; recruit neutrophils; contribute to autoimmune disease
TFH Cell Differentiation
driven by IL-6 and IL-21; Bcl-6 is the master transcription factor
TFH Cell Function
support germinal center reactions; promote affinity maturation and class switching
TFH Cell Cytokines
produce Il-21 and IL-4
Regulatory T cells (Tregs) Origin
arise in the thymus from intermediate-affinity self-reactive CD4+ T cells; Tregs can also be induced in the periphery by IL-2 and TGF-𝛽)
Regulatory T cell (Tregs) Master regulator
FoxP3 is the master transcription factor of Tregs
Regulatory T cell (Tregs) Effector Cytokines
Tregs produce IL-10 and TGF-𝛽
Regulatory T cell (Tregs) Mechanims of immune suppresion
IL-2 consumption via high CD25 expression, inhibition of APC activation, and secretion of IL-10 and TGF-𝛽
Regulatory T cell (Tregs) Function
maintain immune tolerance and prevent autoimmunity
B cell Lifecycle includes:
devlopement in bone marrow --> maturation in periphery --> peripheral trafficking --> activation --> effector response
B cell Trafficking and Localization
Naive B cells enter lympnodes vis HEV --> Localize in follicles within secondary lymphoid organs --> Chemokines regulate follicular
Activation of Naive B cells activate in periphery via:
1. Thymus (T) dependent
2. T independent