Cancer therapy I: Understanding the cancer therapy fundamentals
1/43
Earn XP
Description and Tags
Dr. Kim Phar 643
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
44 Terms
Cell cycle
Go (Gap 0):
A resting phase where the cell has left the cycle and has stopped dividing.
G1 (Gap1):
Phase ready for DNA synthesis (Cells increase in size)
S (Synthesis):
DNA replication occurs during this phase.
G2 (Gap2):
During the gap between DNA synthesis and mitosis
M (Mitosis):
Cell division
Cancer cellular characteristics
Loss of growth control
Resistance to cell death
Tissue invasive and angiogenesis
Abnormal Morphology and Genetic Instability
angiogenesis = making new blood vessels; if inhibited, then the cell cannot survive

Apoptosis: Programmed cell death
Initiation
Execution
Regulation
Therapeutic target
Initiation
Intracellular signals activate a cascade of caspases in response to cellular damage or stress
Execution
Caspase enzymes cleave specific proteins, leading to controlled cell dismantling
Regulation
Process is tightly regulated by pro- and anti-apoptotic factors, such as Bcl-2 family proteins
Therapeutic target
Inducing apoptosis in tumor cells in a promising strategy for cancer treatment
Cellular senescence: permanent cell cycle arrest
Definitions and triggers
Senescence is a state of permanent cell cycle arrest that can occur in response to normal cellular aging, DNA damage, or oncogene activation. It serves as a natural tumor suppression mechanism by preventing potentially damaged cells from dividing.
This process is regulated by complex signaling pathways, including the p53 and p16INK4a tumor suppressor pathways, which can be activated in response to various cellular stressors.
Telomeres and cancer: The immortality connection
Normal telomere function
Replicate senescence
Telomere reactivation in cancer
Therapeutic targeting
Normal telomere function
Telomeres are protective DNA-protein complexes at chromosome ends that prevent degradation and fusion. They naturally shorten with each cell division, acting as a biological clock that limits cellular lifespan.
Replicate senescence
When telomeres reach a critically short length, cells enter senescence or undergo apoptosis, preventing further division. This natural mechanism helps prevent genomic instability and tumor formation.
Telomere reactivation in cancer
Cancer cells often reactivate telomerase, an enzyme that adds telomeric DNA sequences to chromosome ends. This allows cancer cells to maintain telomere length and divide indefinitely, contributing to cellular immortalization.
Therapeutic targeting
Telomerase inhibitors like Imetelstat, BIBR1532, RHPS4, and GRN163L are being developed as potential anticancer agents. By blocking telomerase activity, these drugs aim to restore telomere shortening and limit cancer cell proliferation.
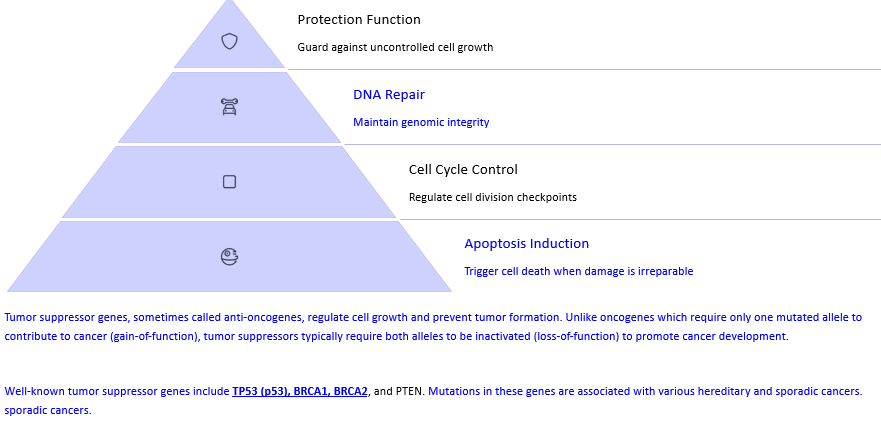
Oncogenes vs Tumor Suppressor Genes
Oncogenes
•Genes that have the potential to cause cancer when mutated or overexpressed.
•They are normal genes that code for proteins involved in cell growth and division. However, when these genes are altered or mutated, they can become overactive and promote uncontrolled cell growth, leading to the development of cancer.
•Proto-oncogenes are normal genes that regulates cell growth and division. When they are mutated or overexpressed, they can become oncogenes and contribute to the development of cancer.
Tumor Suppressor Genes (=anti-oncogenes)
•Genes that regulate cell growth and prevent the formation of tumors
•Inactivation or mutation of tumor suppressor genes can lead to loss of their function, resulting in uncontrolled cell growth and division, leading to the development of cancer.
Oncogenes: Drivers of Cancer Development

Tumor Suppressor Genes: Cancer's Natural Inhibitors
Genetic Testing in Cancer: Types and Applications
Somatic vs Germline mutations
Single vs panel testing
Somatic mutations are detected in cancer tissue and only affect tissues derived from the mutated cell. These mutations are not inherited and cannot be passed to offspring.
Germline mutations are tested using blood or saliva samples. These hereditary mutations are present in every cell of the body and can be passed to offspring, potentially increasing cancer risk in future generations.
bolded terms distinguish their differences
Single gene testing examines one specific gene for mutations, often used when a particular mutation is suspected based on family history or cancer type.
Panel gene testing examines multiple genes simultaneously. Next Generation Sequencing (NGS - screen all the genes at the same time) is a powerful technology that enables massively parallel sequencing, allowing for comprehensive genetic profiling of tumors and personalized cancer treatment approaches.
Predictive biomarkers
Provide information about the likely effect of a specific therapeutic intervention. Examples include HER2 testing for targeted therapies in breast cancer and PD-L1 testing for immunotherapy response across multiple cancer types.
Prognostic Biomarkers
Offer information about a patient's overall cancer outcome, regardless of therapy. Ki-67, a marker of cellular proliferation in cancer cells, is a classic example that helps predict how aggressive a tumor might be.
do not have an agent to target the Ki-67??
be sure to check for this biomarker
Clinical Applications
These biomarkers guide treatment decisions, help monitor disease progression, and allow for more personalized cancer care approaches tailored to individual tumor characteristics.
Predictive biomarkers in cancer therapy
HER2 (Human Epidermal Growth Factor Receptor 2) overexpression or amplification predicts response to HER2-targeted therapies like trastuzumab, pertuzumab, and T-DM1. Testing is standard for all invasive breast cancers to identify patients who will benefit from these targeted treatments.
Epidermal Growth Factor Receptor (EGFR) mutations predict response to EGFR tyrosine kinase inhibitors like osimertinib, erlotinib, and gefitinib in non-small cell lung cancer. These mutations are more common in never-smokers and Asian populations.
Programmed Death-Ligand 1 (PD-L1) expression levels help predict response to immune checkpoint inhibitors across multiple cancer types. Higher expression is generally associated with better response to PD-1/PD-L1 inhibitors (Checkpoint inhibitors).
Microsatellite Instability-High (MSI-H) or Deficient Mismatch Repair (dMMR) status predicts response to immunotherapy regardless of cancer type, leading to tumor-agnostic approvals for certain checkpoint inhibitors.
Prognostic biomarkers in cancer
Ki-67 Proliferation Index - most well known, although there are others
Basic tumor nomenclature
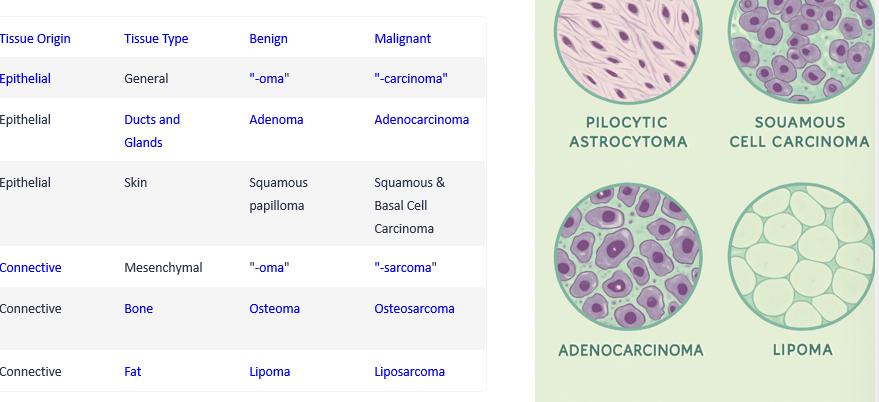
Essential cancer terminology
Doubling time
the time required for a tumor to double in size
Malignancy