Chapter 7 - Quantum, Atomic, and Nuclear Physics
Basics
- Einstein’s postulates of special relativity * All laws of physics remain the same in a uniformly moving frame of reference * The speed of light in a vacuum is always 3 x 10^8 no matter the motion of the source of light or the observer * Summary: time and distance are relative according to your frame of reference
- ==E = mc^2== * Mass is a solid form of energy and can be converted into energy and vice versa
- Big 4 subatomic particles * Proton (p) * Mass = 1.67 x 10^-27 kg = 1 amu * Charge: positive * Electron (e) * Mass = 9.11 x 10^-31 kg * Charge: negative * Neutron (n) * Mass = 1.67 x 10^-27 kg = 1 amu * Charge: 0 * Photon (ɣ) * Mass = 0 * Charge: 0
- Electron-Volts (eV) * : a unit of energy - the amount of energy needed to change the potential of an electron by 1 volt * ==1 eV = 1.6 x 10^-17 J==
- Photons * Light is made of photons * ==E = hf = hc/λ== * E: energy of a photon * h: Planck’s constant = 6.63 x 10^-34 Js = 4.14 x 10^-15 eVs * f: frequency (Hz) * c: speed of light (3 x 10^8 m/s) * λ: wavelength (m)
Photoelectric Effect
- Applications: solar panels, photosynthesis, tanning, photographic film
- __Photoelectric effect__: when incident light is shined on a metal, electrons detach
- ==K(max) = hf - ɸ== * K(max): max kinetic energy of the emitted electron * h: Planck’s constant * f: frequency * hf: energy of the incident photon * ɸ: work function - the energy required to remove an electron from a specific element/material * When the frequency of incident light increases, the maximum kinetic energy of the emitted electron increases linearly * Threshold frequency: minimum frequency for electron emission
- Photon Momentum * When a photon collides with an atom and the atom emits an electron, momentum and energy are conserved * ==p = h/λ = E/c==
DeBroglie Wavelength
- If a particle has a shorter wavelength, it behaves more like a particle
- If a particle has a longer wavelength, it behaves more like a wave
- To find the wavelength for a particle (de Broglie’s wavelength), use ==λ = h/p = h/mv== * λ: de Broglie’s wavelength * p: momentum of particle
- Particles have a __wave function__ representing the probability of finding the particle at a specific location * Ѱ: wave function * Ѱ = 0: no probability of finding the graph
Energy Levels in an atom
- For an electron to move from one energy level to another, it will either have to absorb or emit energy in the form of a photon
- The nucleus of an atom is positive and electrons are negative so it takes energy to pull the electron away from the nucleus by overcoming their attractive force * Electrons take less energy if they’re in a higher energy level
- Key points * n1 is called the ground state - the lowest possible energy level for the electron * Moving from lower to higher energy levels tells you the atom absorbed a photon * Moving from higher to lower energy levels tells you the atom emitted a photon * There are no intermediate levels between energy levels * ==E (photon) = E (final) - E (initial)==
- If there is extra energy after jumping from one energy level to another, that energy is converted to kinetic energy of the emitted electron
Nuclear Decay
- Particles involved with nuclear decay: * __Alpha particle (𝞪): two protons and two neutrons together (helium nucleus) * : either an electron or positron * Gamma particle (Ɣ__): a gamma ray photon - massless and chargeless
- Isotopes - same atomic number of an element but different mass numbers * Notation for Isotopes: element symbol with two small numbers to the left (one on top of the other) * Top number on the left side of the symbol: mass number = neutron # + proton # * Bottom number on the left side of the symbol: # of protons in the nucleus = atomic number
- __Alpha (𝞪) decay__: a Helium nucleus is emitted from the original isotope
- __Beta (β) decay__: either a positron or electron is emitted * β+ (also symbolized as e+): positron - +1 charge with negligible mass * β- (also symbolized as e-): electron - -1 charge with negligible mass
- __Gamma (Ɣ) decay__: massless and chargeless photon * The photon carries away some energy and momentum so the nucleus recoils
- __Neutron deca__y: a neutron is emitted
- __Mass defect__: the slight difference in mass between the total mass present before the decay and after the decay * This difference in mass is destroyed and converted into kinetic energy * ==E = Δmc^2== * Δm: mass defect * c: speed of light * E: energy produced * ==1 u = 931 MeV/c^2== * The mass defect may become the nuclear binding energy and will be equal to the strong nuclear force that holds the nucleus together
- Half-life: the time it takes for a radioactive isotope to decay half its original amount
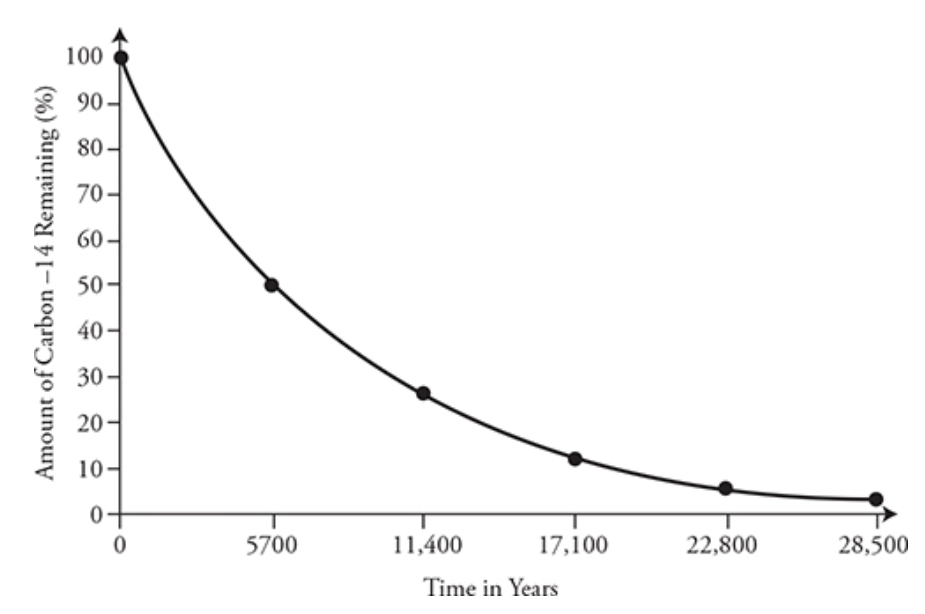
\ * Longer half life → slow decay rate
- __Fission reactions__: when a heavy nucleus is split into two chunks * Begun by shooting a neutron into the nucleus * Nuclear power plants and weapons
- Fusion reactions: when two light nuclei combine to make a heavier and stable nucleus
- Induced Reaction: scientists bombard a nucleus with high-speed particles to induce the emittance of a proton
- Antimatter: every normal particle has an antimatter to match it (electron and positron) * When matter and antimatter meet, they annihilate each other * Ex: electron and positron can turn into photon energy * ==E (electron) + E(positron) = (2m)c^2 = hf== * m: mass of electron * c: speed of light * h: Planck’s constant * f: frequency
\