Chapter 12 - Thermodynamics
12.1 Spontaneous Processes
Spontaneous Process: process that proceeds without any ongoing intervention, self-sustaining once initiated; does not necessarily mean a fast or exothermic reaction
Non-spontaneous Process: cannot happen on its own
12.2 Entropy and the Second Law of Thermodynamics
First law of Thermodynamics: energy cannot be created or destroyed - cannot do more work than allowed by available energy
Second law of Thermodynamics: not all energy released by a spontaneous reaction is available to do useful work
- energy unavailable to do useful work spreads out and becomes less concentrated over time
Entropy: measure of energy dispersion of a system; always increases during a spontaneous process
Accessible Microstates: probable number of arrangements of particles in a system at a given temperature
Third Law of Thermodynamics: a perfect crystalline solid has zero entropy at a temperature of absolute zero
12.3 Absolute Entropy and Molecular Structure
Standard Molar Entropy (S°): entropy of one mole of a substance in its standard state
- linked to masses and molecular structures (rotational motion and rigidity)
Entropies of States: Ssolid<Sliquid<Sgas
Entropies increase as temperature, volume and number of independent particles increases
12.4 Applications of the Second Law
A spontaneous process produces an increase in entropy of the universe
- ΔSuniv > 0; spontaneous process
- ΔSuniv < 0; nonspontaneous process
- ΔS = q/T
 Reversible Process: happens slowly, can be reversed by changing reaction conditions to restore original state of system with no net flow of energy in/out of system
Reversible Process: happens slowly, can be reversed by changing reaction conditions to restore original state of system with no net flow of energy in/out of system
12.5 Calculating Entropy Changes
Entropy is a state function: ΔSsys = ΔSfinal - ΔSinitial
![]()
Each S° value for product/reactant is multiplied by number of moles derived from balanced equation
12.6 Free Energy
- Endothermic reactions have thermal energy flowing from surroundings into the system, lowering surrounding temperature and decreasing its entropy
- Exothermic reactions have thermal energy flowing into surroundings from system, increasing its entropy
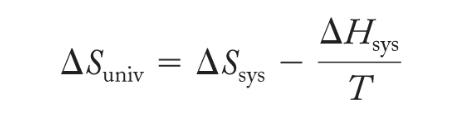
Free Energy: measure of maximum work a thermodynamic system can perform, energy that is freed during process
- ΔE = q + w
- ΔH = ΔG + TΔS
Gibbs free energy: maximum amount of energy available to do useful work in processes happening at a constant temperature and pressure or once temperatures and pressures of reaction mixtures have returned to their initial values, represented by ΔG; is a state function
- ΔGsys = -TΔSuniverse = ΔH - TΔS
- ΔG < 0, then ΔS > 0, reaction is spontaneous
- ΔG > 0, then ΔS < 0, reaction is not spontaneous
- ΔG = 0, then ΔS = 0, reaction has reached equilibrium

A decrease in free energy and a spontaneous process at constant temperature and pressure occur when there is an increase in entropy or the process is exothermic
Standard Free Energy of Formation (ΔG°f): change in free energy associated with formation of one mole of it in its standard state from its elements in their standard states
![]()
12.7 Temperature and Spontaneity
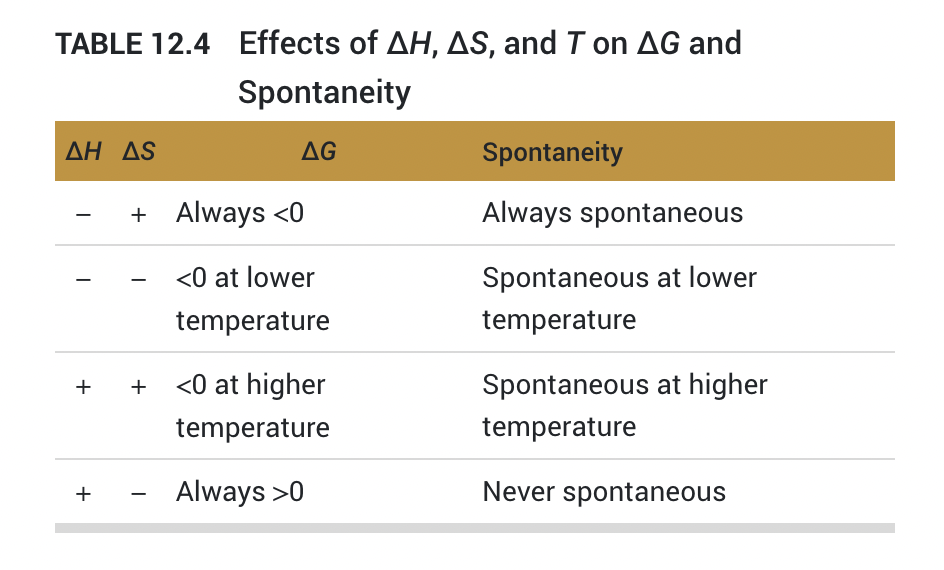 12.8 Coupled Reactions
12.8 Coupled Reactions
- living systems use energy from spontaneous reactions to run non spontaneous reactions
Glycolysis: series of reactions converting glucose to pyruvate; major anaerobic pathway of glucose metabolism
Phosphorylation reaction: reaction resulting in the addition of a phosphate molecule to an organic molecule