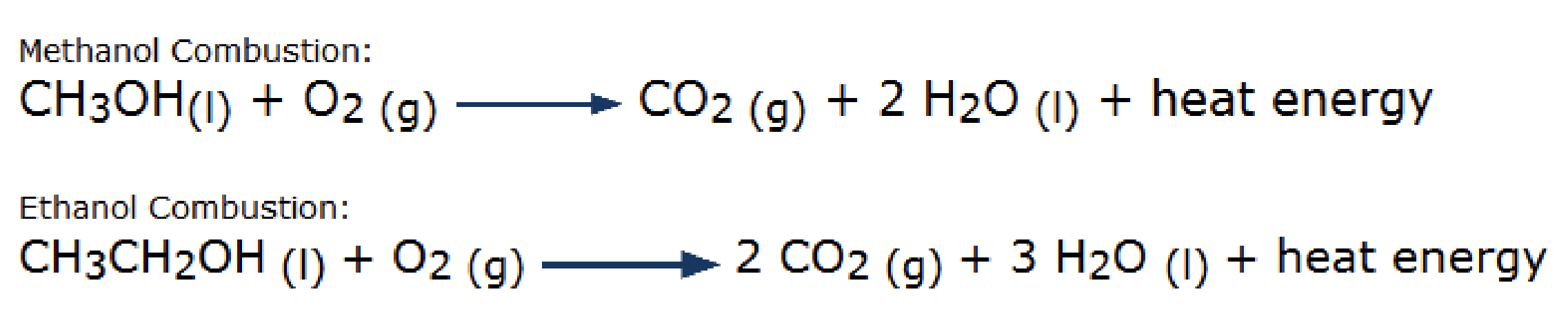1.4 Alcohols
Alcohols
- General Formula: R-OH
- Name of the functional group: Hydroxyl
Naming Alcohols
- Add “ol” to the end of the alkane name.
Alcohol Types
- Primary (1°) alcohols – hydroxyl is bonded to a terminal carbon
- Secondary (2°) alcohols – hydroxyl is bonded to a carbon attached to 2 alkyl groups (Cs)
- Tertiary (3°) alcohols – hydroxyl is bonded to a carbon attached to 3 alkyl groups (Cs)

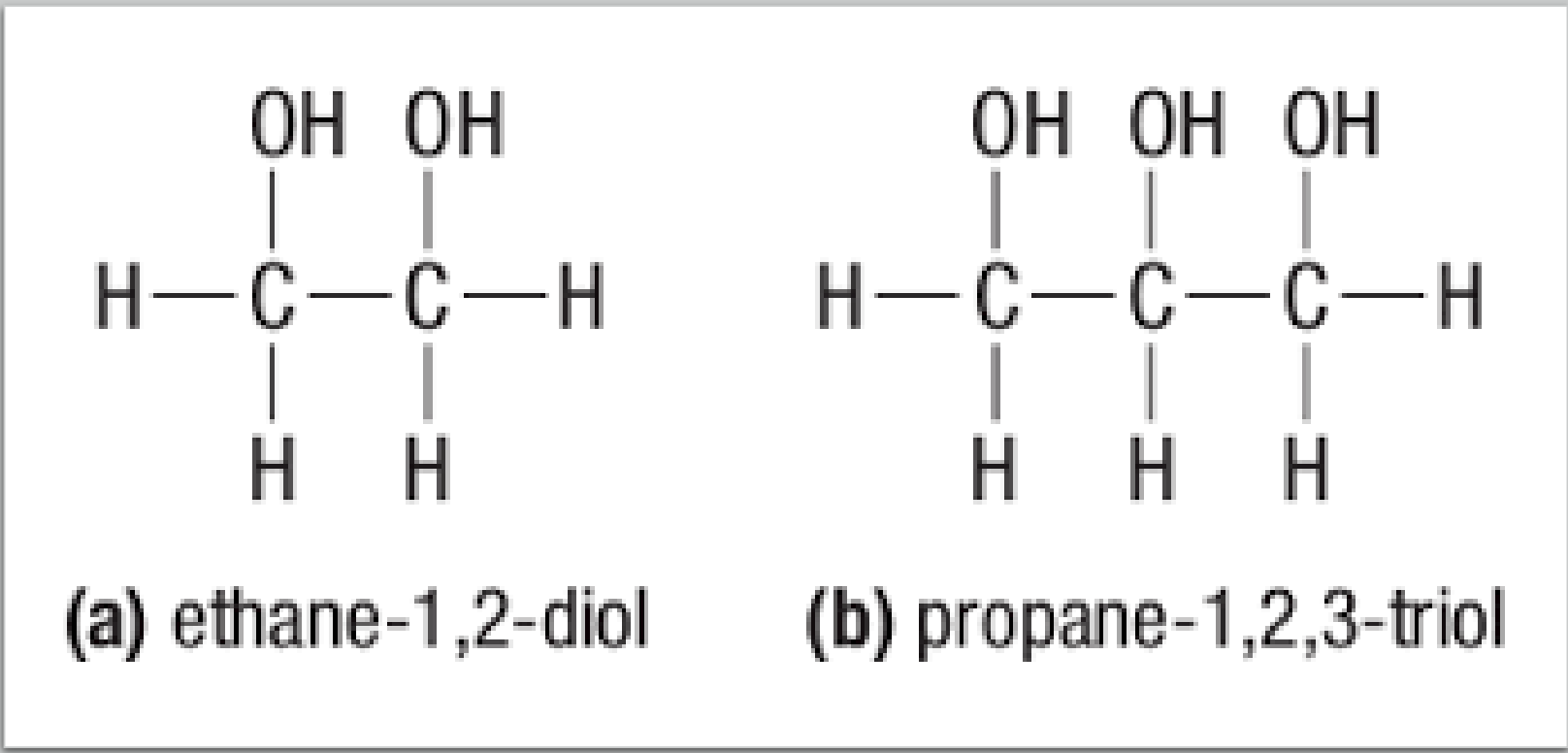
- Polyalcohols: alcohols with more than one hydroxyl group
- Cyclic alcohols: cyclohexanol
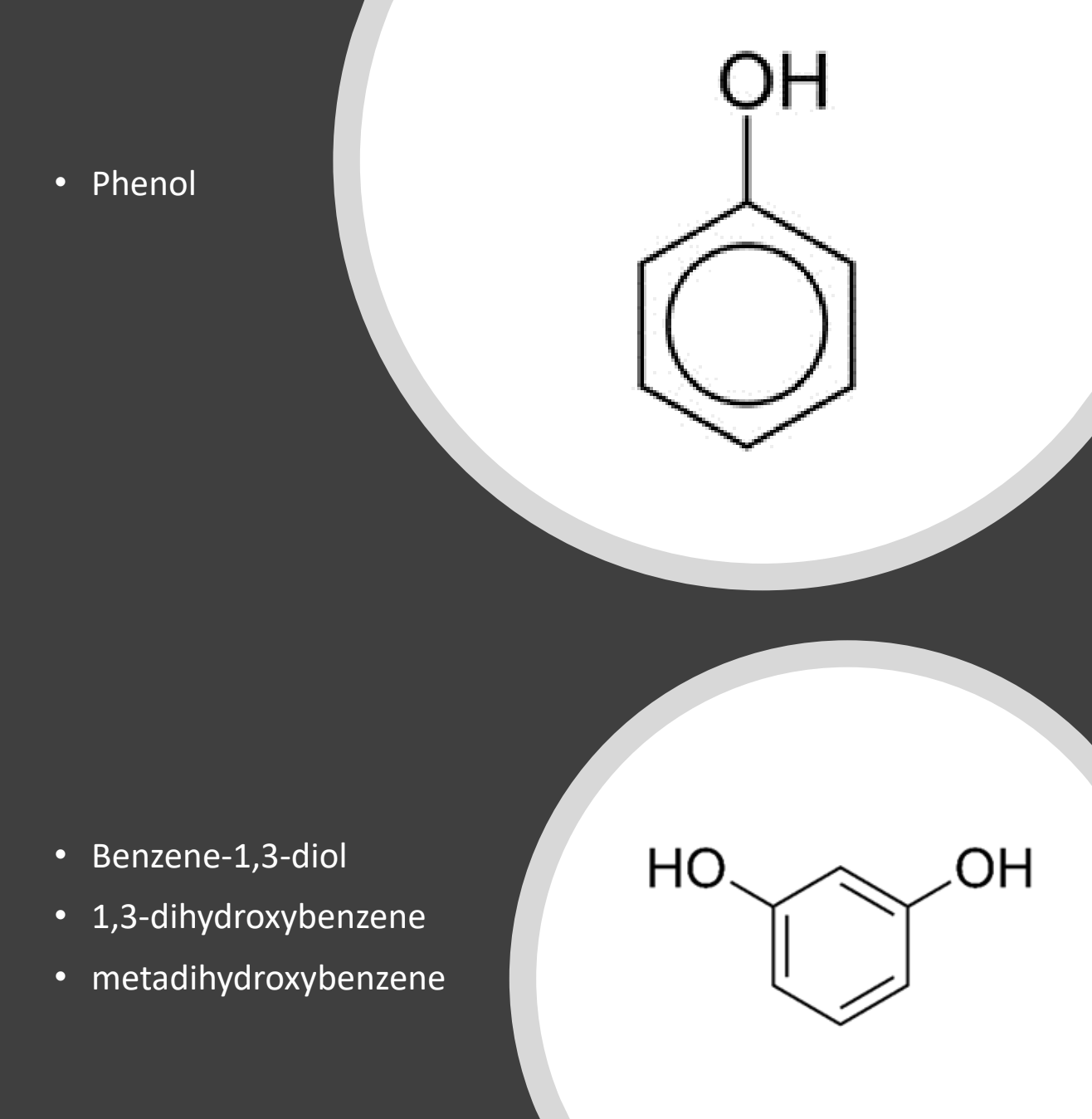
- Aromatic alcohols:
Properties of Alcohols
- Alcohols have a much higher boiling point than alkanes because of the hydroxyl group
- This makes the alcohol molecule polar and allows them to form hydrogen bonds, increasing the intermolecular forces
- Small alcohols have a high solubility in polar solvents (water)
- In long-chain alcohols, the hydrocarbon portion is nonpolar making larger alcohols good solvents for non-polar compounds.
Reactions
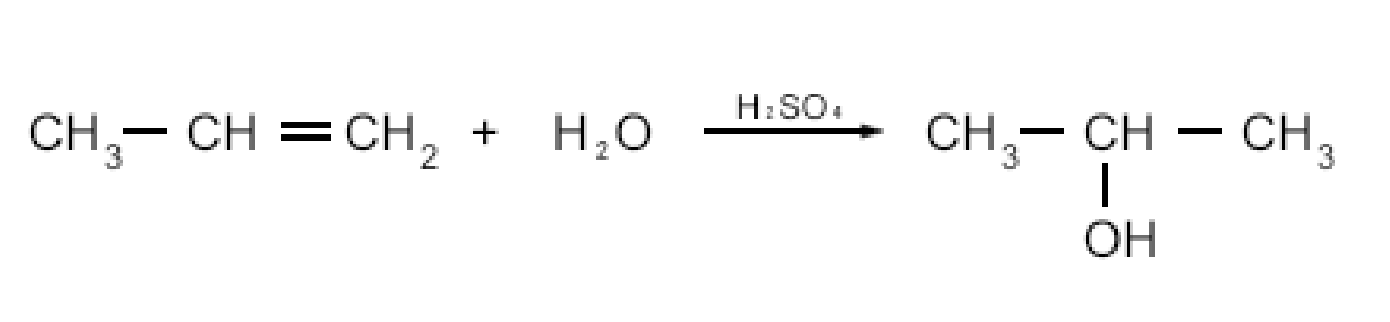
- Hydration of alkenes
* Requires a catalyst such as H2SO4
* Addition reaction
* Makes 2 & 3 alcohols (Markovnikov’s rule)
- Hydration of alkyl halides
* Substitution reaction
* Makes 1 alcohols

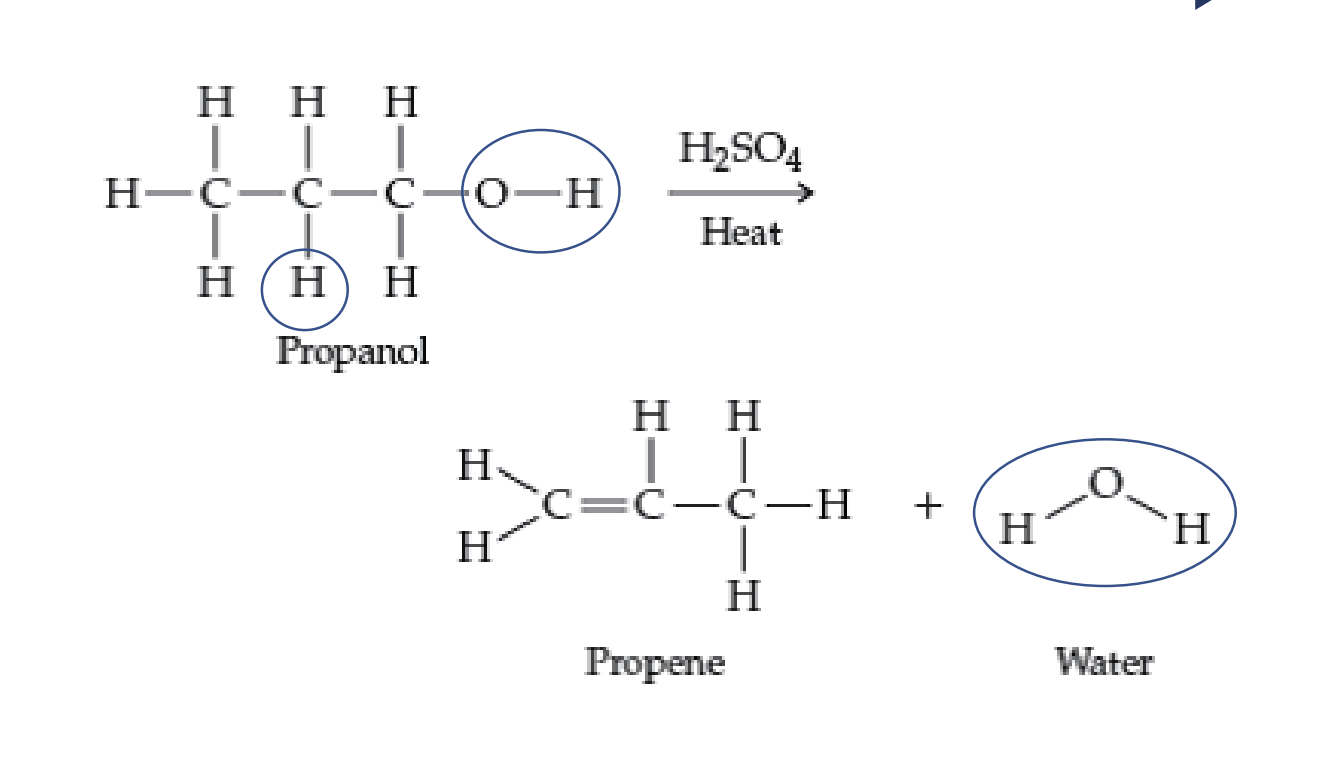
- Elimination/Dehydration
* Prepares alkenes from alcohols
* Requires catalyst (H2SO4)
Combustion of Alcohols