d and f block (transition metal )
Transition elements are a group of metals that are studied separately from non-transition elements.
However, the theory of valence that applies to non-transition elements can also be applied to transition elements.
Precious metals like silver, gold, and platinum, as well as industrially important metals like iron, copper, and titanium, belong to the transition metals series.
The d-block of the periodic table, which is located between the s-block and p-block, contains the transition elements.
The d-orbitals of the penultimate energy level of atoms receive electrons, resulting in four rows of transition metals: 3d, 4d, 5d, and 6d.
The electronic configuration of the outer orbitals of these elements is generally (n-1)d1-10ns1-2, except for Pd which has a configuration of 4d105s0.
The inner d orbitals can have one to ten electrons, while the outermost ns orbital can have one or two electrons.
However, there are exceptions to this generalization due to small energy differences between the d and ns orbitals and the stability of half and completely filled sets of orbitals.
For example, Cr has a configuration of 3d5 4s1 instead of 3d44s2
and Cu has a configuration of 3d104s1 instead of 3d94s2.
The electronic configurations of outer orbitals of Zn, Cd, Hg, and Cn are represented by the general formula (n-1) d10ns2.
These elements have completely filled d orbitals in their ground state as well as in their common oxidation states.
Therefore, they are not considered transition elements.
The d orbitals of transition elements have a greater influence on the atoms or molecules surrounding them compared to the s and p orbitals.
Ions with the same dn configuration have similar magnetic and electronic properties.
Transition elements with partly filled d orbitals exhibit characteristic properties such as multiple oxidation states, formation of colored ions, and complex formation with ligands.
Transition metals and their compounds are also catalytic and show paramagnetic behavior.
The properties of transition elements in a horizontal row are more similar to each other compared to non-transition elements.
However, there are also some group similarities.
The transition elements generally exhibit typical metallic properties such as high tensile strength, ductility, malleability, high thermal and electrical conductivity, and metallic lustre.
However, exceptions to this include Zn, Cd, Hg, and Mn, which do not have typical metallic structures at normal temperatures.
The transition metals have high melting and boiling points, which can be attributed to the involvement of a greater number of electrons from the (n-1) d orbitals in addition to the ns electrons in interatomic metallic bonding.
The melting points of these metals rise to a maximum at d5, except for anomalous values of Mn and Tc, and then fall regularly as the atomic number increases.
These metals also have high enthalpies of atomization
Metals with a higher enthalpy of atomisation tend to have stronger interatomic interactions and stronger bonding.
The second and third series of metals have greater enthalpies of atomisation compared to the first series, which leads to more frequent metal-metal bonding in compounds of heavy transition metals.
The number of valence electrons in a metal affects the strength of bonding.
The lattice structures of transition metals vary, with some metals adopting hexagonal close-packed (hcp) or body-centered cubic (bcc) structures.
The standard electrode potential of a metal is influenced by its enthalpy of atomisation.
n a given series of ions with the same charge, there is a progressive decrease in radius with increasing atomic number.
This is because each time the nuclear charge increases by unity, a new electron enters a d orbital.
The shielding effect of a d electron is not very effective, so the net electrostatic attraction between the nuclear charge and the outermost electron increases, causing the ionic radius to decrease.
The same trend is observed in the atomic radii of a given series, although the variation within a series is relatively small.
When comparing the atomic sizes of one series with the corresponding elements in another series, there is an increase in atomic sizes from the first (3d) to the second (4d) series of elements.
However, the radii of the third (5d) series are virtually the same as those of the corresponding members of the second series.
This phenomenon is due to the intervention of the 4f orbitals, which must be filled before the 5d series of elements begins.
The filling of the 4f orbitals before the 5d orbitals results in a regular decrease in atomic radii, known as Lanthanoid contraction, which compensates for the expected increase in atomic size with increasing atomic number.
The lanthanoid contraction refers to the decrease in atomic and ionic radii from lanthanum to lutetium.
This contraction is caused by the imperfect shielding of one electron by another in the same sub-shell.
However, the shielding of one 4f electron by another is less effective than the shielding of one d electron by another.
As a result, there is a regular decrease in the size of the entire 4f n orbitals as the atomic number increases.
This decrease in size leads to an increase in atomic density.
The lanthanoid contraction is responsible for the similar radii and properties observed in the second and third d series of elements, such as Zr and Hf.
The lanthanoid contraction is caused by the imperfect shielding of one electron by another in the same set of orbitals. The shielding of one 4f electron by another is less effective than that of one d electron by another. As the nuclear charge increases along the series, there is a regular decrease in the size of the entire 4f orbitals. This decrease in metallic radius, coupled with an increase in atomic mass, leads to a general increase in the density of these elements.
IONISATION ENTHALPY
The ionisation enthalpy of transition elements increases along each series from left to right due to an increase in nuclear charge.
The increase in ionisation enthalpy is less steep compared to non-transition elements.
The first ionisation enthalpy generally increases, but the increase in the second and third ionisation enthalpies for successive elements is much higher along a series.
Transition elements are located in the d-block of the periodic table and have electronic configurations of (n-1)d1-10ns1-2.
The electronic configurations of transition elements can have exceptions due to small energy differences between (n-1)d and ns orbitals.
The highest oxidation number in transition metal oxides is attained in Sc2O3 to Mn2O7, and oxygen can stabilise high oxidation states better than fluorine.
The stability of different oxidation states in transition elements can be explained by factors such as the energy difference between d orbitals and the ability of oxygen to form multiple bonds.
Transition metal ions can have magnetic moments due to the presence of unpaired electrons, and the magnetic moment increases with the number of unpaired electrons.
The stability of transition metal ions in different oxidation states can be determined by comparing their experimental magnetic moments with those calculated using the "spin-only" formula.
Copper ions can exist in both Cu+ and Cu2+ oxidation states, but Cu2+ is more stable due to its more negative ∆hydHV and the ability of oxygen to stabilise high oxidation states.
The irregular variation of ionisation enthalpies in the first series of transition elements is attributed to the varying stability of different d configurations.
The increasing oxidising power in the series VO2+ < Cr2O72- < MnO4- is due to the increasing stability of the lower species to which they are reduced.
Exchange energy stabilises energy states in electrons.
exchange energy is proportional to the total number of possible pairs of parallel spins in degenerate orbitals.
The lowest energy state occurs when multiple electrons occupy a set of degenerate orbitals with maximum single occupation and parallel spins.
The loss of exchange energy increases stability.
The ionisation enthalpy of Mn+ is lower than that of Cr+.
The ionisation enthalpy of Fe2+ is lower than that of Mn2+.
The third ionisation enthalpy of Fe is lower than that of Mn.
reason 😀
The ionisation enthalpy of Mn+ is lower than that of Cr+ because Mn+ has a 3d54s1 configuration while Cr+ has a d5 configuration.
Similarly, the ionisation enthalpy of Fe2+ is lower than that of Mn2+ because Fe2+ has a d6 configuration and Mn2+ has a 3d5 configuration.
Therefore, the third ionisation enthalpy of Fe is lower than that of Mn.
The lowest common oxidation state of these metals is +2. The second ionisation enthalpy plays a dominant role in the formation of M2+ ions, particularly for Cr and Cu with d5 and d10 configurations, respectively.
The ionisation enthalpy of Zn is low because the removal of one 4s electron results in a stable d10 configuration.
The trend in the third ionisation enthalpies shows the difficulty of removing an electron from d5 and d10 ions.
The third ionisation enthalpies are generally high, and the high values for copper, nickel, and zinc explain why it is difficult to obtain oxidation states greater than two for these elements.
However, the relative stabilities of oxidation states are complex and cannot be easily generalised based on ionisation enthalpies.
Transition elements, such as those in the first row, can exhibit multiple oxidation states due to the incomplete filling of d orbitals.
The common oxidation states of the first row transition elements are as follows:
Scandium (Sc): +3
Titanium (Ti): +2, +3, +4
Vanadium (V): +2, +3, +4, +5
Chromium (Cr): +2, +3, +6
Manganese (Mn): +2, +3, +4, +6, +7
Iron (Fe): +2, +3
Cobalt (Co): +2, +3
Nickel (Ni): +2
Copper (Cu): +1, +2
Zinc (Zn): +2
Transition elements exhibit a wide range of oxidation states, with the greatest number of oxidation states occurring in or near the middle of the series
Manganese, for example, exhibits oxidation states from +2 to +7.
The lesser number of oxidation states at the extreme ends of the series is due to either too few electrons to lose or share (Sc, Ti) or too many d electrons, resulting in fewer available orbitals for electron sharing (Cu, Zn).
Scandium(II) is virtually unknown, and titanium (IV) is more stable than Ti(III) or Ti(II).
Zinc only exhibits an oxidation state of +2, as it does not have any d electrons.
The maximum oxidation states of reasonable stability correspond to the sum of the s and d electrons up to manganese, followed by a decrease in stability for higher oxidation states.
The variability of oxidation states in transition elements is due to the incomplete filling of d orbitals, resulting in oxidation states that differ by unity.
In contrast, oxidation states of non-transition elements typically differ by a unit of two. In the d-block elements, there is an interesting trend among groups 4 through 10, where the lower oxidation states are favored by the heavier members.
However, in the p-block, the opposite trend is observed.
Low oxidation states can also be found in complex compounds with ligands capable of π-acceptor character in addition to σ-bonding, such as in Ni(CO)4 and Fe(CO)5 where the oxidation state of nickel and iron is zeroEle
The EV (M3+/M2+) values for transition metals vary, reflecting the stability of different oxidation states.
The low value for Sc reflects the stability of Sc3+ with a noble gas configuration.
The high value for Zn is due to the removal of an electron from the stable d10 configuration of Zn2+. Mn2+ (d5) is particularly stable, while Fe3+ (d5) is less stable.
The low value for V is related to the stability of V2+ (half-filled t2g level).
The highest oxidation numbers are achieved in TiX4 (tetrahalides), VF5, and CrF6.
Mn can reach a +7 state in MnO3F, while Fe and Co have trihalides (FeX3 and CoF3).
Fluorine can stabilize high oxidation states due to higher lattice energy or higher bond enthalpy in covalent compounds like VF5 and CrF6.
Fluorides are unstable in low oxidation states like VX2 (X = Cl, Br, or I).
Copper (II) halides are known, except for the iodide.
Copper (I) compounds are unstable in aqueous solution and undergo disproportionation. The stability of Cu2+ (aq) is due to its more negative ∆hydHV compared to Cu+.
Oxygen can stabilize high oxidation states in oxides, and the highest oxidation number is attained in Sc2O3 to Mn2O7.
Oxygen can form multiple bonds to metals, making it superior to fluorine in stabilizing high oxidation states.
Transition metals exhibit a wide range of chemical reactivity.
Most of them can dissolve in mineral acids, although some are considered noble and are unaffected by acids.
The first series of transition metals, except for copper, are relatively more reactive and can be oxidized by 1M H+.
However, the rate of reaction with oxidizing agents like hydrogen ion (H+) can be slow.
Titanium and vanadium, for example, are passive to dilute non-oxidizing acids at room temperature.
The EV values for M2+/M indicate a decreasing tendency to form divalent cations across the series.
This trend is related to the increase in the sum of the first and second ionization enthalpies.
It is worth noting that the EV values for Mn, Ni, and Zn are more negative than expected from the general trend.
The stability of half-filled d subshell in Mn2+ and completely filled d subshell in zinc is related to their Ee values, while for nickel, the Eo value is related to the highest negative enthalpy of hydration.
In terms of redox reactions, Mn3+ and Co3+ ions are the strongest oxidizing agents in aqueous solutions, while Ti2+, V2+, and Cr2+ ions are strong reducing agents that can liberate hydrogen from a dilute acid.
Paramagnetism is a type of magnetic behavior observed when a magnetic field is applied to substances.
Paramagnetic substances are attracted to the applied field.
This behavior arises from the presence of unpaired electrons, which have a magnetic moment associated with their spin and orbital angular momentum.
In the case of transition metals, the contribution of orbital angular momentum is negligible, so the magnetic moment is determined by the number of unpaired electrons.
The magnetic moment increases with the number of unpaired electrons.
The observed magnetic moment can provide information about the number of unpaired electrons in an atom, molecule, or ion.
The magnetic moments calculated from the "spin-only" formula
magnetic moment (μ) is equal to the number of unpaired electrons (n) multiplied by 2, and then multiplied by the Bohr magneton (BM).
The Bohr magneton is a unit of magnetic moment.
When an electron is excited from a lower energy d orbital to a higher energy d orbital, it absorbs light of a specific frequency.
The color observed corresponds to the complementary color of the absorbed light.
The frequency of the absorbed light is determined by the nature of the ligand.
Formation of complex compounds is a common characteristic of transition metals.
Complex compounds are formed when metal ions bind to anions or neutral molecules, resulting in complex species with unique properties.
Transition metals are particularly prone to forming complex compounds due to their smaller size, high ionic charges, and availability of d orbitals for bond formation.
The colors observed in aqueous solutions of transition metal ions depend on the nature of the ligand.
Water molecules, for example, can act as ligands and produce specific colors for different metal ions.
Transition metals and their compounds are known for their catalytic activity.
This is due to their ability to adopt multiple oxidation states and form complexes.
Catalysts at a solid surface involve the formation of bonds between reactant molecules and atoms of the catalyst surface.
This increases the concentration of reactants at the catalyst surface and weakens the bonds in the reacting molecules, lowering the activation energy.
Transition metal ions can change their oxidation states, making them more effective as catalysts.
For example, iron(III) catalyzes the reaction between iodide and persulfate ions.
The catalytic action can be explained by the following reaction:
2 Fe3+ + 2 I– → 2 Fe2+ + I2
2 Fe2+ + S2O82– → 2 Fe3+ + 2SO42–
Interstitial compounds are formed when small atoms like H, C, or N are trapped inside the crystal lattices of metals.
These compounds are usually non-stoichiometric and do not correspond to any normal oxidation state of the metal.
They have high melting points, are very hard, retain metallic conductivity, and are chemically inert.
Alloys are blends of metals that are formed by mixing different components.
In homogeneous solid solution alloys, the atoms of one metal are randomly distributed among the atoms of the other metal.
These alloys are formed when the metallic radii of the atoms are within about 15 percent of each other.
Transition metals, which have similar radii and other characteristics, readily form alloys.
These alloys are often hard and have high melting points.
Some well-known alloys include ferrous alloys (chromium, vanadium, tungsten, molybdenum, and manganese) that are used in the production of various steels and stainless steel.
Additionally, alloys of transition metals with non-transition metals, such as brass (copper-zinc) and bronze (copper-tin), are also of significant industrial importance.
The oxides of transition metals are formed by the reaction of metals with oxygen at high temperatures.
Most metals form MO oxides, which are ionic. ( except scandium )
The highest oxidation number in the oxides is attained in Sc2O3 to Mn2O7.
However, beyond group 7, no higher oxides of iron above Fe2O3 are known.
In addition to the oxides, the oxocations stabilize V^v as VO2+, V^IV as VO2+, and TiIV as TiO2+.
As the oxidation number of a metal increases, the ionic character decreases.
For example, Mn2O7 is a covalent green oil, while CrO3 and V2O5 have low melting points and exhibit acidic character.
The oxides of manganese, chromium, and vanadium show different properties.
Manganese(VII) oxide (Mn2O7) gives rise to permanganic acid (HMnO4), while chromium(VI) oxide (CrO3) forms chromic acid (H2CrO4) and dichromic acid (H2Cr2O7).
Vanadium pentoxide (V2O5) is amphoteric, meaning it can act as both an acid and a base.
It can form vanadate ions (VO43-) as well as vanadium(IV) ions (VO2+).
On the other hand, chromium(III) oxide (Cr2O3) is amphoteric.
Potassium dichromate is an important chemical used in the leather industry and as an oxidant for preparing azo compounds.
It is prepared from chromate by fusion of chromite ore with sodium or potassium carbonate in the presence of air.
Sodium dichromate is more soluble than potassium dichromate, so potassium dichromate is prepared by treating the solution of sodium dichromate with potassium chloride.
The chromates and dichromates are interconvertible in aqueous solution depending on the pH, and the oxidation state of chromium in both chromate and dichromate is the same.
The chromate ion is tetrahedral whereas the dichromate ion consists of two tetrahedra sharing one corner with a Cr–O–Cr bond angle of 126°.
Sodium and potassium dichromates are strong oxidizing agents, with the sodium salt having greater solubility in water and being extensively used as an oxidizing agent in organic chemistry.
Potassium dichromate is used as a primary standard in volumetric analysis.
In acidic solution, its oxidizing action can be represented by the half-reaction:
Cr2O72– + 14H+ + 6e– → 2Cr3+ + 7H2O (EV = 1.33V).
Acidified potassium dichromate can oxidize iodides to iodine, sulfides to sulfur, tin(II) to tin(IV), and iron(II) salts to iron(III).
The full ionic equation can be obtained by adding the half-reaction for potassium dichromate to the half-reaction for the reducing agent,
such as: Cr2O72– + 14H+ + 6Fe2+ → 2Cr3+ + 6Fe3+ + 7H2O.I
Potassium permanganate (KMnO4) is prepared by fusing MnO2 with an alkali metal hydroxide and an oxidizing agent like KNO3.
This reaction produces dark green K2MnO4, which then disproportionates in a neutral or acidic solution to form permanganate.
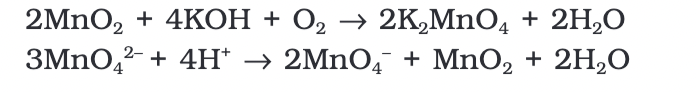
Commercially, potassium permanganate is prepared through the alkaline oxidative fusion of MnO2 followed by the electrolytic oxidation of manganate (VI) ions.
o
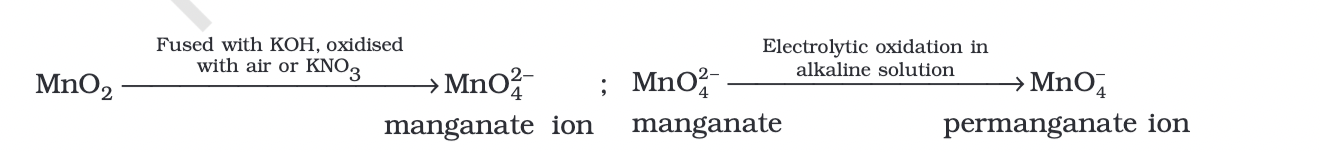 nisatio
nisatioin the laboratory, a manganese (II) ion salt is oxidized by peroxodisulphate to permanganate.
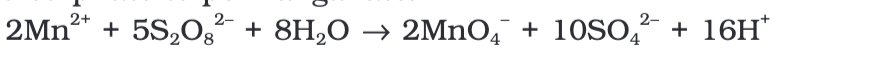 Enthalpies-
Enthalpies-Potassium permanganate forms dark purple crystals and is not very soluble in water. When heated, it decomposes.
It has intense color and diamagnetism.
The manganate and permanganate ions are tetrahedral.
their bonding is formed by the overlap of p orbitals of oxygen with d orbitals of manganese.
The manganate ion is paramagnetic because it has one unpaired electron, while the permanganate ion is diamagnetic because it does not have any unpaired electrons.
Acidified permanganate solution oxidizes oxalates, iron(II), nitrites, and iodides. The half-reactions of reductants involve the production of carbon dioxide, iron(III), nitrates, and iodine.
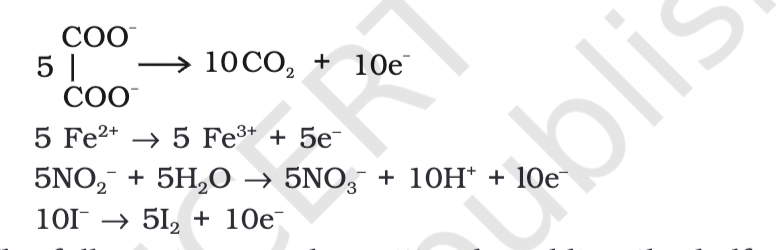 Ionisation
IonisationThe reduction of permanganate to manganate, manganese dioxide, and manganese(II) salt can be represented by the following half-reactions:
MnO4– + e– → MnO42– (EV = + 0.56 V)
MnO4– + 4H+ + 3e– → MnO2 + 2H2O (EV = + 1.69 V)
MnO4– + 8H+ + 5e– → Mn2+ + 4H2O (EV = + 1.52 V)
The hydrogen ion concentration of the solution is an important factor in influencing the reaction.
Although many reactions can be understood by considering redox potential, the kinetics of the reaction also play a role.
In acid solutions, potassium permanganate can oxidize iodine from potassium iodide and convert Fe2+ ions to Fe3+ ions.
