Polysaccharides: Cellulose and Chitin
Cellulose
Cellulose is a complex carbohydrate made of a polymer of β-glucose molecules. The β-1,4 glycosidic linkages result in the −CHX2OH groups being on opposite sides of the chain of adjacent glucose molecules.
Within a cellulose chain, adjacent glucose molecules are rotated 180° relative to each other. This means that OH groups are aligned and a water molecule can be removed to form a glycosidic bond.
Therefore, hydrogen bonds do not form between glucose molecules within the same chain, but between glucose molecules in different chains.
The hydrogen bonds form cross-linkages which hold the chains together. This makes cellulose form into long threads called microfibrils.
Cellulose is completely insoluble and the microfibrils are laid down in overlapping layers in plant cell walls.

Cellulose is called a structural polysaccharide. It is very difficult to digest because of the very high numbers of hydrogen bonds between the chains of beta glucose. This also gives cellulose very high tensile strength; it is difficult to break when stretched. This means that cells with cellulose in their cell wall are more resistant to osmotic lysis (they are not likely to burst because cellulose stops too much water entering the cell).
Chitin
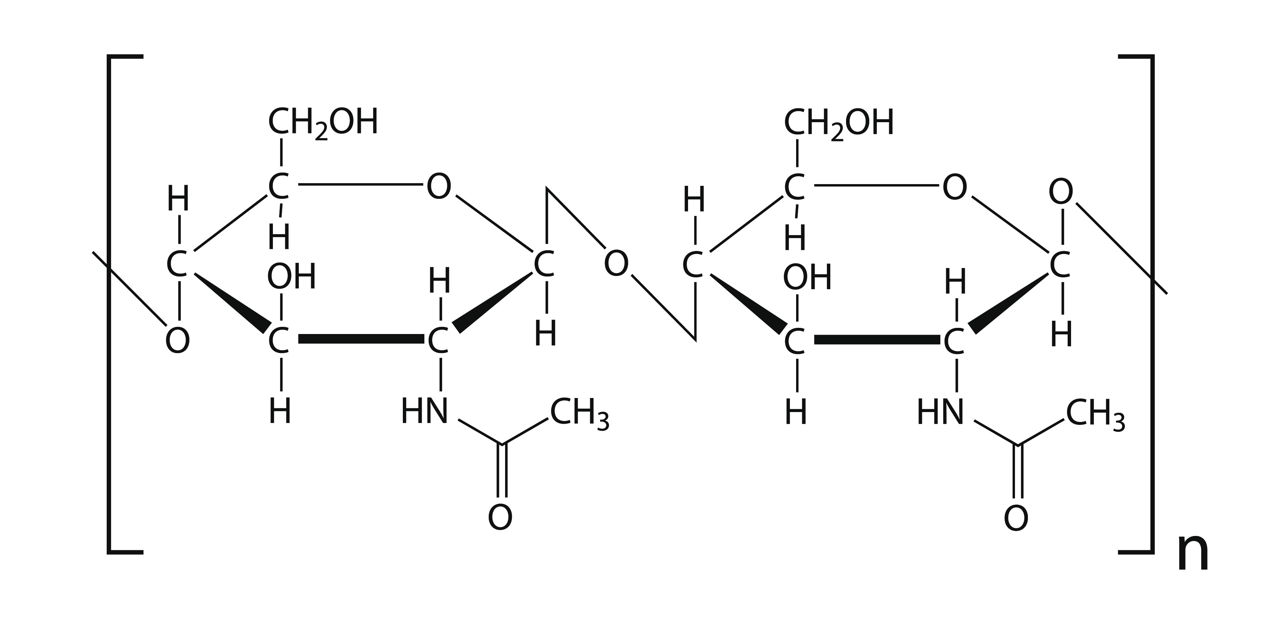
Chitin is found in the cell walls of fungi and in the exoskeletons of insects. It is not a true polysaccharide as it contains the element nitrogen – it is called a heteropolysaccharide.
It has a similar structure and function as cellulose but because it contains side groups containing N, more hydrogen bonds can form. Chitin microfibrils, therefore, have greater tensile strength than those of cellulose.
The diagram shows two of the monosaccharides (N-acetyl glucosamine) found in chitin joined together by a β-glycosidic bond.