Hematology - MSU PSL 310
Compositon of Blood
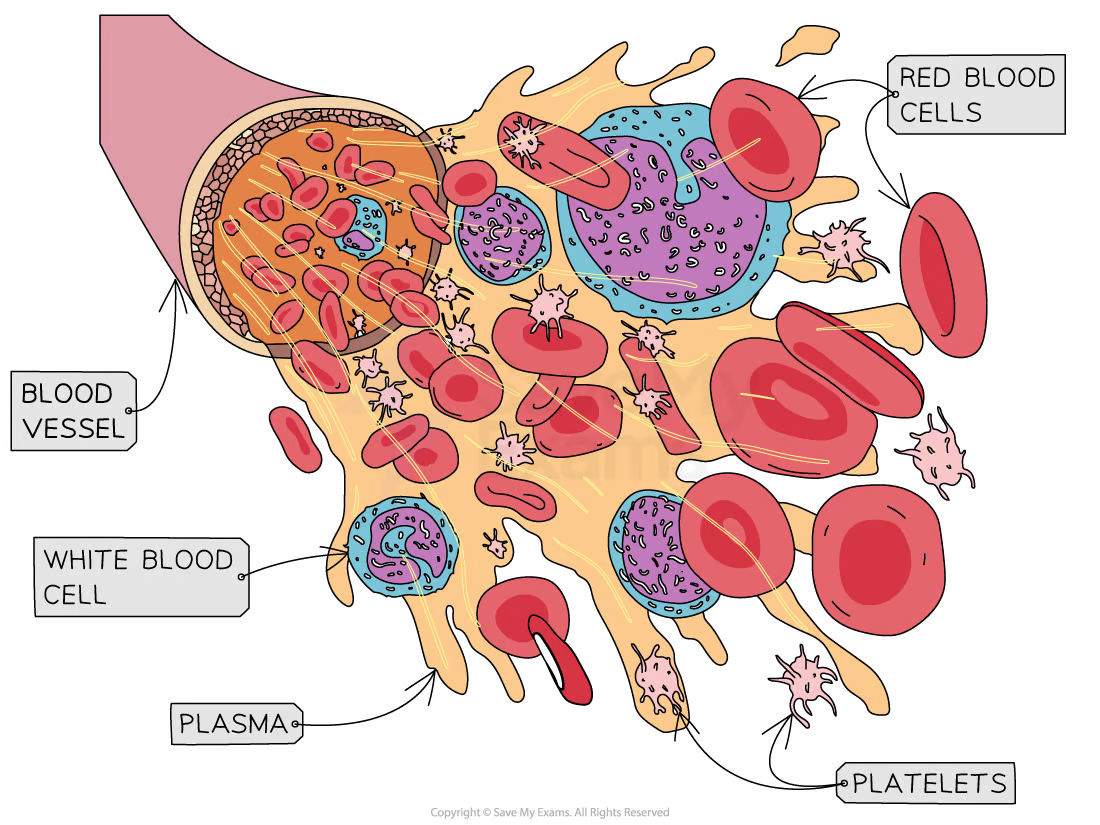
Plasma (55%)
Composition
Water (90%)
Plasma proteins (7%)
Albumins (60%) → major contributor to osmotic pressure regulation in plasma, also transport protein for lipids, and steroid hormones
Globulin (35%) → transport protein for ions (ex: iron), hormones (TeBG → testosterone binding globulin), lipids and had immune functions
Fibrinogen (4%) → inactive protein essential for the clotting system, can be converted into insoluble fibrin (active from of fibrinogen)
Regulatory proteins (<1%) → enzymes, proenzymes and hormones
Other Solutes (1%)
Electrolytes → normal extracellular fluid ion composition for vital cellular activities. Also contribute to osmotic pressure
most common ones → sodium (Na+), potassium (K+), chloride (Cl−), magnesium (Mg2+), calcium (Ca2+), phosphate (P), and bicarbonates (HCO3-)
Organic nutrients → used for ATP production, growth & maintenance of cells, include lipids, carbohydrates, and amino acids
Organic waste → carried to cites for breakdown or excretion, include urea, uric acid, creatine, bilirubin and amonium ions
most of these are nitrogenous wastes
inorganic waste → CO2
Functions
Primary
transport
exchange
Secondary
pH regulation
fluid volume regulation
thermoregulation
90% of it is water → water can absorve a lot of energy without changing temp., aka vasodilation/-constriction
coagulation
ability to form blood clots
immunity
Formed elements (45%)
White blood cells (WBC) (0.1%)
Nucleophils (50%)
most abundant type of WBC
Lymphocytes (25%)
same size as RBC
Monocytes (2-8%)
migrate btw endothelial cells, fuse to make macrophages and gobble up oxidized LDL to form plaques (atherosclerosis)
Eosinophils (2-4%)
Basophils (<1%)
least numerous
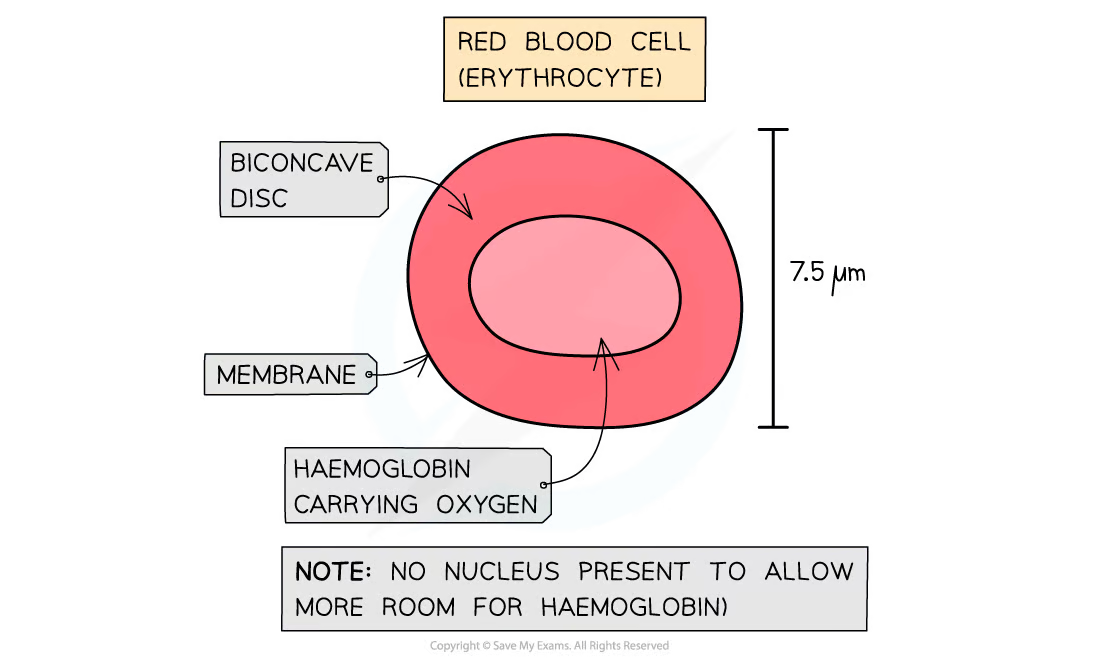
Red blood cells (RBC)(99%)
make about 1/3 of all body cells (ab 30 trillion in adult body)
only living cell without nucleus (anucleate) or organelles
make ATP through glycolisis
contain hemoglobin
O2 transport protein
RBC also referred to as “hemoglobin sacs”
biconcave shape
increases SA
barely any hemoglobin in the middle making it not pick up any stain leading to making it look like they have hole in the middle under the microscope
capillaries are about the same size (or smaller) as the diameter of RBC this makes them have to fold to cruze through them, this shape facilitates this
Platelete (0.1%)
cell fragments
huge cells found in bone marrow pinch pieces off their membrane to form platelets
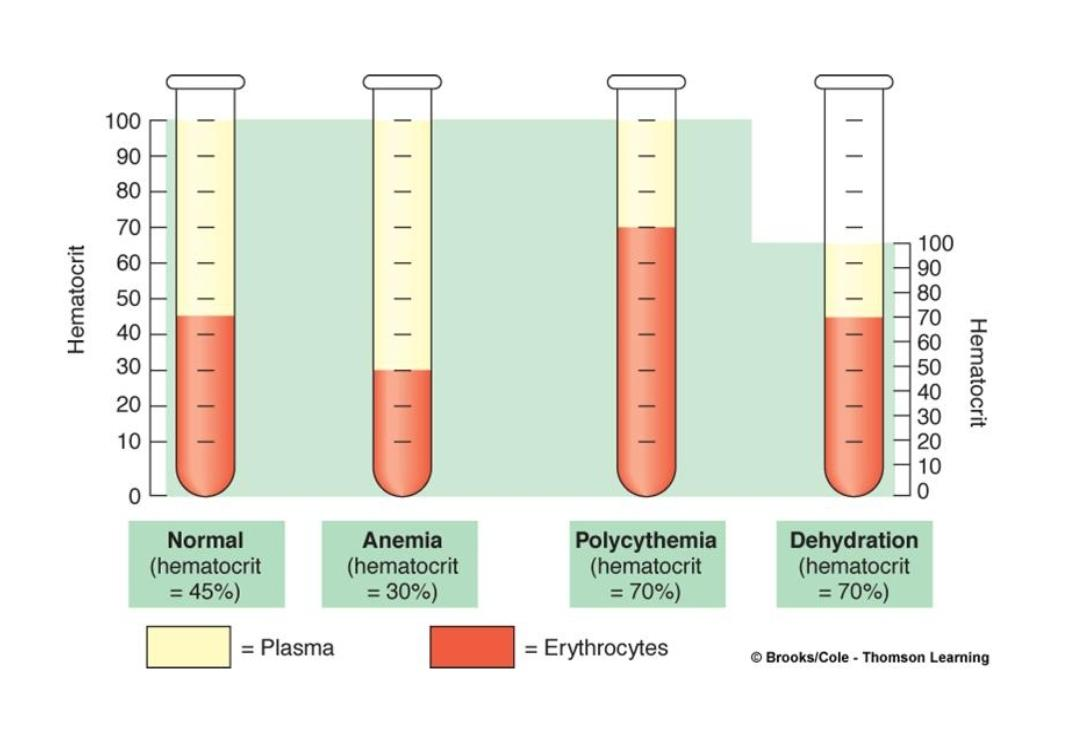
Hematocrit (Hct)
% of a sample of whole blood that is occupied by RBCs
multiple ways to do it
Manual Hct:
derermined by microcentrifugation, followed by dividing the volume of packed RBCs (PCV → packed cell volume) by the total volume of the blood sample
Hct = tot. volume RBC/PCV
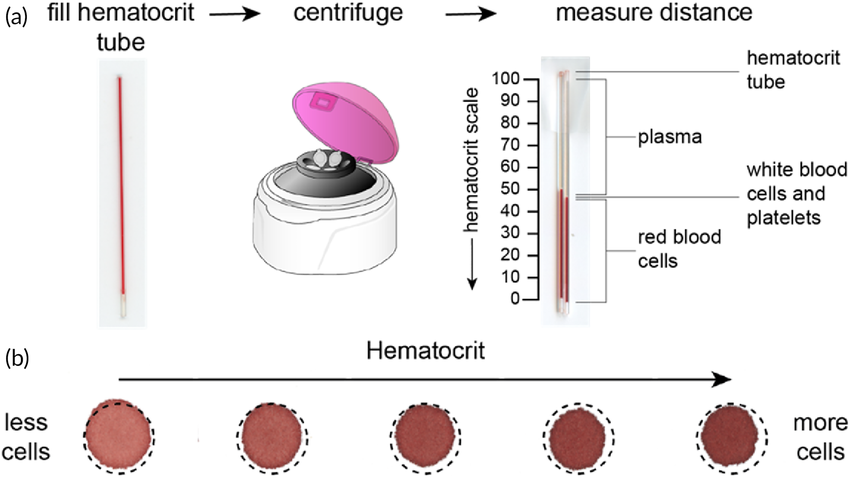
Automated Hct
done through the use of a hematology analyzer
this analyzer counts the # of RBCs and determines their mean corpuscular volume (MCV) then it is multiplied by the # of RBCs to get Hcl
Hct = # RBCs x MCV
Hct values
Males (41-50%) & Females (36-48%)
this difference btw males and females is due to testosterone levels
What affects the values?
androgens
estrogens
erythropoitetin (EPO)
RBC profuction stimmulating hormone
Abnormal Hct typically results from changes in the # of RBCs and or Plasma volume
topHat question: Could you be anemic and have a normal Hct?
Answer: Yes, you can have a normal Hct and be anemic because an anemia can also be caused by issues with hemoglobin or an iron deficiency
Abnormal Hcts
Anemia - 30%
Polycythemia - 70%
primary (polycythemia vera) - bone marrow cancer causing ↑ #RBC and ↑Hct
secondary - normal physiological response to ↓ PO2 causing ↑ #RBC and ↑Hct
used in high altitude training
also artificially done through blood doping
Dehydration - 70%
lack of watter
Red Blood Cells
RBC production (erythropoiesis)
RBC is formed in bone marrow of long bones (sternum, ribs, pelvic bones, and cranium)
cranium bones are composed of two layers of bone and in the middle a section of spongy bone (thats where RBC are cranked out)
Requires iron & amino acids (for hemoglobin synthesis) and coenzyme vitamin B12
Gastric intrinsic factor (GIF) is essential for B12 absorption
↓ GIF → pernicious anemia
treated with B12 injections
Erythropoietin (EPO) → hormone released by chemoreceptive kidney (JG) cells during hypoxic conditions (↓ PO2) which stimulates erythropoiesis
Erythropoiesis is influenced by EPO, Androgens, Estrogen, GH, T3, etc…
Lifespan & destruction
On average the RBC lifespan is about 3-4 months
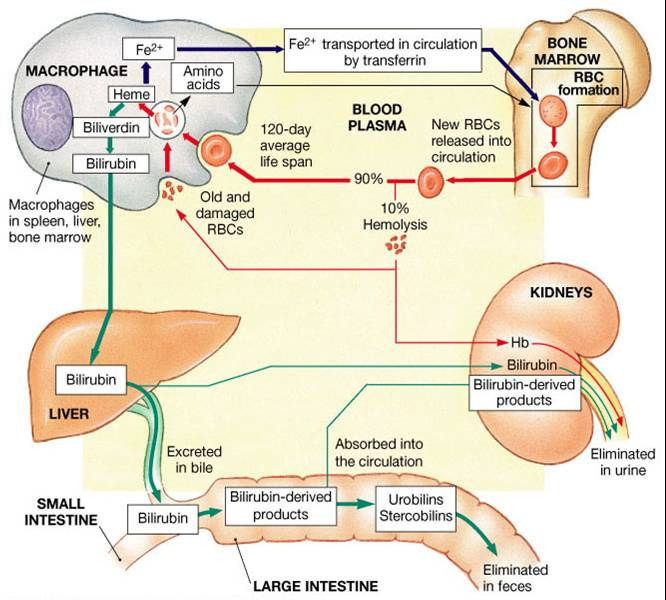
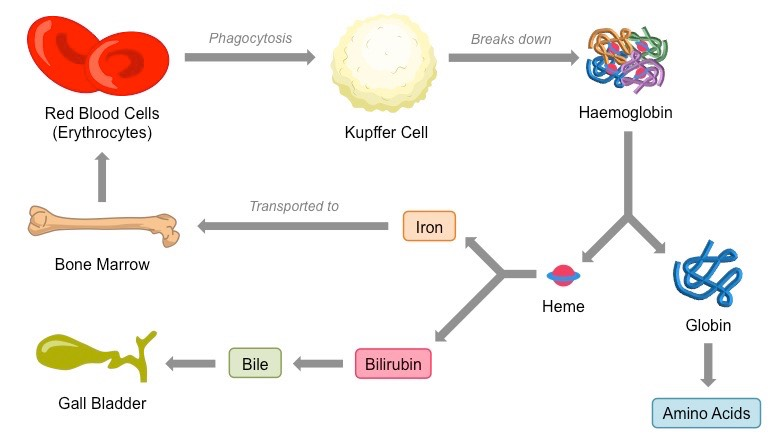
Jaundice
characterized by a yellow hue on the skin and sclera (white part of the eye)
on white skin it shows yellow skin but in dark skin it is more apparent by the yellow sclera
caused by liver unable to convert bilirubin into less toxic forms
seen in sorosis of the liver, and pancreatic, liver and duc cancers
also found in premature babies
liver has not fully developed
treated with UV light (synthesizes bilirubin into less toxic form)
Hemoglobin
4-subunits, quaternaty protein complexed with 4 heme groups (phorphyrin ring-Fe2+)
types
oxyhemoglobin (HgbO2)
deoxyhemoglobin (Hgb)
carbaminohemoglobin (HgbCO2)
carbon monoxide bonded with hemoglobin
carbon monoxide has a higher affinity to heme than oxigen
it’s a competitive inhibitor for it
always outcompetes oxigen, this is why its poisonous
maternal (adult) hemoglobin (HgbA)
fetal hemoglobin (HgbF)
sickle-cell hemoglobin (HgbS)
glycated hemoglobin (HgbA1c)
glucose bound to the n-terminus of the polypeptide chains in hemoglobin
no change in O2 affinity
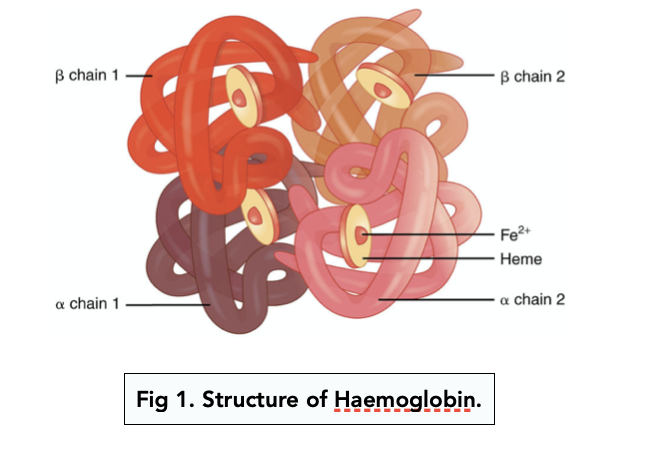
it binds to CO2, H+, 2,3-BPG, NO, CO, glucose, etc…
CO2, H+, 2,3-BPG are byproducts of metabolism that bind to hemoglobin and change the conformation of its 3D shape making it lose its ability to bind to O2
binding cites:
CO2 → polypeptide chain
forms an amine
H+ → some R- groups in the amino acids
when it binds it changes the confirmation of hemoglobin decreasing O2 affinity (due to change in pH)
2,3-bisphosphoglycerate (BPG) → center of hemoglobin
two conformations
Tense (T-hemoglobin)
low O2 affinity
favors the unloading of O2
induced by CO2, H+, and 2,3-bisphosphoglycerate
Relaxed (R-hemoglobin)
high O2 affinity
favors the loading of O2
Hgbs and Sickle-cell anemia
Inherited point-mutation in DNA coding for hemoglobin
makes it so that it codes for the protein to be nonpolar instead of non-polar thus making it hydrophilic
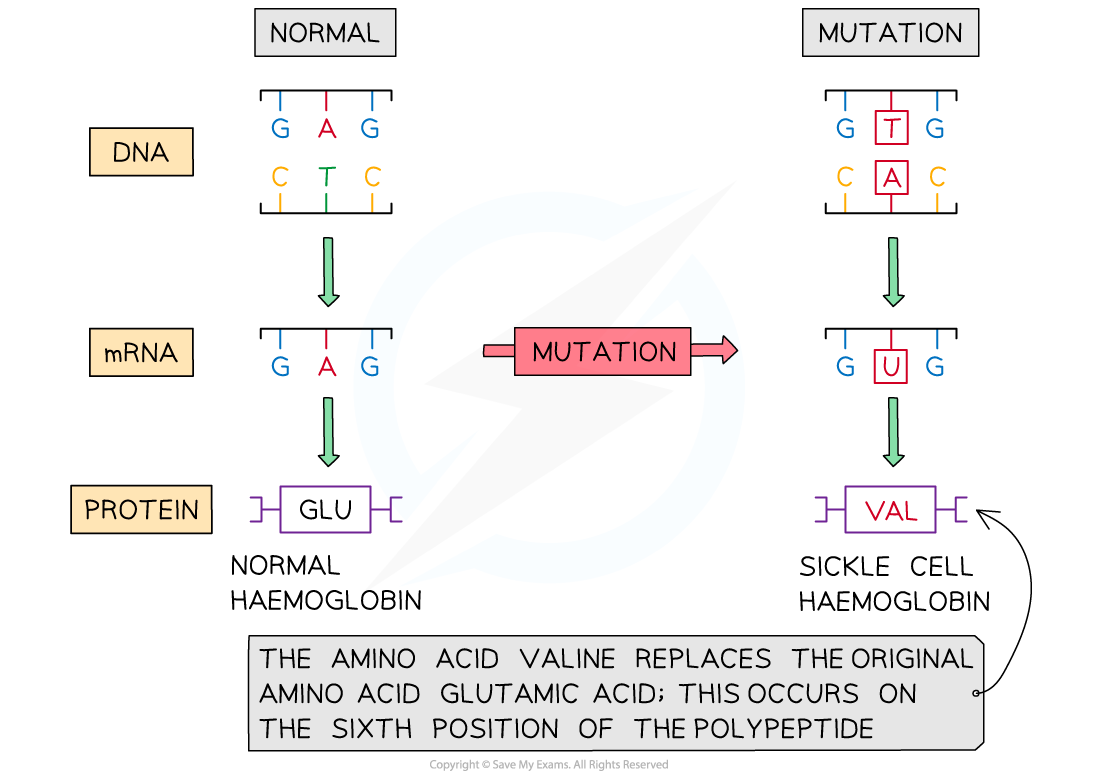
this leads to the sickling of RBCs, sickle-cell crises, premature RBC destruction and anemia
this small change makes the hemoglobin sticky which causes the hemoglobin proteins to stick with each other creating sickle-cell crises
this anemia can cause ischemia, perfusion and lead to organ failure
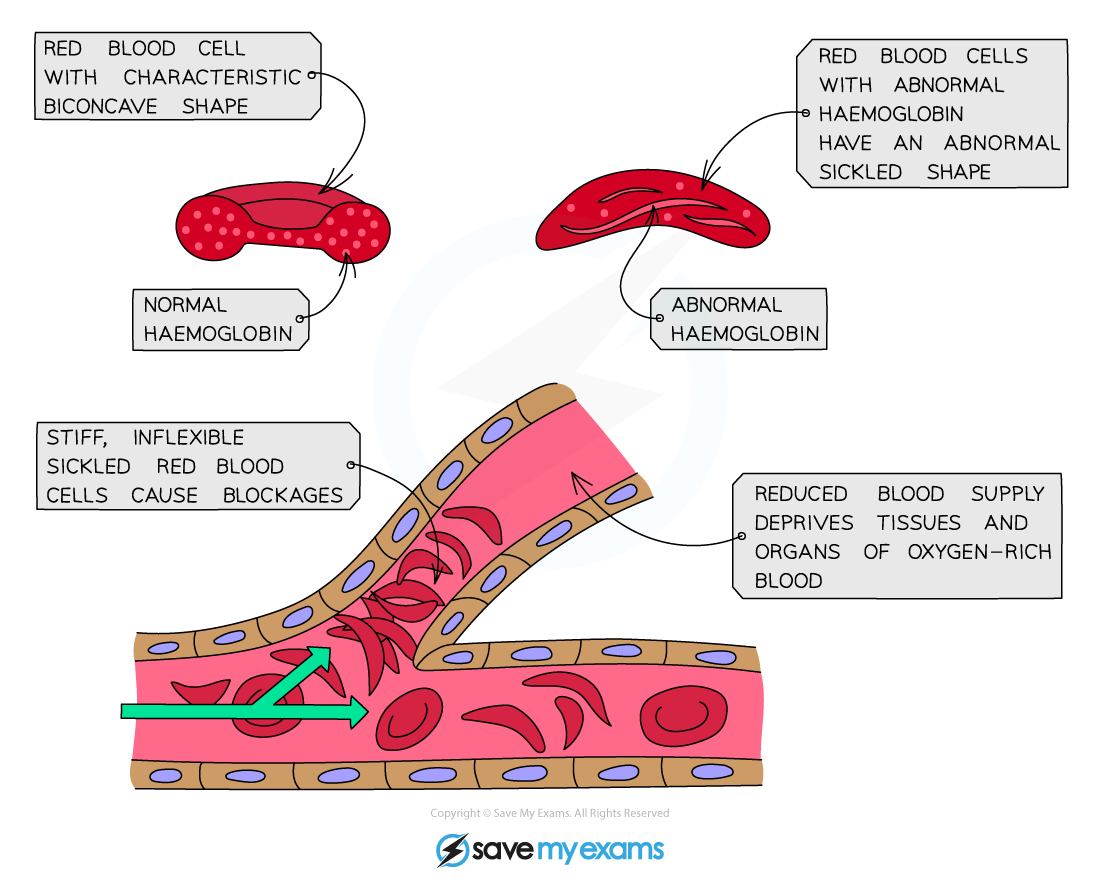
Trait vs disease
Sickle-cell disease (HgbSS or HgbAS) → has the disease
only has sickle cells
treatments:
monthly blood transfusions (most common)
bone-marrow transplants (not common, a few have been completely cured)
gene therapy (currently on the horizon)
Sickle-cell trait (HgbSs or HgbAS) → carries the gene for the disease
also has both sickle and normal RBC
1/13 of African americans in the US are carriers
Malaria and Sickle-cell
malaria is a superimposable disease to Sickle-cell
the malaria parasite uses human RBC to mature (it gets into them)
carriers of the disease have both sickle and normal RBC, meaning that some of their RBC carry low oxygen capacity and it can stick together a little bit
Malaria is unavle to live in those and it messes up their lifecycle
as a result the individuals who are gene carriers are most likely to survive Malaria in comparison to someone who is HgbSS (disease) or HgbAA (normal)
this is why the trait is still in our gene pool → natural selection
those who live pass the gene down to their offspring
HgbA1c and diavetes
HgbA1c is glycated hemoglobin (i.e. covalently bound to monosacharides)
glycated hemoglobin is a high indicator for diavetes (lasts 3-4 months)
RBC blood typing
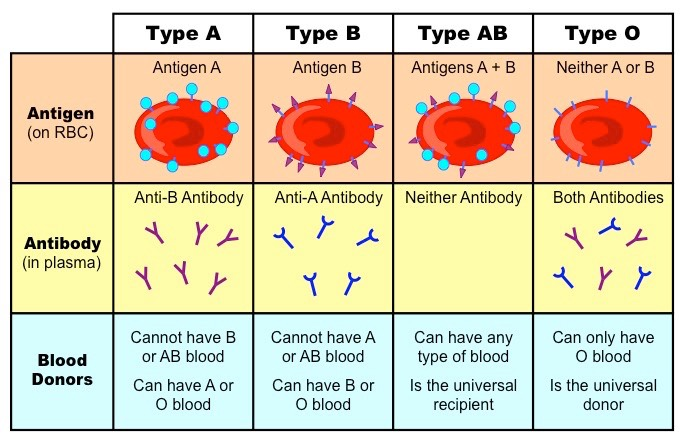
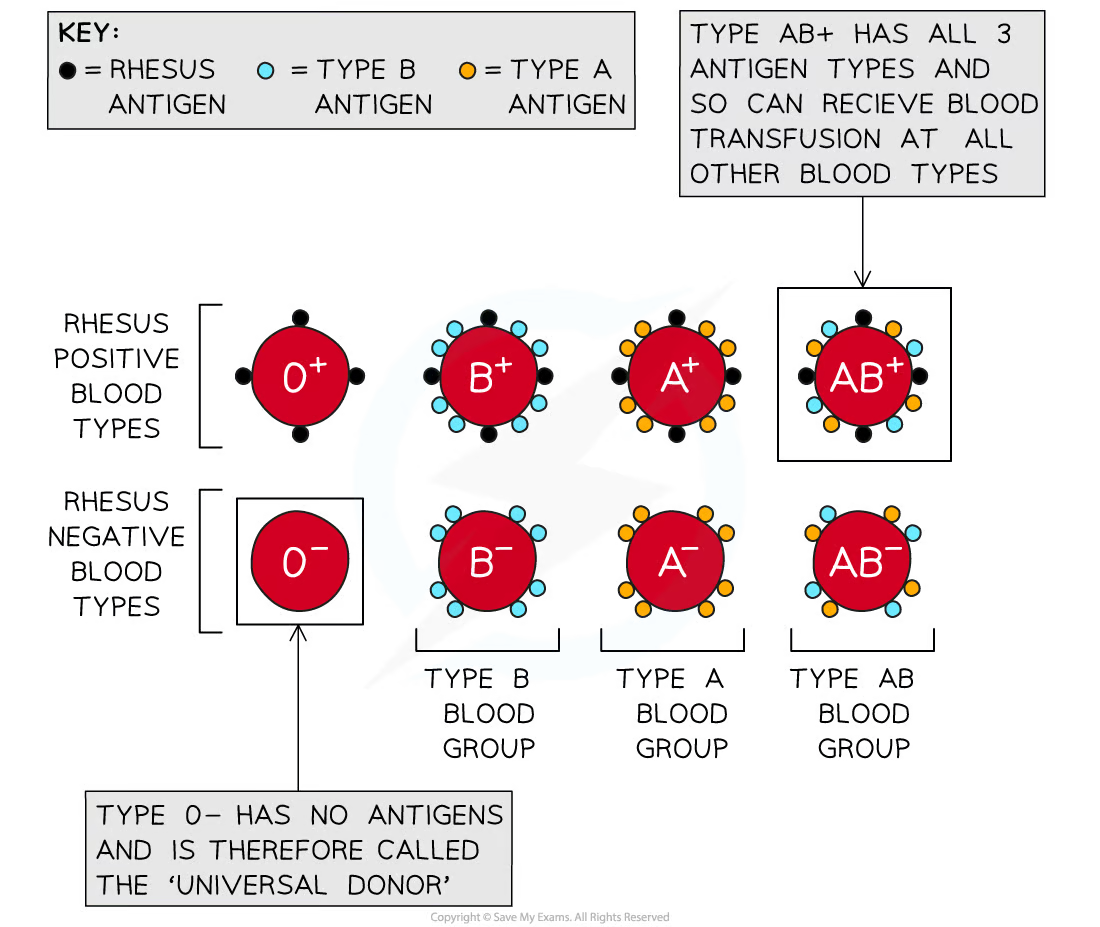
ABO blood group system
blood type is determined by the antigens attached to the end of the base unit
type O has no antigens (its only the base unit) but has both A and B antibodies → makes it the universal donor (there are no antibodies for type O)
60% of all blood transfusions are type O
type AB has both A and B antigens and has no antibodies → makes it the universal receiver (can get blood from any type)
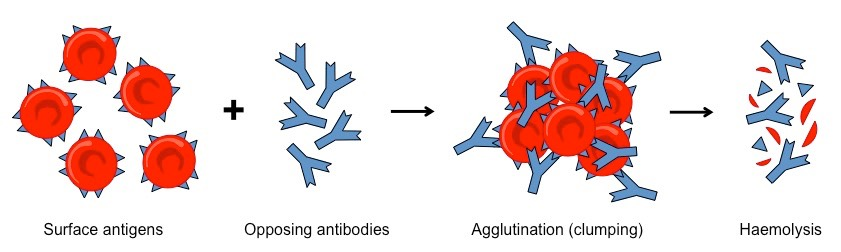
What happens if you get the wring type of blood?
the antibodies attack bind to the surface antigens causing Agglutination and Haemolysis (destruction of the blood cells)
Rh blood group system
where we get + or - in our blood types
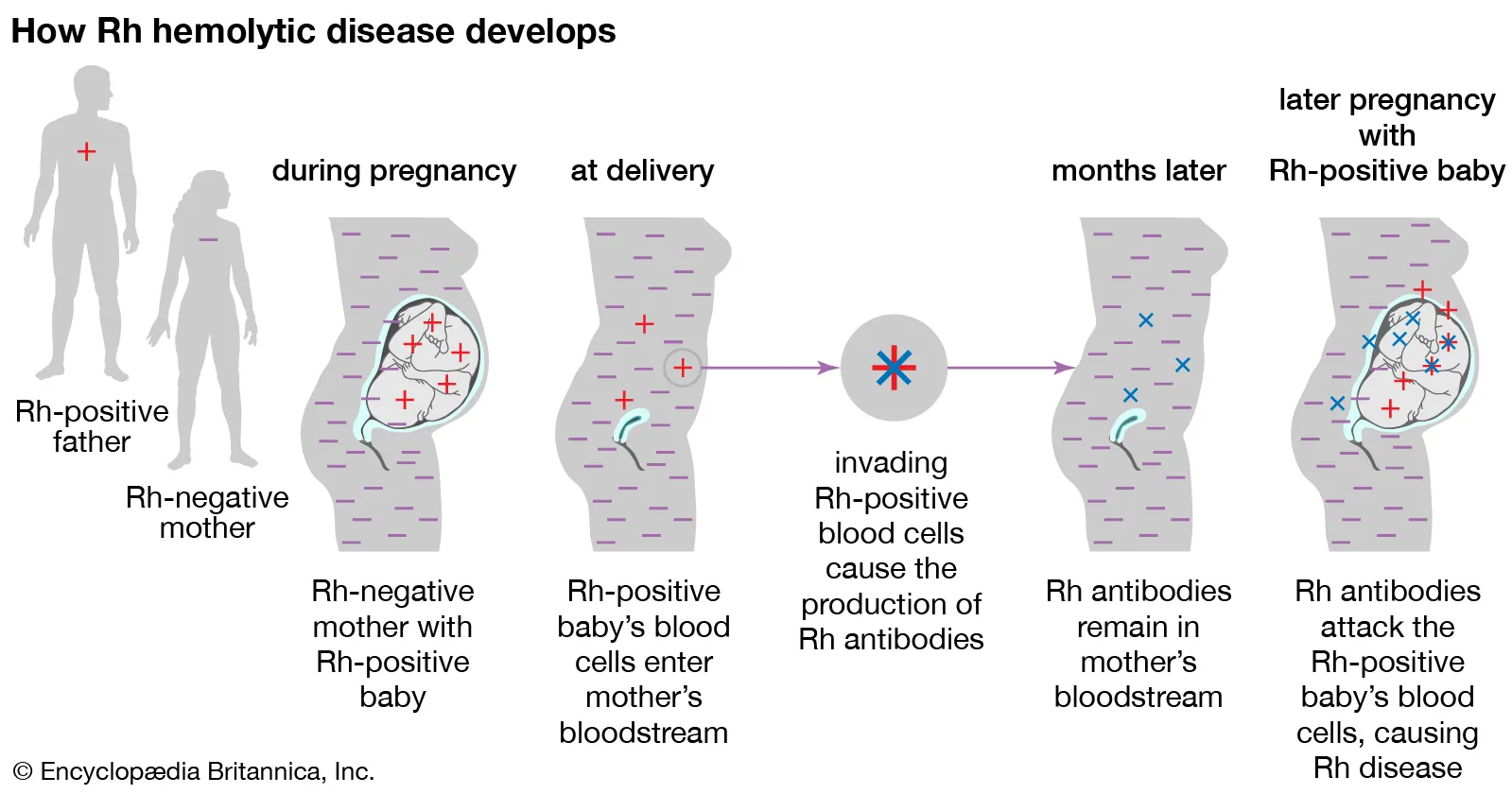
Rh hemolytic disease
occurs in mothers who are Rh- that have Rh+ after first pregnancy
treatment: at 28 weeks and within 72h of delivery they are given doses of RhoGAM (Rh+ antibody) that will bind to them when the blood of the foetus mixes with the mother during the hemorrhaging occurring during delivery preventing her from getting sensitized
her immune cells are unable to detect the Rh+ antigens thus preventing the disease from occurring in the following pregnancy
ABO types + Rh blood
Blood typing: ABO and Rh blood group systems
+ types can donate only to other + types
- types can donate to both -/+ types
true universal donor → O-
true universal recipient → A-
Leukocytes (WBC)
unlike RBC they do have nuclei and organelles
function → immunity, removal of waste, toxins and damage cells
primary site of action → lose connective tissue (CT)
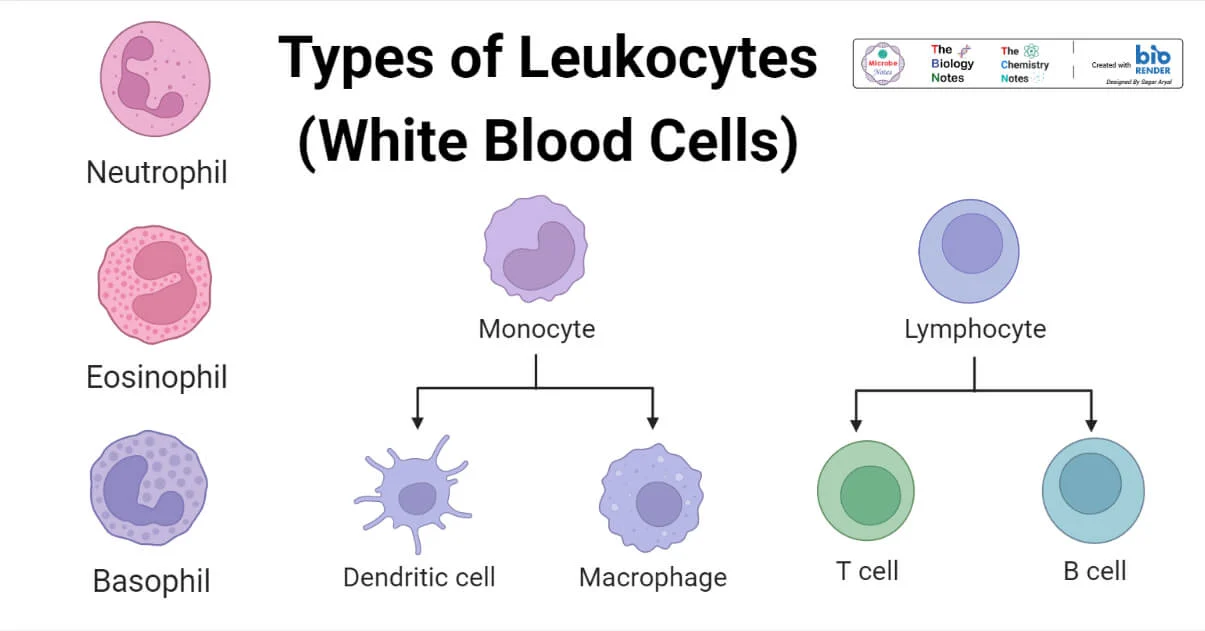
Types of WBCs
Granulocytes
Nucleophils (50%)
most abundant type of WBC
soecualized in attacking and digestin “marked” backteria
phagocytosis → lysosomes → respiratory burst (H2O2 and O2)
Eosinophils (2-4%)
attack and phagocytose objects coated with antibodies
will be in high ammounts during allergic or anaphylactic reactions
phagocytosis of pollen granules that have antibodies bound to them
also common in parasitic worm infections
exocytose cytotoxic compounds
In parasitic worm infections: eben though they are way smaller than the multicellular parasitic worms they exocytose chemicals that damage them
Basophils (<1%)
least numerous WBC
histamine (inflammation) and heparin (anticoagulant) granules
histamine makes blood vessel walls leaky (increase gaps btw epithelial cells) allowing blood to crowl
heparin prevents blood clots in blood vessesl
analogous to mast cells (resident cells in lose CT)
mast sells are in the connective tissue, basophils are in the blood
Agranulocytes
Monocytes (2-8%)
largest WBC
enter CT to become macrophages (syncytium)
phagocytosis
Lymphocytes (25%)
smallest WBC
abundant in CT and lymphoid tissue
3 functional classes →
T-cells → cell-mediated immunity
physically bind to cells to destroy them
B-cells → antibody- mediated immunity
secrete antibodies into the blood
NK cells → immune surveillance
Natural Killers, don’t need sensitizing
cruise around the body making sure you got normal sugars and stuff
sometimes they go haywire leading to autoimmune diseases
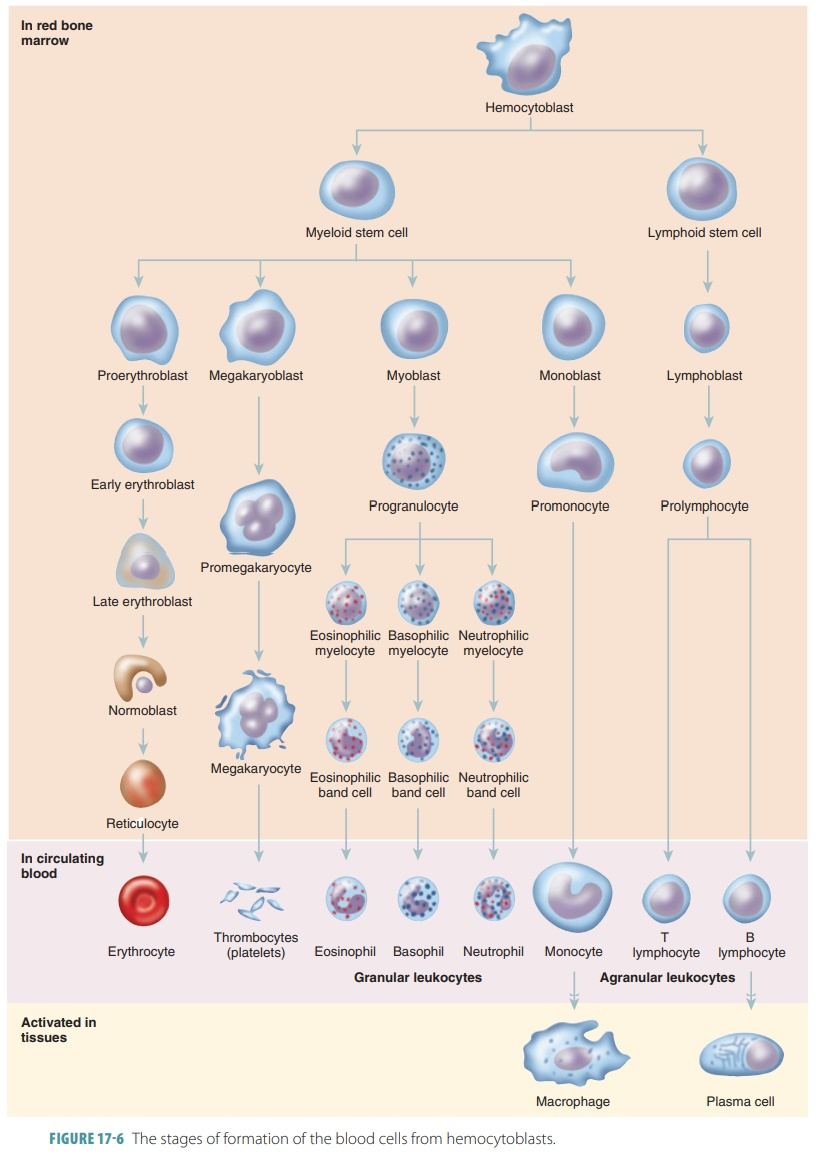
WBC production
occurs in red bone marrow (just like RBC)
CBC and differential WBC counts
CBC count (#WBC, #RBCs, #platetes, hemoglobin, hematocrit, etc.)
differential WBC count provides relative numbers of WBCs (#/100)
what does count indicate?
↓ WBC → leukopenia
↑ WBC → leukocytosis
could be leukemia (WBC cancer)