3. X-Ray Imaging
How it Works
- X-ray particles are called photons
- X-ray photons are delivered in packets called quanta.
- If the particle energy is greater than the binding energy of the electron, then the photons \n are capable of ionizing atoms.
- Diagnostic radiation is typically in the range of 100 nm to about 0.01 nm, or from 12 eV to 125 \n keV.
Components
- The number of X-ray photons produced depends on the number of electrons striking the target material (so tube current)
- The anode is made of either tungsten or molybdenum. The cathode is composed of two parts: the filament made of tungsten, and a focusing cup.
- A change in filament current changes the intensity of the X-ray photons.
- The X-ray beam coming off the cathode material is polychromatic.
- Filtering out the undesired portion of the X-ray spectrum can substantially reduce the radiation dose delivered to the patient.
Math
- c = λ * f
- c = 3E8 m/s
- 1 angstrom = 10E-10 m
- High frequency range is from 3E16 to 3E19 Hz
- E = h * f
- h = Plank’s constant = 6.63E-34 J*s = 4.13E-18 keV*s
- f is frequency, or ν (Greek letter nu)
- eV is an electron volt, a unit of energy representing the amount of energy one electron can obtain from accelerating between the potential difference of 1 volt
- 1 V = 1.602E-19 columbs = 1.602E-19 J
- A pjoton with 3E18 Hz frequency has what energy?
- 4.13E-18 keV*s x 3E-18 Hz = 12.39 keV
Ionization in X-Rays
- Simplest atom to ionize is an H atom (only 1 e-, super easy to ionize because we have a lot of H in our bodies)
- If it can ionize, it has enough energy to eject an electron
- 13.6 eV is enough to kick out an electron, and is the threshold of ionizing
X-Ray Generation
- X-rays are generated from an x-ray tube
- High potential difference between cathode and anode
- Acelerated electrons from a heated filament
- Electrons strike the target (sometimes tungsten
- Heat and x-rays are generated
- 99% of generated energy goes to heat
- Electrons interact with the target material mainly in 2 ways to generate radiation…. Braking and Characteristic
Braking “Bremsstrahlung” Radiation
- Electrons are slowed down (lose E)
- Change on energy is emitted as photon energy
- Generally, more photons in lower energy
- Max energy is related to max kV across tube
- E tube = kV * e
Characteristic Radiation
- Electron strikes another inner shell electron
- Inner electron is ejected with lower energy
- Electrons reconfigure to fill the void
- Photon is produced with specific photon energy
- Photon energy depends on the shell (closer to nucleus = more E)
X-Ray Spectrum
- Spectrum can also be characterized by its “effective energy” defined as the energy of a mono-energetic beam with the same penetrating ability
- Effective energy is a weighted sum of the spectrum
- Filtration whether intended or not, increases the effective energy of spectrum
- Number of photons is the “quantity” of the x-ray beam
- Energy level of the beam is the “quality”
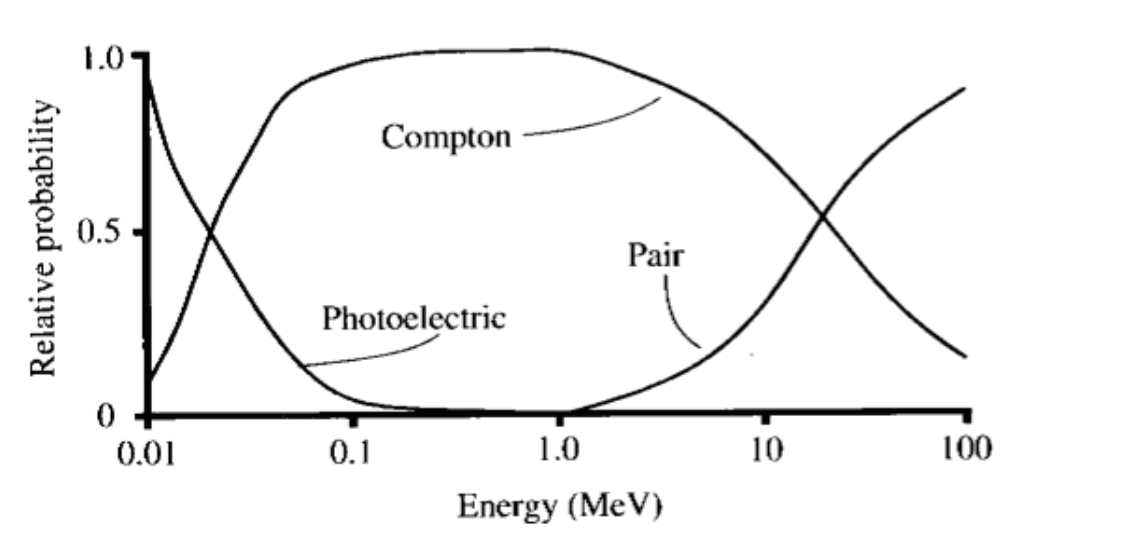
How Might X-Rays Interact with Matter?
Coherent (Rayleigh) scattering
- Photon bounces off in a new direction with little energy change
- The electric field of the incident photon’s EM wave expends energy by making all of the electrons in the atom to oscillate in phase
- Atom’s electron cloud then radiates the energy as a scattered photon
- Coherent scattering is used mostly with low energy diagnostic x-rays (mammography, thyroid scans)
- Electrons are not ejected so ionization does not occur
Compton scattering
- If it’s above 30keV with soft tissue, it’s probably compton scattering
- Steps
- Photon interacts with an electron (usually valence) and only some energy from the photon goes to the electron
- Photon moves on with reduced energy and new direction
- Electron is ejected
- Energy of the initial photon must be equal to the energy of the scattered photon + energy of ejected electron
- More dense the tissue = more likely Compton scattering occurs
- Compton scattering makes up most of the background noise & tissue damage
- If the initial energy is low, then the scattered energy doesn’t matter on the scattering angle
- If the initial energy is high, the scattered energy is higher for a smaller scattering angle
- Scattered photons with higher energies will continue in pretty much the same direction
- Compton scattering in which a photon is not absorbed but rather scattered. The photon energy is reduced, and an electron is ejected. This is the major source of noise in X-ray (and CT) images.
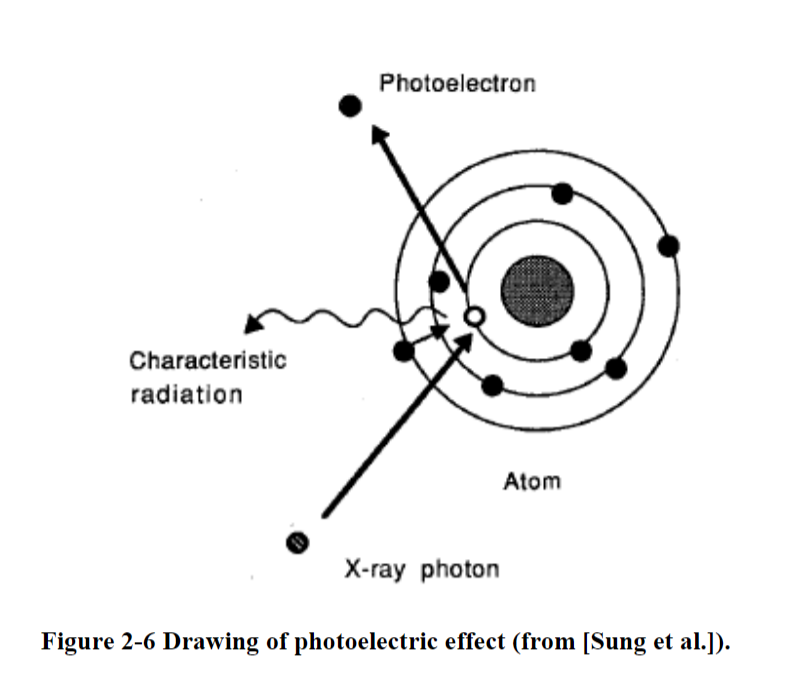
Photoelectric effect
- In the photoelectric effect, all of the initial energy is transferred to an electron
- Photoelectric effect in which a photon is absorbed, characteristic radiation is emitted along with photoelectrons, and possibly Auger electrons.
- Steps
- All photon energy transfers to electron
- Electron ejects
- Electron becomes a photoelectron
- Energy of the photoelectron is the energy of the initial photon minus the energy it took to bind to the orbital electron
- Called an Auger electron
- A lower orbital electron will jump up to take its place
- Energy needs to decrease now, so energy is given off as fluorescent energy
- Probability of characteristic x-ray emission (dangerous) decreases as the atomic number of the absorber atom decreases (less protons = less possibility of radiation)
- Soft tissue has lower atomic number so it’s not super frequent
- Probability of characteristic x-ray emission also decreases with increasing photon energy
Pair Production
- Pair production can occur when the energy of the incident photon exceeds 1.02 MeV
- Steps
- High energy photons are absorbed by a nucleus
- A positron (positive electron, a form of anti-matter) is emitted with an electron
- Energy above 1.02 MeV goes to the electron as kinetic energy
- The positron and electron interact and shoots oppositely directed 511 keV annihilation photons
- Unusual because it takes so much energy
- Describes the same anti-matter formation used in PET scans
- Pair production in which a photon is absorbed by the nucleus, a positron is emitted, and an electron is ejected.
Photo-disintegration
- Interaction of an incident photon with a nucleus, which produces one (or more) ejected nuclear particle
- One element becomes a different element
- Super unusual so it takes so much energy