Modern Atomic Theory Review
- ^^light is a form of electromagnetic radiation^^
* properties of both waves and particles:
* wavelength (λ) - the distance between adjacent wave crests, meters
* red light (750 NM) has longest wave length
* violet light (400 NM) has shortest wave length
* ==1 NM = 1 * 10^-9 meters==
* frequency (v) - number of cycles or crests that pass through a stationary point in one second
* amplitude - the height of the wave from zero to crest
* ^^wavelength and frequency are inversely/indirectly related^^ - the shorter the wavelength, the higher the frequency
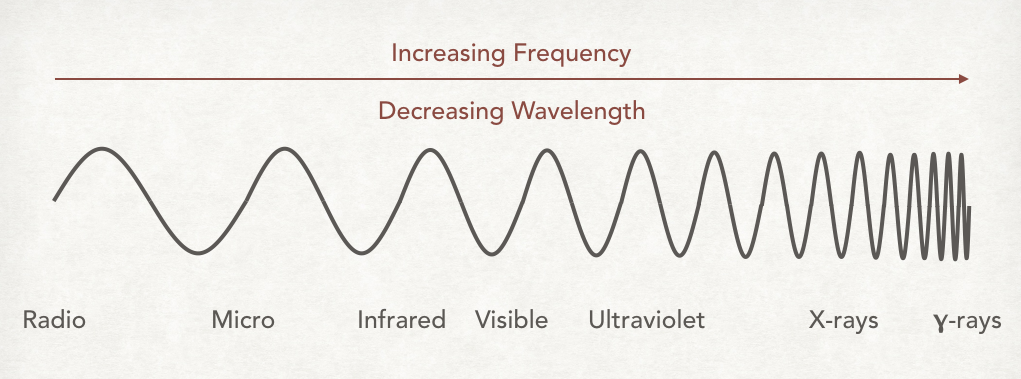
* ==speed of light====: 2.998 * 10^8 meters/second = λv==
- electromagnetic radiation
* light can be viewed as a stream of particles
* particle of light is a ^^photon^^
* photon - a single packet of light energy
* has specific wavelength, determines what light we see
* wavelengths of spectral lines are characteristics of the element
* make up atomic emission spectra
* no two elements have the same emission spectra
* amount of energy carried in the packet depends on the wavelength of the light - ^^the shorter the wavelength, the greater the energy^^
* light waves that carry more energy in their crests are closer
* violet light carries more energy per photon than red light
* the photoelectric effect - the emission of electrons from a metal when light shines on the metal
* ^^quantum of energy^^ - the minimum quantum of energy that can be lost or gained by an atom
* ^^quantized^^: an electron has to absorb/emit a specific amount of energy to move from one energy level to another
* ^^ground state^^: the normal energy level any given electron occupies
* ^^excited state^^: the energy level an electron occupies when it has absorbed the specific quantum of energy to move up to that level
* %%Planck’s Law%% - ==E=Hv==
* E - energy, joules
* H - ==Planck’s constant, 6.626 * 10^-34 J*S==
* v - frequency - %%Bohr’s Model%%
* Niels Bohr changed Rutherford’s model to include newer discoveries about how the energy of an atom changes when the atom absorbs/emits energy
* proposed electron is found only in specific circular paths/orbits around the nucleus ❌
* incorrect - if the orbits were truly circular, the electron would spiral into the nucleus
* each possible electron orbit has a fixed energy - ^^energy level^^ ✅
* each orbit is a specific distance from the nucleus and at each specific energy
* ^^impossible for an electron to exist between orbits^^
* ^^amount of energy is directly related to the frequency → wavelength^^ - %%de Broglie%%: proposed “electrons be considered as waves confined to the space around an atomic nucleus”
- %%Heisenberg Uncertainty Principle%%
* Werner Heisenberg
* states that it is impossible to determine simultaneously both the position and velocity of an electron
* “we cannot know both the position and speed of a particle, such as a photon or electron, with perfect accuracy”
* %%Schrödinger Wave Equation%%
* Erwin Schrödinger developed an equation that treated electrons as waves
* ^^Quantum Theory^^ - describes mathematically the wave properties of electrons
* electrons exist in certain regions called orbitals
* orbitals - 3D regions around the nucleus that indicate the probable location of an electron
* represent probability maps showing a statistical attribution of where the electron is likely to be found
* 4 Wave Properties
* ^^Energy Level^^: Principal Quantum Numbers - number specifying the principle shell of orbital
* n - indicates the energy level
* energy increases with principal quantum number
* maximum of 7 energy levels
* ==n^2== - how many orbitals in any energy level
* ==2n^2== - maxim. number of electrons possible in any energy level
* ^^Sub Level^^: Shapes of Quantum Mechanical Orbitals
* letter indicates subshell of orbital, specifies shape
* possible letters - s, p, d, f
* electrons are more likely to be found closer to the nucleus than farther away
* ^^Orbital^^: Orientation
* s - 1 orbital
* p - 3 orbitals
* d - 5 orbitals
* f - 7 orbitals
* ^^Spin^^: clockwise or counterclockwise
| ENERGY LEVEL | SUB-LEVEL | # ORBITALS (n^2) | ELECTRONS (2n^2) |
|---|---|---|---|
| n=1 | 1s | 1 | 2 |
| n=2 | 2s 2p | 4 | 8 |
| n=3 | 3s 3p 3d | 9 | 18 |
| n=4 | 4s 4p 4d 4f | 16 | 32 |
- Electron Configuration
* arrangement of electrons in an atom and the way in which the electrons are arranged in various orbitals around the nucleus
* %%Aufbau Principle%%
* the electrons will fill the orbitals in a very specific order
* lowest → highest energy
* ^^The Diagonal Rule^^

* %%Pauli Exclusion Principle%%
* an individual orbital may describe at most TWO electrons
* in order to occupy the orbital, the two electrons must have opposite spins: ⬆⬇
* EXAMPLES
* Carbon 6e- : 1s^2, 2s^2, 2p^2
* Aluminum 13e- : 1s^2, 2s^2, 2p^6, 3s^2, 3p^1
* Noble Gas Configuration
* Aluminum 13e- : [Ne] 3s^2, 3p^1
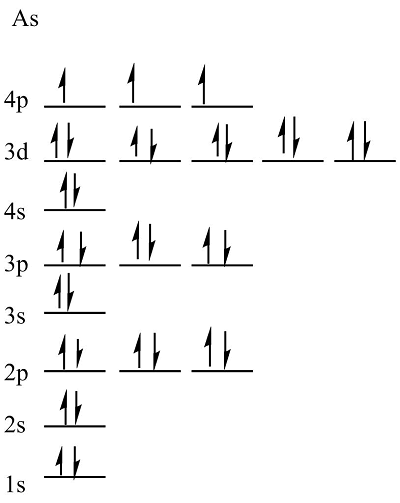
- %%Hund’s Rule%%
* orbitals in the same sub-level must all fill with one electron before a second electron is added to any of the orbitals: (n)p^4 - ⬆⬇ ⬆ ⬆
* the “single” electrons will all have the same spin direction
* Orbital Diagram
- @@Valence@@ and Electrons
* valence electrons are the electrons on the outermost energy level
* the noble gases always have full valence shells
* Selenium 34e- : @@4s^2@@, @@4p^4@@
* Silicon 14e- : @@3s^2, 3p^2@@
* the chemical properties of elements are largely determined by the number of valence electrons they contain
* properties vary in a periodic fashion because the number of valence electrons is periodic - Atomic Physical Properties
* Atomic Size ⬇⬅
* left-right ^^decreases ⬅^^
* across a period, the amount of protons in the nucleus increases which has a stronger pull on the electrons, causing them to move closer to the nucleus
* top-bottom ^^increases ⬇^^
* size of the orbital increases with increasing principal quantum shell number
* electrons occupying the outermost orbitals are farthest from nucleus
* Ionization Energy ⬆➡
* amount of energy needed to remove a single electron
* left-right ^^increases ➡^^
* electrical pull on electrons from the # protons in nucleus causes increases amount of energy needed
* top-bottom ^^decreases ⬆^^
* electrons in outermost orbitals are less affected by the electrical pull from nucleus
* Electronegativity ⬆➡
* ability of an atom of an element to attract electrons when the atom is in a compound
* left-right ^^increases ➡^^
* top-bottom ^^decreases ⬆^^