8.2 Strong and Weak Acids and Bases
\
Acids:
- Strong acids/bases: ionize almost to 100% in water, producing hydrogen ions (acids) or hydroxide ions (bases).
- Weak acids/bases: only partly ionize in water.
\
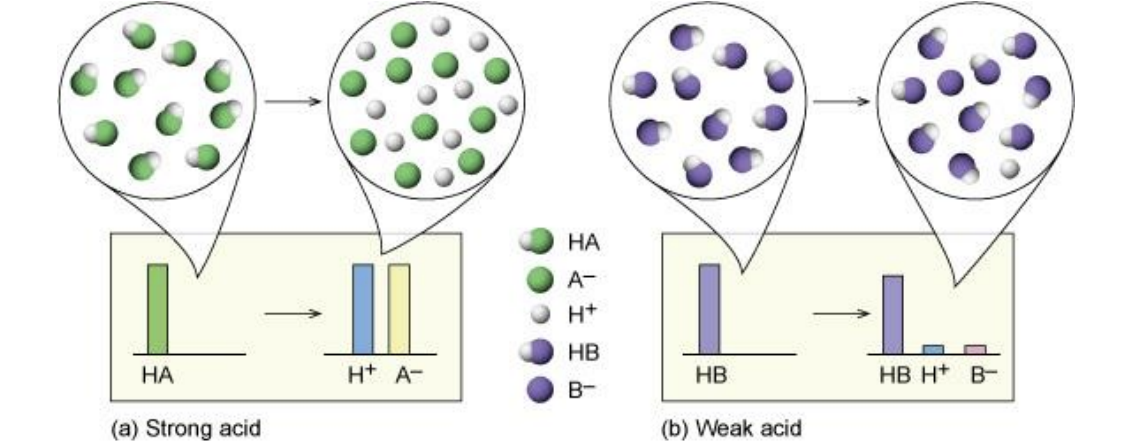
- Strong acids have a very large Ka, equilibrium position is far to the right, [HA(aq)]initial [H (aq)]equilibrium
- Weak acids have a small Ka, equilibrium position is far to the left, [HA(aq)]initial >> [H+ (aq)]equilibrium
- List of Ka values on pg. 496 Table 2 or pg. 726 appendix B5
\
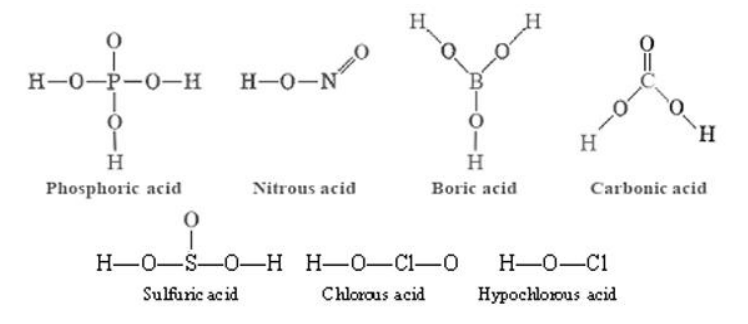
- Oxyacids: an acid in which the acidic hydrogen atom is attached to an oxygen atom:
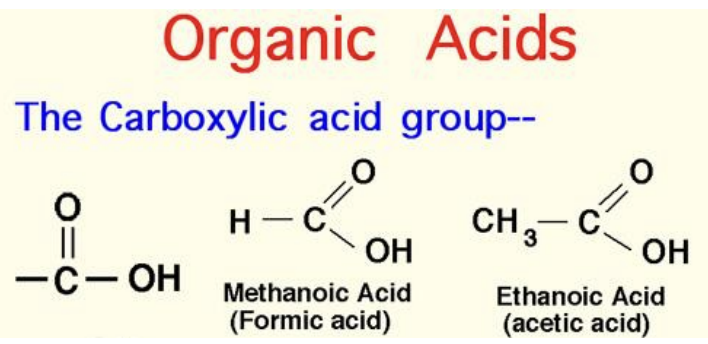
- Organic acids: an acids containing a carboxyl group (COOH)
Bases
- Strong Bases – hydroxides formed with metals from group I and II on the periodic table. Group I bases have a high solubility, and high dissociation. We can say that virtually all of the base dissociates to form the ions:
- BIG K
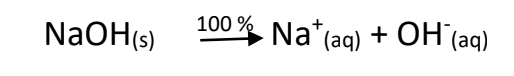
- Although group II hydroxides are strong bases, they have low solubility in water: * Not a lot dissolves, but what dissolves forms a lot of hydroxides. Interesting dichotimies.
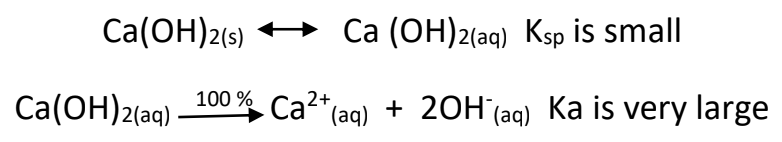
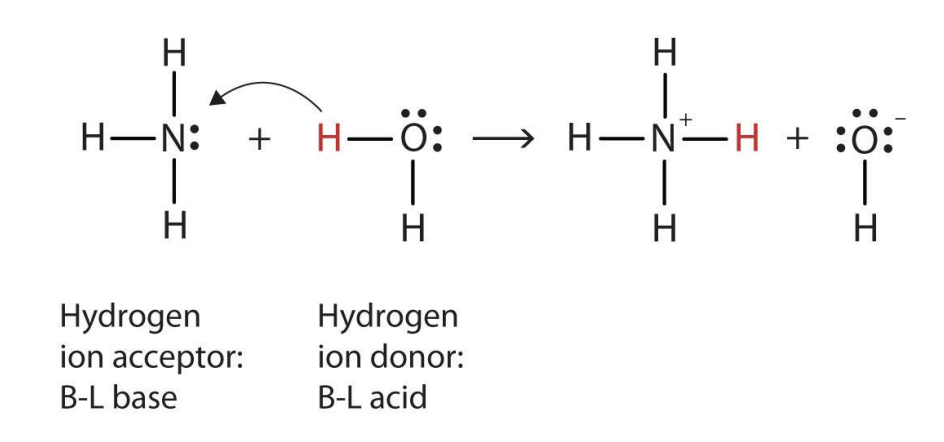
- Weak Bases – only partially react to produce hydroxide ions. A good example is ammonia: has one lone electron pair capable of forming coordinate bonds.
- In general, the reaction of a weak base in water is:
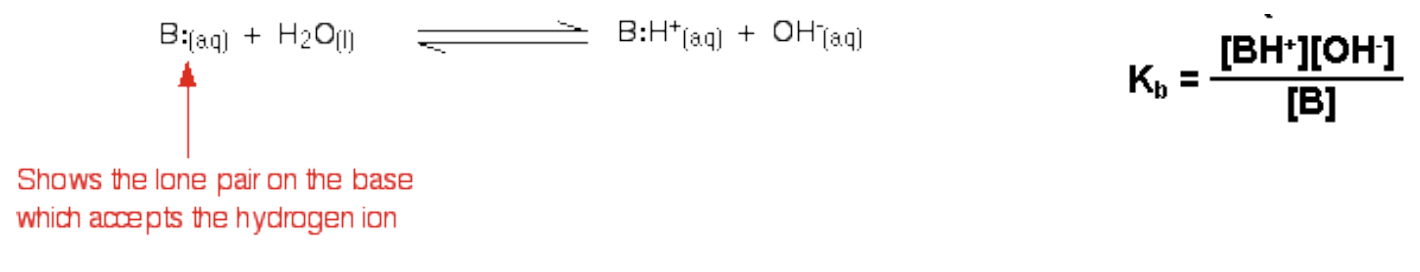
- Write the expression for Kb when ammonia reacts with water:

- Autoionization of water: the transfer of a hydrogen ion from one water molecule to another: * This is called the ion-product constant for water (Kw)
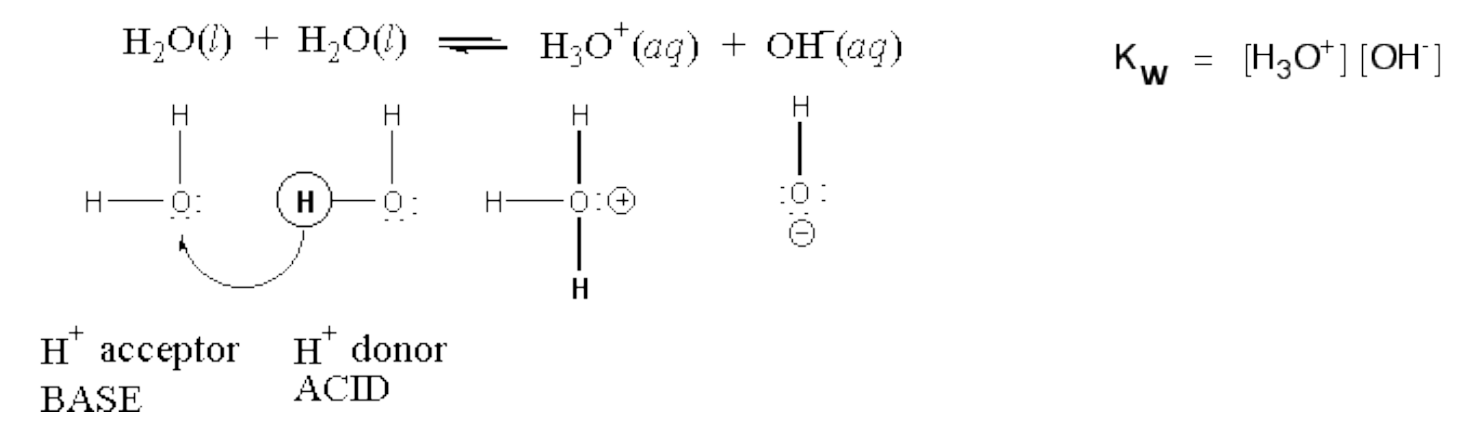
- At 25 oC, [H+ (aq)] is 1.0 x 10-7 mol/L and [OH- (aq)] is 1.0 x 10-7 mol/L. Calculate Kw: * Kw = )concentration of hydronium)(concentration of hydroxide) * 1.0 x 10^-14 * Always the equilibrium constant for the autoionization of water
\
- Three situations: * Kw = KaKb

- Overall, as the strength of the acid increases, strength of the conjugate base decreases, and vice versa:
1. A strong acid/base will have a very weak conjugate (only forward rx) 2. A weak acid/base will have a weak conjugate (equilibrium) 3. A very weak acid/base will have a strong conjugate (no reaction)
If you need concentrations:
