16 Sulfur
Sulfur sources and uses
- Sulfur is found in its elemental state underground in the USA, Mexico and Poland
- It is also a by-product from the removal of sulfur from petroleum and natural gas
- Sulfur can also be obtained from sulfide ores
- The main use of sulfur is in making sulphuric acid which is a very important chemical used in many industries
- It is also used extensively in making rubber tyres more flexible (vulcanising), where the rubber is heated with sulfur
Sulfur dioxide
- Sulfur dioxide can be made by burning sulphur in air
- This is the method used in the first stage of the manufacture of sulfuric acid
- Sulfur dioxide is used as a bleach in the manufacture of wood pulp for paper, and as a preservative for foods and drinks by killing bacteria
- Sulfites are often added to foods and these release sulfur dioxide in acidic conditions


\
Sulfuric Acid : Properties, Manufacture and uses

\ \
- Sulfuric acid is synthesised by the Contact process which use sulfur and oxygen from air and is done in three distinct stages
The Contact Process
Stage 1
- The first stage is the oxidation of sulfur: * S + O2 → SO2
Stage 2
- The main stage is the oxidation of sulfur dioxide to sulfur trioxide using a V2O5 catalyst:

\
- The conditions for the main stage of production of sulfur trioxide need to be considered
Conditions during Stage 2
Temperature 450°
- The reaction is exothermic, so increasing the temperature shifts the position of equilibrium to the left in the direction of the reactants
- Therefore the higher the temperature, the lower the yield of sulfur trioxide
- The optimum temperature is a compromise between a higher rate of reaction at a higher temperature and a lower equilibrium yield at a higher temperature
Pressure: 2 atm
- An increase in pressure shifts the position of equilibrium to the right in the direction of a smaller number of gaseous molecules
- However the position of equilibrium lies far to the right (the equilibrium mixture contains about 96% sulfur trioxide)
- So the reaction is carried out at just above atmospheric pressure because: * it is not worth spending the extra energy or money required to produce high pressures * a higher pressure causes the sulfur dioxide to liquefy
Stage 3
- Once stage 2 is completed, the sulfur trioxide is absorbed into a solution of 98% sulphuric acid to produce a thick liquid called oleum: * SO3 + H2SO4 → H2SO7
- It is not absorbed into water because a fine mist of sulfuric acid would be produced and this would be difficult to condense and is also highly dangerous
- Oleum is added to water to form concentrated sulfuric acid:
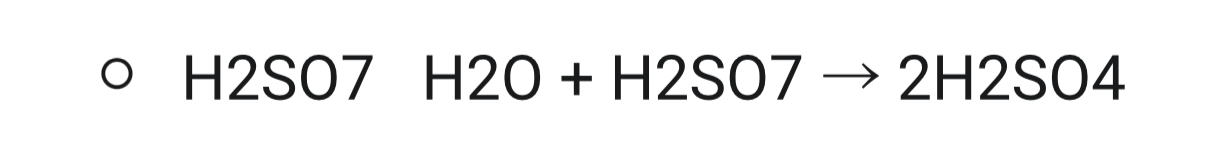
Properties of sulfuric acid
- Sulfuric acid is a strong dibasic acid as two of its hydrogen atoms can be replaced by a metal * Mg + H2SO4 → MgSO4 + H2
- It reacts in a similar way to other acids with metal carbonates, oxides, hydroxides (and ammonia) and metals, e.g: * Na2CO3 + H2SO4 → Na2SO4 + CO2+ H2O * ZnO + H2SO4 → ZnSO4 + H2O
\
- Concentrated sulphuric acid is corrosive and a powerful oxidising agent
- Concentrated sulphuric acid is also a very powerful dehydrating agent and is very good at removing water from other substances
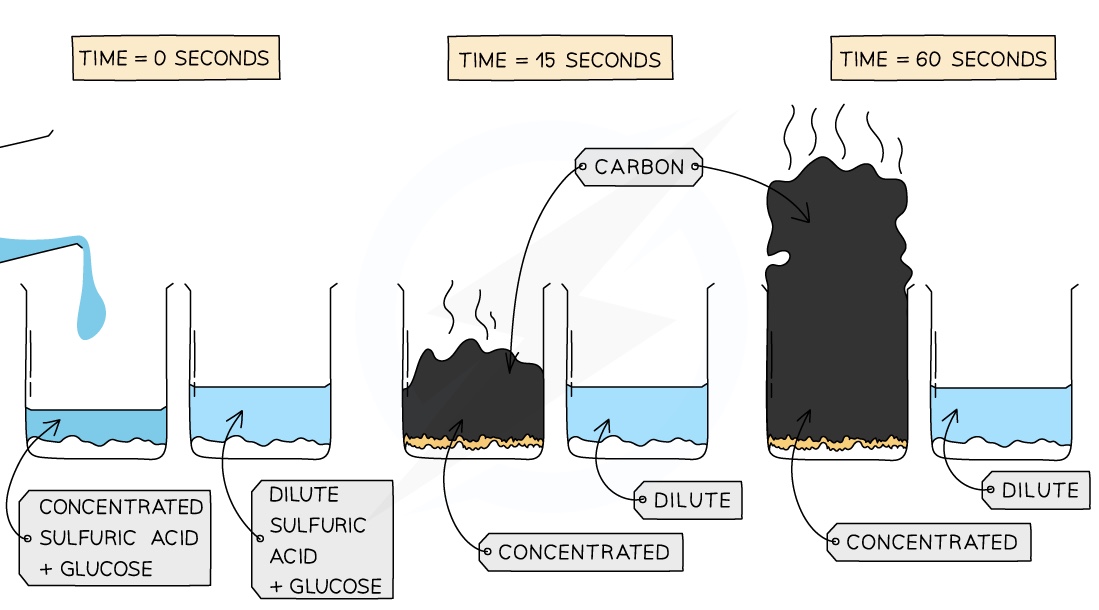
- For example, if mixed with sugar (C6H12O6), concentrated H2SO4 will remove water molecules and leave behind carbon in a spectacular looking black tower
\
Uses of Sulfuric Acid
In dilute solution it is used as a catalyst in many organic reactions and to clean the surface of metals
- Concentrated sulfuric acid is used in car batteries, making fertilisers, soaps and detergents
- It is also used to make acid drain cleaners and in the production of paints and dyes

\ \n \n \n \n
\