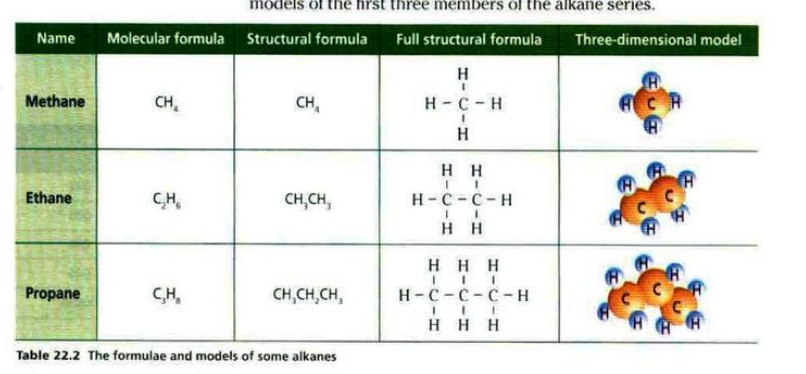
Chapter 22- Alkanes
- Alkanes are hydrocarbons that contain only single covalent bonds between carbon atoms.
- They have the general formula: C
nH2n+2 - A structural formula is the formula which shows how atoms are arranged in a molecule.
- Alkanes are saturated hydrocarbons because they contain only single carbon-carbon bond.
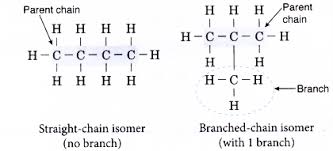
ISOMERISM
- Compounds that have the same molecular formula but different structural formula are isomers.
- Branched alkyl groups are isomers of alkanes.
PROPERTIES OF ALKANES
- As alkane molecules get bigger, the attractive forces become stronger.
- Melting and boiling points, and density of alkanes increases as their molecular sizes increases.
- Viscosity also increases as it becomes difficult to pour out bigger molecule chains.
- The flammability decreases as the carbon chains become longer.
- Complete combustion of alkanes produces carbon dioxide and water while incomplete combustion produces carbon monoxide and water.
CRACKING
- Longer chains of alkanes are broken down to smaller chains of hydrocarbons which are more in demand. This process is cracking.
SUBSTITUTION REACTIONS
- Alkanes are generally unreactive but they react with halogens.
- One mole of chlorine substitutes one hydrogen atom. Another product of hydrogen chloride is formed.
