1.1C Energy and Changes of State
Temperature Change vs Energy Input when heating a solid at constant pressure
Energy added at a change of state goes into the breaking of inter-particle forces, not into raising the temperature
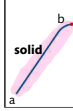
a-b: solid is heated, vibrational energy of particles increases so the temperature increases
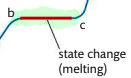
b-c: melting point, vibrations are energetic enough for particles to move away from fixed positions and form a liquid. Energy added breaks inter-particle forces, but does not raise kinetic energy so temperature remains constant
c-d: liquid is heated, particles gain kinetic energy and temperature increases

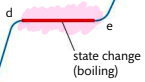
d-e: boiling point, sufficient energy to break all inter-particle forces and form a gas.
Requires more energy than melting
Temperature remains constant
Bubbles of gas visible throughout volume of liquid
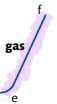
e-f: gas is heated under pressure, kinetic energy of particles continues to rise, temperature also rises
A note about units of temperature
In work this year, it is essential to use values for temperature recorded in kelvin (K), not in Celsius (°C). Temperature in kelvin is known as absolute temperature, where absolute zero (0 K) is the point of zero kinetic energy of particles. This coincides with -273.15 °C. To convert between the two, simply add or subtract 273.15 (or just 273).
Temperature (K) = Temperature (°C) + 273.15