4.1 Types of Chemical Bonds
\
- Electrostatic forces that hold atoms together in compounds
- Involves the interaction of the valance electrons
<<Ionic<< <<Chemical Bonds<<
- electrons and transferred between oppositely charged ions
- form ionic compounds with ionic bonding
- ions form to become isoelectronic with a noble gas, same electron configuration * Isoelectronic: same electron configuration as another
<<Covalent Chemical Bonds<<
- electrons are shared
- forms molecular compounds
- Nonpolar, polar or pure
- Molecular element: pure substance made up of 2 atoms like Oxygen
- As two atoms move close together, the electron cloud of one attracts the nucleus of the other
- At the same time the nuclei repel each other as do the electron cloud
- The atoms stay a distance from one another that has the lowest overall energy of the system like the hydrogen molecule.
- Inter: how the molecules are attracting with one another like London Dispersion, Hydrogen Bonding & Dipole-Dipole
- Intra: what is holding the molecule togehter like Covalent or Ionic
<<Lewis Theory of Bonding<<
- Atoms & ions are stable if they have a full valence shell of electrons
- Electrons are most stable when they are paired
- Atoms form chemical bonds to achieve a full valence shell of electrons
- A full valence shell of electrons may be achieved by an exchange of electrons between metal and non-metal atoms
- The sharing of electrons results in a covalent bond
<<Duet / Octet Rule<<
- Hydrogen is stable with 2 electrons
- Most atoms are stable with 8 electrons
<<Lewis Structures<<
- Draw the central atom (highest bonding capacity)
- Arrange the symbols of the atoms for the rest of the elements around equal distance apart
- Add up the number of valence electrons of each atom. Add to this any negative charge or subtract any positive charge.
- Place a pair of bonded electrons between central atom and each of the others (single bond).
- Place lone electron pairs on outer atoms first (follow duet and octet rule)
- Dump rest of electrons on central atom in pairs
- Move electrons around to form double or triple bonds until all atoms follow octet/duet rule.
- Resonance Structures: models that give the relative position of atoms in a Lewis Structure, but show different places for their bonding and lone pairs
Exceptions
- Under filled octets: molecules who’s central atoms are surrounded by fewer than 8 electrons * Ex: Boron Trifluoride
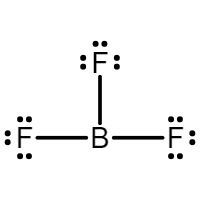
- Overfilled octets: molecules whose central atoms are surrounded by more than 8 electrons * Sulfur has its valence electrons in the third energy level * There is space for 12 electrons in the valence shells * Sulfur, phosphorus, chlorine
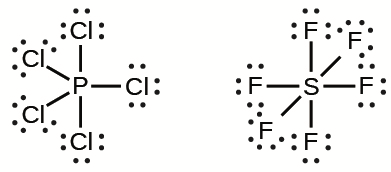
Determining Formal Charge
- Formal charge of a molecule to determine its most stable structure
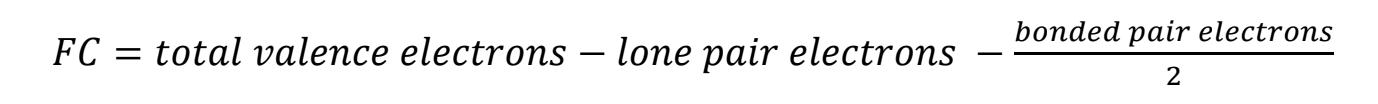
- Determine for each atom, then add all atoms together to determine charge of molecule
- The arrangement with which charges are the most stable
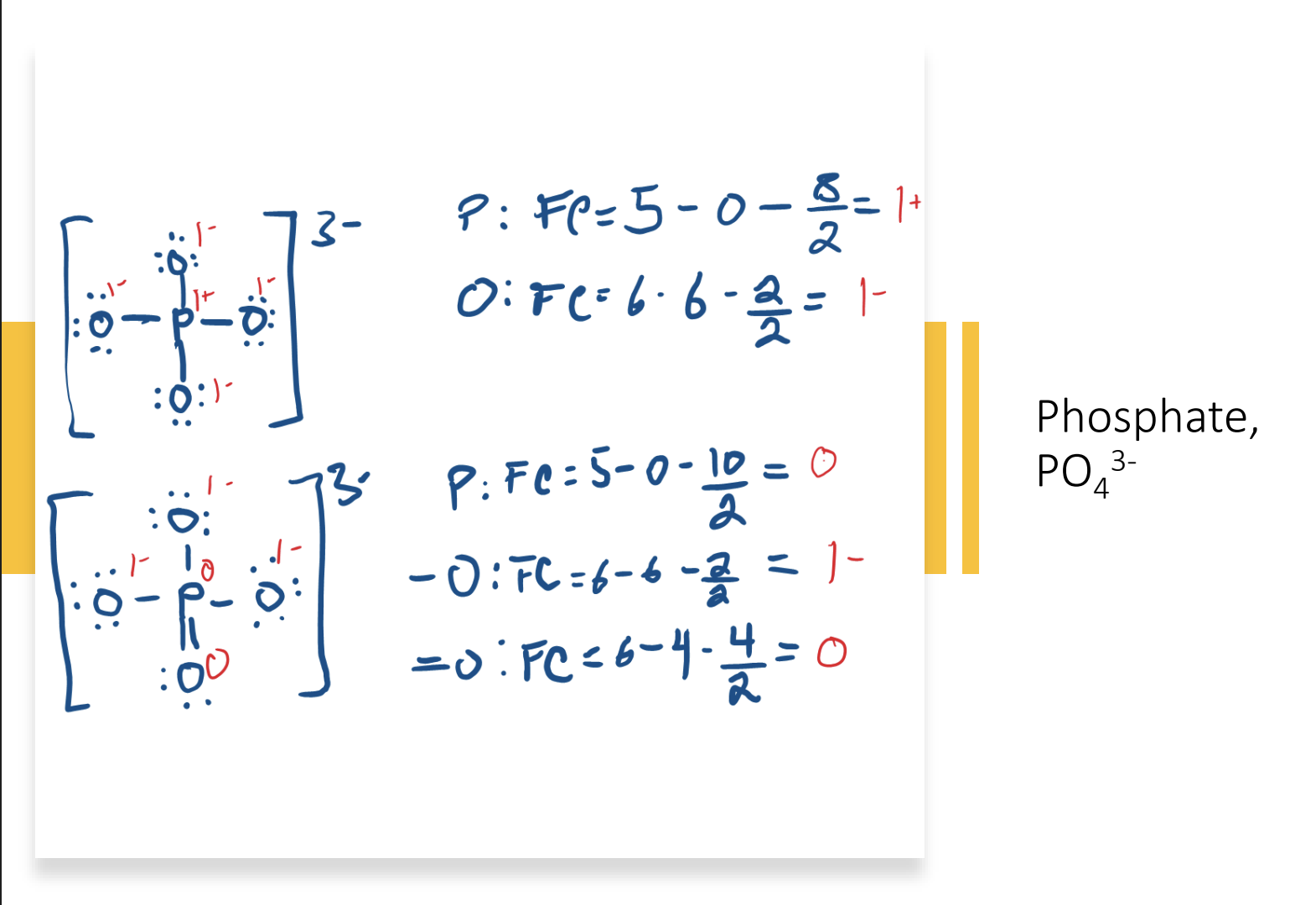
- The arrangement where P has an overfilled octet contains fewer charges than the arrangement where P has a normal octet
- Certain elements are able to do this because they have an empty 3d orbital that electrons can fill
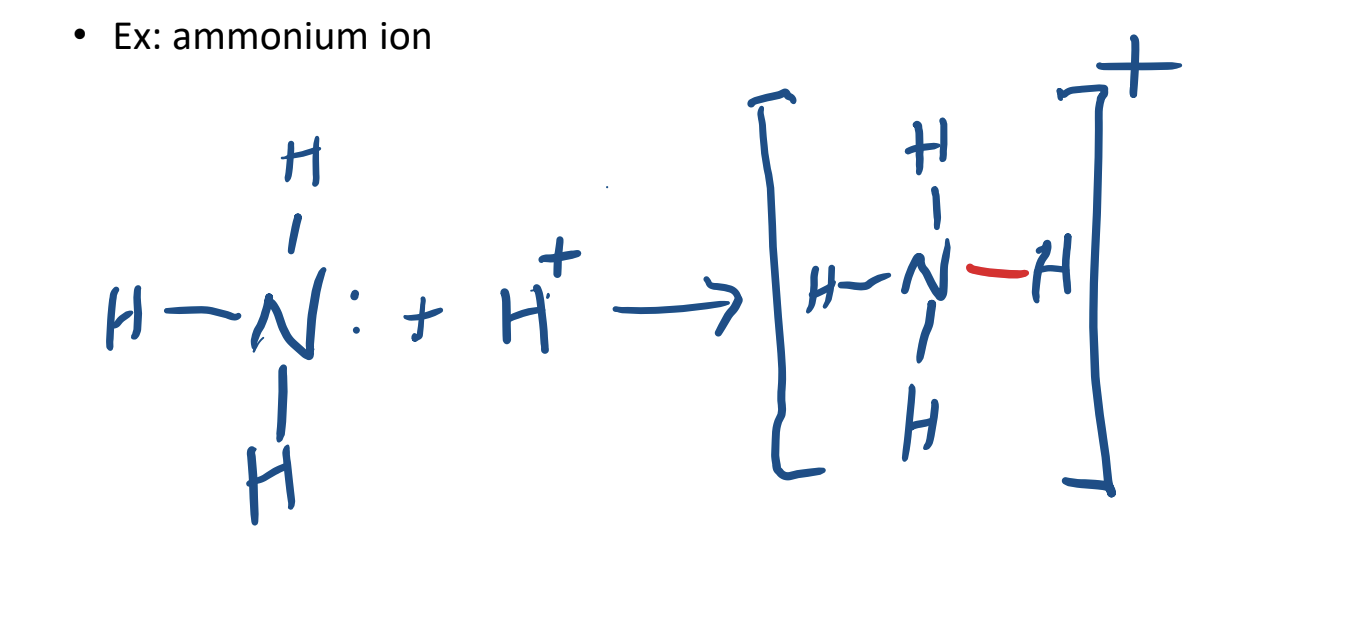
Coordinate Covalent Bonding
- a covalent bond in which the both of the bonding electrons are from one atom
\