Physical and Chemical Properties of Carboxylic Acids
Introduction to Carboxylic Acids
- Carboxylic acids contain the : a carbonyl with oxygen attached to the carbon that’s attached to a hydrogen
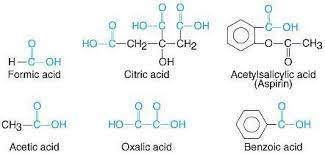
- Some common carboxylic acids are shown below:
Physical Properties of Carboxylic Acids
- They are the most polar functional group due to the carboxyl group
- They have the highest melting and boiling points of all the functional groups
- They tend to form dimers in the solid, liquid, and gaseous phases
- this makes their apparent molar mass higher than their actual molar mass
- carboxylic acids up to 6 carbons long tend to be water soluble
- benzoic acid (benzene ring attached to carboxyl group) is soluble in hot water
Chemical Properties of Carboxylic Acids
- Carboxylic acids are weak acids
- Most carboxylic acids have a pKa of approximately 5
- The dissociation reaction of acetic acid is shown below
- it will be similar for all carboxylic acids
- Acetic acid has a pKa of 4.74
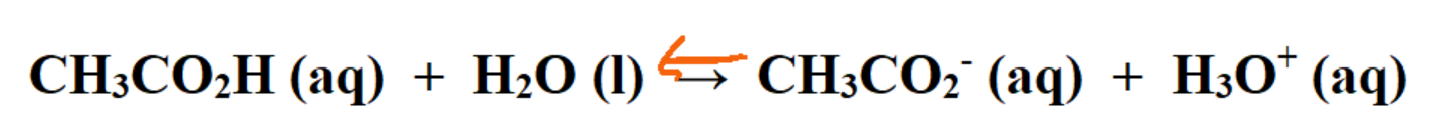
- carboxylic acids are more acidic than alcohols because of
- the resonance effect of the anion
- inductive effect of the carbonyl group
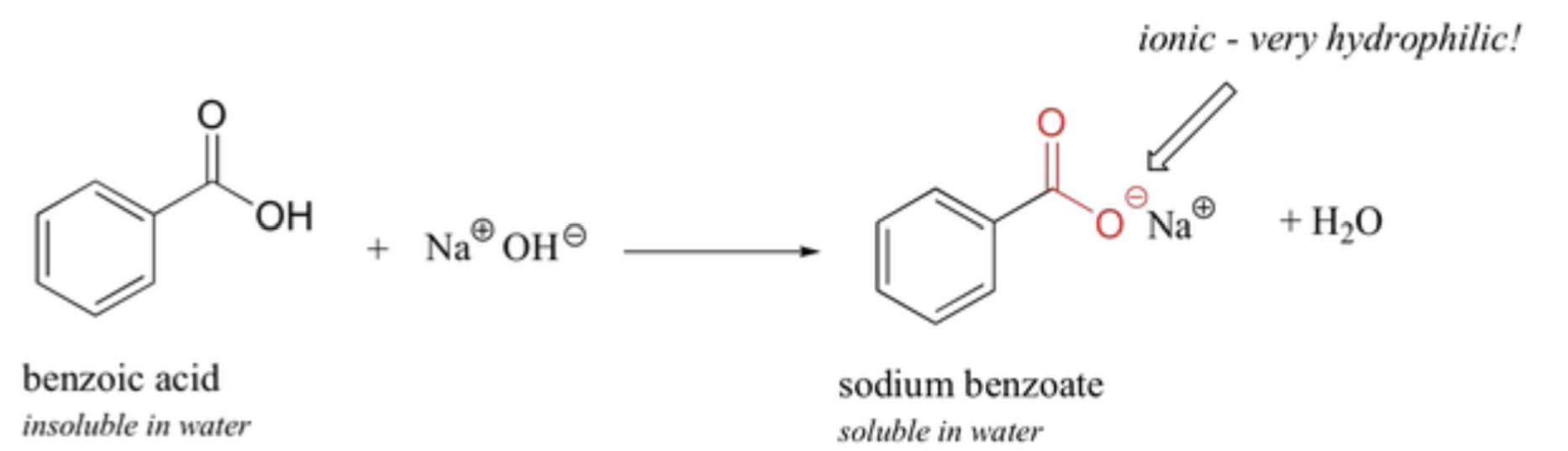
- They can react with sodium hydroxide and sodium bicarbonate to form (salts of carboxylic acids)
- The reaction of benzoic acid with sodium hydroxide is shown below