Unit 1: Bio Chemistry and Macromolecules
Slogan: Structure determines Function
Pre Unit 1: Statistics
Null hypothesis- States that there is no difference between groups or no relationship between variables
Positive Control Group- Experiment with definite results from the variable
used to confirm the system is working correctly
Negative Control Group- Experiment where no response to the variable occurs
used to check false positive
Independent Variable- The part of the experiment you change
Always on the x-axis
Dependent Variable- The part of the experiment that changes as a result of manipulation
Found on the y-axis
Standard Deviation- A measure of how each value in a data set varies of deviates from the mean
68% of data will be within 1 standard deviation
95% of data will be within 2 standard deviations
99% of data will be within 3 standard deviation
Formula: SD :
Standard error- A statistical measure of how far the sample average of the data is likely to be from the true population mean
Standard error bars- lines that represent the variability in the data
If they overlap, the data is NOT significant
Unit 1
Cohesion (Company)- the same molecules interact
Water + water
Adhesion (Alternate)- different molecules come together
Water + Penny
What property of water has to do with heat capacity?
Hydrogen bonding
Functional Groups
Group | Alt. Name | Structure | Characteristics |
Methyl | Alkyl | 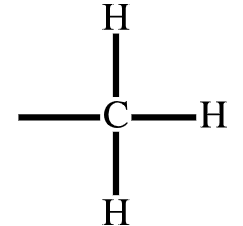 |
|
Amino | Amines | 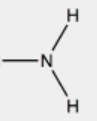 |
|
Phosphate | Organic Phosphate | 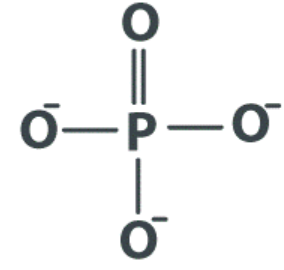 |
|
Hydroxyl | Alcohol |  |
|
Carbonyl | Aldehydes /ketones | 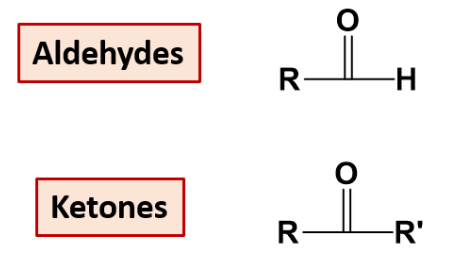 |
|
Sulfhydryl | thiols |  |
|
Carboxyl | Carboxylic Acids | 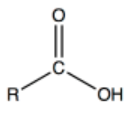 |
|
Macromolecules
CHONPS- Carbon, Hydrogen, Oxygen, Nitrogen, Phosphorus, Sulfur
Chemical reactions:
Anabolic: using energy to build larger molecules from smaller molecules
dehydration synthesis takes out water to combine two monomers
Catabolic: releases energy by breaking down larger molecules into smaller molecules
Hydrolysis breaks down monomers by “splitting“ water and “adding” it to the respective monomers
Metabolism: the sum of all the chemical reactions in your body
Carbohydrates
ONLY CONTAIN: Carbon, Hydrogen, Oxygen
has a 1:2:1 ratio of the above (C1:H2:O1)
Monomers are called monosaccharides
Monomers are single sugar units
Examples↴
Glucose
Ribose
Galactose
Glyceraldehyde
Disaccharides are covalently bonded
known as Glycosidic linkage (A dead giveaway that something is a carb.)
A weak Glycosidic Bond: ⬠\o/⬠\o/⬠ [down:down:down / up:up:up]
A strong Glycosidic Bond: ⬠\o/⬠/o\⬠\o/⬠ [Alternate- up:down:up ]
Lipids
ONLY CONTAIN: Carbon, Hydrogen, Oxygen
Doesn’t have the 1:2:1 ratio for the above
Forms long chains of hydrocarbons
Are always hydrophobic
Saturated fats: Carries all the possible hydrogen atoms a carbon chain can hold
Solid at room temperature
Maintains a straight hydrocarbon chain
A diet in saturated fats can lead to cardiovascular disease
Butter and lard are saturated fats
Unsaturated fats: Does not have all the possible hydrogen atoms that the carbon chain could fit
Liquid at room temperature
Creates a bend in a hydrocarbon chain
Good for a blood stream as it flows smoothly
Oils are an example of unsaturated fats
Phospholipids: molecules that contain a fatty acid attached to phosphate
The head (Phosphate) is hydrophilic whilst the tail (the two lipid chains) is hydrophobic
Proteins
ONLY CONTAIN: Carbon, Hydrogen, Oxygen, Nitrogen, and sometimes Sulfur
NEVER contains Phosphorus
Monomers of Proteins are called Amino acids made up of:
Amino Group Terminus
Central Carbon (known as the α carbon)
Carboxyl Terminus
Central R group
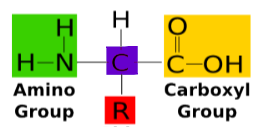
Peptide bonds hold amino acids together
through dehydration synthesis, the -OH from the carboxyl and H from the amino group is removed to form HOH or H2O
This leads to the formation of two amino acids bonded together + H2O
The R-group determines the chemical properties of the amino acids (there are 20)
9 are non-polar
None of them contain Oxygen
6 are polar
All contain Oxygen except Cysteine which contains an “SH“ instead
2 are acidic
Contain negatively charged atoms
3 are basic
Contain positively charged atoms
There are 4 levels of protein structure
All structures contain primary, secondary, and tertiary
Primary- Covalent Peptide bonds
forms the initial chains
Secondary- 3-Dimensional folding arrangement of the Primary structure into either coils (α) or pleats (β).
Held together by hydrogen bonds
Alpha (α) Helix
Beta (β) pleated sheets
Tertiary- Formed as secondary structure is bent and folded into a more complex 3D arrangement
The R-groups interactions play a role in how it bends/folds (hydrophobic/philic)
Bonds can be: hydrogen bonds, ionic bonds, and disulfide bridges
A polypeptide folds into compact 3-D shape stabilized by interaction between r groups of amino acids
These structures can be referred to as Protein Subunits
Quaternary- Composed of two or more protein subunits
Not all proteins have a quaternary structure
Quaternary structures are in a globular shape
Enzymes are a quaternary structure
Enzymes: Help speed up metabolic reactions
Activation energy allows the reaction to take place faster using less energy, making it more convenient
Most enzymes end in “-ase”
Enzymes are reusable
Nucleic Acids
ONLY CONTAIN: Carbon, Hydrogen, Oxygen, Nitrogen, Phosphorus
NEVER contains sulfur
The monomers of nucleic acids are called nucleotides
Made up of a nitrogen base, pentose sugar, and a phosphate
A nucleoside refers to the nitrogen base and pentose sugar alone
Two types of Nucleotides ↴
Pyrimidines | Purines |
Cytosine | Guanine |
Thymine (DNA) Uracil (RNA) | Adenine |
Chargaff’s rule:
Adenine will always pair with Thymine
Guanine will always pair with Cytosine
Phosphodiester bonds: Connect nucleotides to each other on a single strand
hydrogen bonds connect the nitrogen bases of the two strands in a DNA molecule
DNA
stands for Deoxyribonucleic acid
The Phosphate end is considered 5’ (five prime)
the hydroxyl group on the pentose sugar is considered 3’ (three prime)
This orientation when they come together is known as antiparallel
RNA
Stands for Ribonucleic acid
Three types: mRNA, rRNA, tRNA
mRNA: a messenger between DNA and the ribosomes
rRNA: combines with proteins to form the large and small subunits of the ribosome
decodes DNA by making and RNA copy
tRNA: assembles amino acids into proteins through Protein synthesis
The sequence of protein synthesis goes like ↴
DNA → RNA → Protein
DNA vs RNA
5 carbon sugar | 5 carbon sugar |
Phosphate group | Phosphate group |
Nitrogenous base | Nitrogenous base |
Deoxyribose sugar | Ribose sugar |
C, G, T, A | C, G, U, A |
Double stranded | Single stranded |
Original Copy | Disposable copy |
Extra Notes!
STRUCTURE DETERMINES FUNCTION
The oxygen atoms of a water molecule- is more electronegative than hydrogen atoms
✍(◔◡◔) | Storage | Structure |
Plants | Starch | Cellulose |
Animals | Glycogen | Chitin |
Essential Questions
What properties of oxygen and hydrogen lead to the unique properties of water?
Which elements are used to build which macromolecules?
How do these contribute to their properties?
What types of bonds are used to create molecules, macromolecules, and most organic compounds?
What reactions are used to make and break polymers?
Which macromolecules are polar vs. non-polar? why?
How does the structure of these components help determine or influence the function of the molecules discussed in this unit?
How do small changes in the structure impact function