10th grade science exam revision | theory
Physics
Semester 1
Acceleration
- A measurement of how much velocity changes over time
- Velocity - vector quantity of distance travelled over time
- Vector quantity - has a direction
- Caused by a force
- Deceleration - acceleration in the opposite direction
- Average speed = total distance travelled over time taken (d/t)
Distance, displacement
- Distance = the total length travelled
- Displacement = length from point A to B using the most direct and shortest route
- You can calculate displacement via pythag (a^2 + b^2 = c^2)
- Average velocity - the overall displacement of a given motion with direction
- Displacement over time + direction (d/t) + a direction
Quadrant bearings
- Refers to the angle between two quadrants - eg, North 30 degrees East is just saying that from North, if you turn 30 degrees in an east direction. you’ll be at that angle.
- SOHCAHTOA - get the values of the lines that make up the angle you want to calculate, and plug them into the calculator (eg, sin^-1(opposite/hypotenuse))
m/s → km/h
- To go bigger, times by 3.6
- To go smaller, divide by 3.6
- \
Position time graphs
- Shows how far/close an object/person is from a set distance. The end of the line on the right represents displacement.
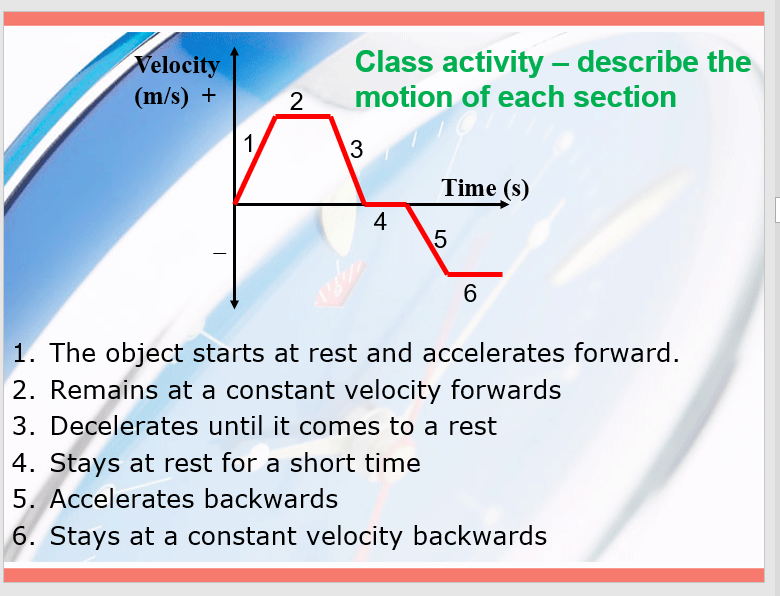
Velocity time graphs
The area is equal to the total displacement
- Look at the area between the origin and under the line for positive axis, and do the same but reversed (over the line, under the origin) for a negative axis.
- Subtract the negative axis stuff from the positive :)
Vector diagrams

Terminal velocity
- Maximum velocity that can be achieved by a mass
Semester 2 N/A
Biology
Semester 1
DNA
- Deoxyribonucleic acid
- Purpose: Stores genetic information
- Is a molecule, known as the blueprints for the human body
- Shape and structure
- Shape: Twisted double helix is formed via two chains.
- These chains are formed via multiple subunits called nucelotides
- Nucleotides - consist of a phosphate group, sugar, and nitrogenous force.
- Nitrogenous force - Adenine and Thymine, Cytosine and Guamine. These form complementary pairs.
- Chromosomes - tightly coiled up DNA
- Each human body cell has 46 of em, or 23 pairs. 22 of those are homologous pairs. Half from your mother, half from your father!
- The 23rd pair is a sex chromosome - these could either be the same (XX) or different (XY)
- Genes - a sequence of DNA on a chromosome that codes for a particular protein that codes characteristics such as eye colour
- Inherited from our parents
- How is DNA replicated?
- Two strands of DNA separate, and then each strand provides a template for making a new strand, and then they link together with those nucleotides.
Mitosis and meiosis
Mitosis
The process that all cells except for sex cells use to divide
A single cell forms into two IDENTICAL daughter cells, which carry the same amount of chromosomes and genetic info as the parent cell.
The stages:
\
- Interphase.
- Prophase
- Metaphase
- Anaphase
- Telophase
- Cytokinesis (split)
Meiosis
The process sex cells (sperm and egg) use to divide.
They have half the number of chromosomes than the parent cells
Genetically different from parent cells.
Produces 4 unique sex cells called gametes.
The stages:
\
- Interphase.
- Prophase
- Metaphase
- Anaphase
- Telophase
- Cytokinesis (split)
- From here, we repeat the process in those two split cells
- Prophase II
- Metaphase II
- Anaphase II
- Telophase II
- Cytokinesis (split, now we should have four varied gametes)
Differences between the two?
| Characteristic | Mitosis | Meiosis |
|---|---|---|
| Goal of process | Identical cells | Variety of haploid cells |
| How many daughter cells made per process? | 2 | 4 |
| Which cells does it occur in? | All sells (somatic) | Sex cells |
| Chromosome number of daughter cells | 46 (same as parent cell) | 23 (1/2 of parent cell) |
| No. of cell divisions | 1 | 2 |
Variations and mutations
- Mutations
- A change in DNA, can cause changes in how an organism looks, behaves, and it’s structures.
- Essential to evolution - mutations are not inherently bad.
- Can be inherited from parent to child, or acquired from the environment or DNA copying wrong.
- Genetic disorders are when DNA has a mutation where there is an altered instruction for making protein.
Homozygous, heterozugous
- Homozygous - if genes in an allele are the same and pure bred. (TT, tt)
- Heterozygous, if the genes in the allele are different. (Tt)
Genotypes and phenotypes
- Phenotype - VISIBLE APPEARANCE.
- Genotype - the genetic makeup.
Dominant and recessive alleles
- Alleles are alternative forms of a gene. Basically, if this was on a WIki, the gene would be a category, and the alleles would be the pages. For example, blue and brown are alleles of eye colour.
- Dominant alleles mask or hide other alleles.
- Recessive alleles are masked or covered up whenever the dominant allele is present.
- The law of dominance states that recessive alleles will always be masked by dominant alleles - recessive alleles cannot be present unless there are no dominant alleles.
- Incomplete dominance happens when genes have equal dominance, and the characteristics blend to give an intermediate characteristic (think like mixing paint)
Parent generation + F1 and F2 gen
- Parent gen is the first gen in a family tree, F1 and F2 just keep going on and on down…
Semester 2
Artificial selection/selective breeding
- The process of breeding for certain favourable traits. Done by humans
Natural selection
- The principle of “survival of the fittest” - animals with traits that suit their environment are more likely to survive and produce offspring with that genetic makeup. Those with traits that do not favour the environment will die.
Speciation
- Variation - variation and mutation is present in species
- Isolation - species are isolated from each other, creating different genetic pools to adapt to different envrionments
- Separation - species cannot interbreed
- Speciation - species formed! yay
Evolution
- The change in the heritable traits of biological populations over successive generations.
Proof of evolution
- Fossils
- Early fossils are quite simple, while later ones are more complex.
- Transition fossils to show the “inbetween” stages of how an organism has evolved, showing traits of the original and developed organism.
- Embryology
- Before a embryo is exposed to it’s environment, they all kinda look pretty similar, depending on what common ancestor they have.
- Anatomy
- Many organisms that share common ancestors have similar anatomy.
- DNA
- Similar anatomy = common genes
- Distribution
- Isolated island -→ high proportions of unique species
- Continents (now separated across oceans) have identical fossil species based on where they were once connected in the past.
Dating techniques
- Relative dating.
- When you date fossils based on youngest to oldest - there’s no date on them, we just try to figure out which came first, and which came last.
- Stratigraphy - we assign the age of rock bands by looking at the fossils contained within them.
- Index fossils - common fossils in different areas which were made at the same time, which can be used to compare fossils in different locations.
- Absolute dating
- Using carbon (or other substances) to get the exact age in years of a fossil. Quantifying the age.
- Radioactive decay
- When elements break down, they give off small particles and radiation.
- Carbon-14 has a half life of 5735 years.
- Tree rings
- Similarly spaced
Chemistry
Semester 1
Atomic structure and the periodic table
- How are atoms structured?
- Well, they’re made up of a nucleus. This consists of the proton and neutron. Then, it’s surrounded by electrons. The number of protons and electrons in a balanced atom are equal.
- These protons are equal to the atomic number!
- The mass number is the amount of molecules in the nucleus - this means that the amount of neutrons is equal to mass no - atomic no.
- Electrons are arranged in energy levels: 2, 8, 8, 16. Remember, the electrons are equal to the protons, which are equal to the atomic number! The amount of electrons on the outer shell are called valence electrons.
- The charge of IONS are linked to the amount of valence electrons. For example, elements in Group 1 form a +1 charge.
Naming and formula
- Metal first, then base
- The best practice for this is honestly just practise questions
Chemical reactions and collision theory.
- Collision theory
- For chemical reactions to occur, particles must collide with another.
- More heat → more energy to collide → more efficient reactions
- These can be sped up by a catalyst, which is something that speeds up a reaction but is chemically unchanged.
- What is a catalyst?