Biology - Biological Molecules 2
Biological Molecules Section 2
Starch, glycogen and cellulose
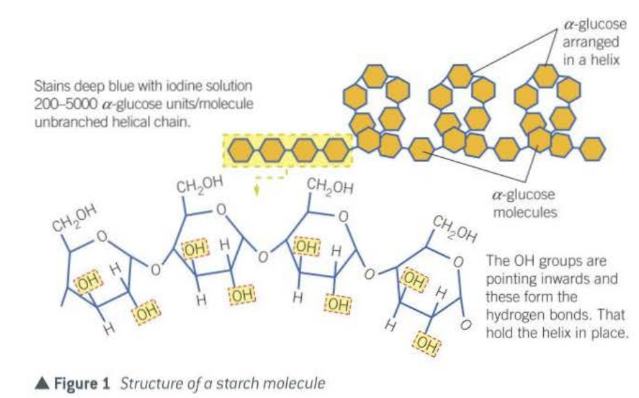
- Starch in plant cells (alpha glucose)
- made up of chains of a-glucose monosaccharides linked by glycosidic bonds
- glycosidic bonds formed by condensation reactions
- chain may be branched/unbranched
- main roles of starch;
- insoluble and doesn’t affect water potential
- large and insoluble, it does not diffuse out of cells
- compact, so a lot can be stored in a small space
- hydrolysed to form a-glucose which is both easily transported and readily used in respiration
- branched form has many ends each of which can be acted on by enzymes so glucose monomers can be released very rapidly
Glycogen in animal, bacteria cells (alpha glucose)
similar structure to starch but shorter chains and highly branched
stored as granules in animals, mainly in muscle and liver cells
its structure is suitable for storage because;
- ==insoluble== and does not tend to draw water into the cells
- ==insoluble==, so cannot diffuse out of cell
- ==compact,== so a lot of it can be stored in small space
- ==more highly branched than starch== so has more ends that can be acted on by enzymes so faster release of glucose monomers
- ==faster release of glucose monomers== as animals have higher metabolic rate
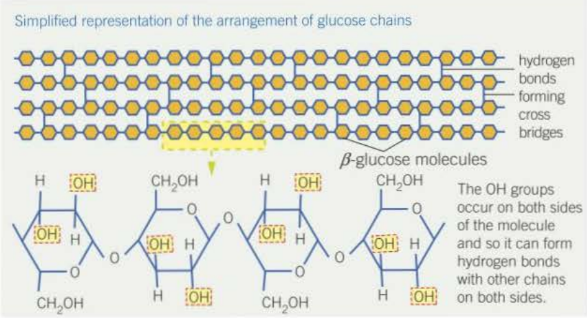
Cellulose
- made of b-glucose
- cellulose a major component of plant cell walls and provide rigidity to the plant cell
- structure of cellulose related to its function;
- cellulose molecules are made up of b-glucose and so form long, unbranched molecules
- the chains run parallel to each other and are crossed linked by hydrogen bonds which adds strength
- these molecules are grouped to form microfibrils which in turn are grouped to form fibres which provides even more strength
Lipids
Characteristics
- contains C, H, O
- proportion of ==oxygen== to ==carbon and hydrogen== is smaller than in carbohydrates
- insoluble in water
- soluble in organic solvents such as alcohol
- main groups of lipids are triglycerides and phospholipids
Roles of lipids
- contributes to the flexibility of the cell membranes and the transfer of lipid soluble substances across them
- source of energy : when oxidised, they produce more than twice the energy the same mass of carbohydrate and release valuable water
- waterproofing : lipids are insoluble in water so it is useful for waterproofing
- insulation : fats are slow conductors of heat and when stored beneath the body surface helps to retain body heat. also acts as electrical insulators in the myelin sheath
- protection : fat is stored around delicate organs such as kidney
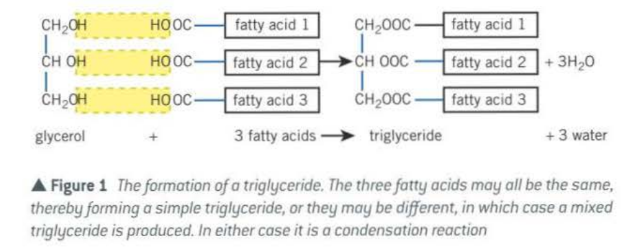
Triglycerides
- three fatty acids + glycerol = triglyceride
- each fatty acid form ==ester bond (-COO-)== with glycerol in a condensation reaction
there are different variations of the triglycerides
no double bond ; saturated
one double bond ; mono-unsaturated
more than one double bond ; polyunsaturated
Structure of triglycerides related to their properties
high ratio of energy-storing carbon-hydrogen bonds to carbon atoms, so excellent source of energy
low mass to energy ratio, good storage molecules, more energy can be stored in a small volume
insoluble in water since they are large non-polar molecules. does not affect water potential of cells
high ratio of hydrogen to oxygen atoms, triglycerides release water when oxidised so it is important source of water
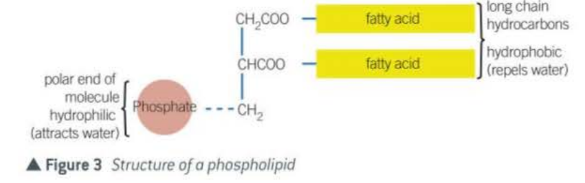
- Phospholipids
similar to lipids but one of the fatty acids are replaced with a phosphate group
has hydrophilic head (phosphates) that are attracted to water
has hydrophobic tails (fatty acid molecules) that repels water but is fat soluble
Structure of phospholipids related to their properties
- polar molecules ; in aqueous environment, phospholipid molecules form a bilayer within cell surface membranes, forming hydrophobic barrier
- hydrophilic phosphate heads help to hold the surface of csm
- phospholipid structure allows to form glycolipids by combining with carbohydrates within the membrane
Test for lipids - emulsion test
1- take a completely dry and grease free test tube
2- to 2cm^3 of the sample being tested, add 5cm^3 of ethanol
3- shake the tube thoroughly to dissolve everything
4- add 5cm^3 of water and shake gently
5- a milky white emulsion indicates presence of lipids
Proteins
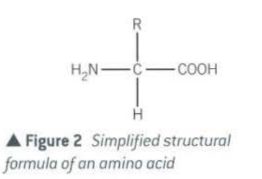
- Structure of amino acid
- amino acids : basic monomer units
- they combine to form polymer; polypeptide
- every amino acid has;
- amino group (-NH2) a basic group
- carboxyl group (-COOH) an acidic group
- hydrogen atom (-H)
- R(side) group , a variety of different chemical groups
The formation of a peptide bond
- amino acids join together to form peptide bonds by condensation reaction and gives water
- water made by combining an -OH from the carboxyl group of an amino acid and a -H from the amino group from another amino acid
- peptide bond can be broken by hydrolysis forming its two constituent amino acids
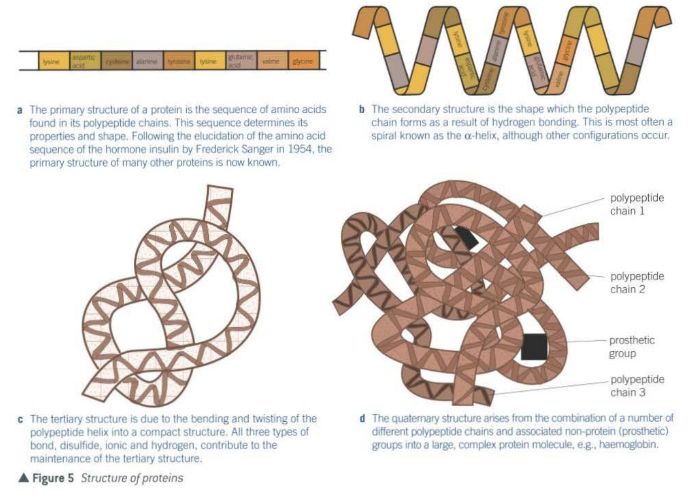
Primary structure of proteins - polypeptides
- many acid monomers joined together in polymerisation
- forms polypeptide
- sequence of the amino acids forms the primary structure of protein
- sequence determined by DNA
- primary structure determines shape so therefore its function
- change in sequence of the amino acids lead to change/stops the function of the protein
Secondary structure of proteins
- linked amino acids have both -NH and -C=O groups on either side of every peptide bond.
- H in -NH (+ve), and the O in -C=O (-ve) form weak hydrogen bonds
- this bond causes the polypeptide chain to twist into 3-D shape such as the a-helix coil
Tertiary structure of proteins
- further twisting and coiling of the secondary structure
- maintained by different bonds
- disulfide bridges - very strong
- ionic bonds - formed between any carboxyl group and amino groups that are not involved in forming peptide bonds. weaker than disulfide bonds
- hydrogen bonds - numerous but easily broken
- specific 3D shape gives its function and allow interaction with other molecules
- Quaternary structure of proteins
- a combination of a number of different polypeptide chains
- there may also be non-protein (prosthetic) groups associated with the molecule
- Test for proteins (blue - purple)
- place a sample of the solution to be tested in a test tube and add an equal volume of sodium hydroxide solution at room temperature
- add a few drops of very dilute (0.05%) copper II sulfate solution and mix gently
- a purple coloration indicates the presence of peptide bonds and hence a protein. if no proteins are present, the solution remains blue