The pH Scale
The pH scale is a logarithmic scale that measures how acidic or basic a substance is. pH stands for “Power of Hydrogen”. It compares the hydrogen ion concentration across a broad range of substances.
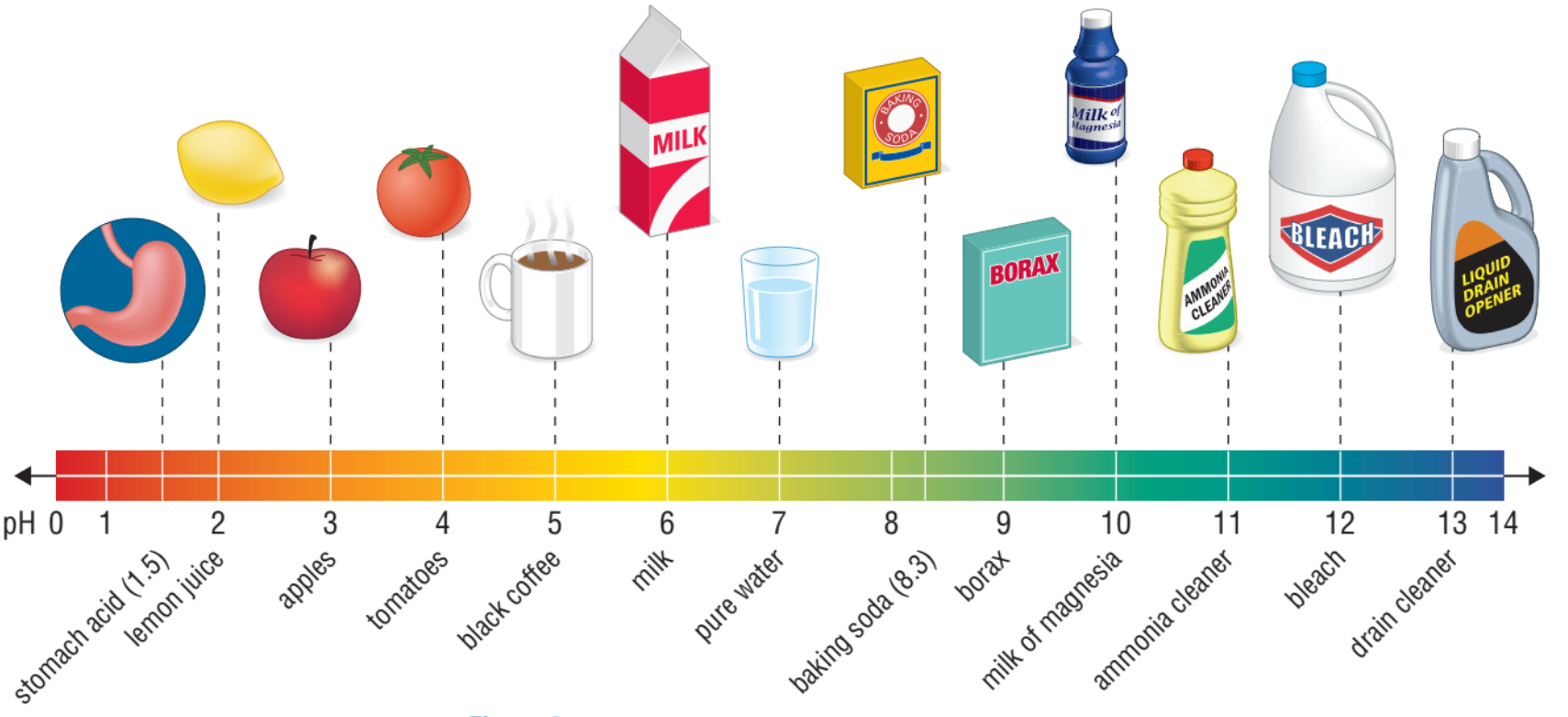
A pH of 7 is considered neutral, neither basic nor acidic. Under 7 indicates that it is acidic, and over 7 indicates that it is basic.
Properties:
Acids are corrosive, and taste sour. When dissolved in water, they form positive H+ ions.
Bases have a bitter taste, feel soapy and slippery to the touch. When dissolved in water, they release hydroxide ions (OH-). Highly basic substances are also corrosive and reactive.
Logarithmic:
Since the scale is logarithmic, a change of 1 on the scale means a 10x increase in H+ concentrations. For example, a solution with a pH of 3 is 10x more acidic than a solution with a pH of 4. A solution with a pH of 5 is 10^5 = 100,000x more acidic than a solution with a pH of 10.
Neutralization:
When an acid and base react, they neutralize, forming a salt (a metal+non-metal) and water (H2O). It is a specific kind of double displacement reaction. The H+ ions combine with the OH- ions, and the positive ion from the acid (metal) and negative ion from the base (metal) form the salt.