Chapter 3- Separation and Purification
- A pure substance consists of a single element or compound.
- Pure substances have a constant and fixed boiling and melting point.
- Impurities cause variation in the melting and boiling points of the substance. Therefore, they can be used to determine if a substance is pure or not.
- Different purification techniques can be used to separate and purify substances.
| Method | Usage | Example |
|---|---|---|
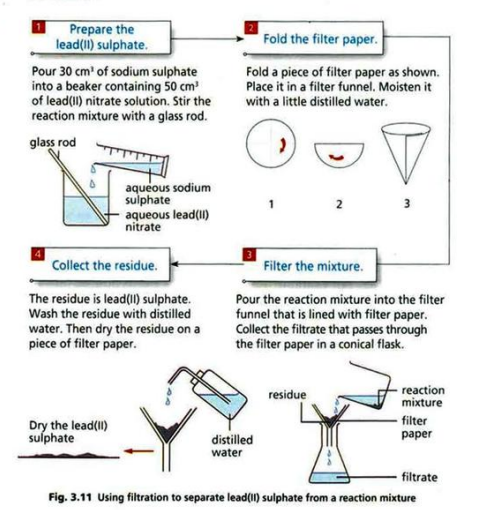
| Filtration | Insoluble solid from liquid | Sand and water |
| Evaporation to dryness | Soluble solid from a solution | To get salt from salt solution |
| Crystallization | Pure solid from a solution | To get Copper (II) sulfate from water |
| Solvent in which only one solid is soluble | A Mixture of two solids | Sand and salt |
| Sublimation | Solid that sublimes with one which doesn’t | Iodine and sand |
| Magnet | Magnetic substance from a non-magnetic one | Iron filings and sulfur |
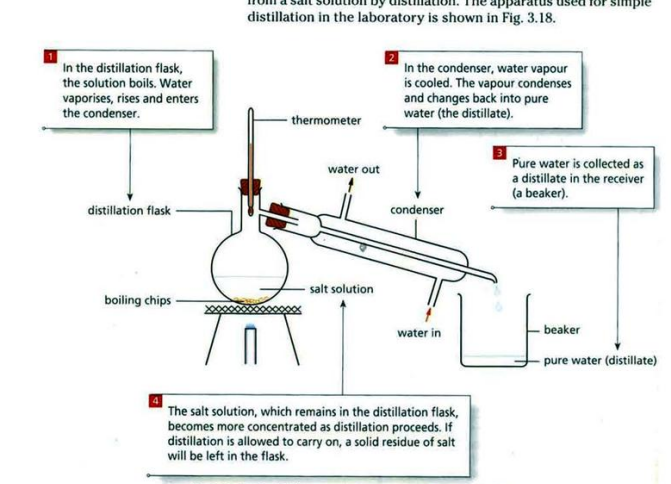
| Simple distillation | Pure solvent (liquid) from the solution | To get water from a salt solution |
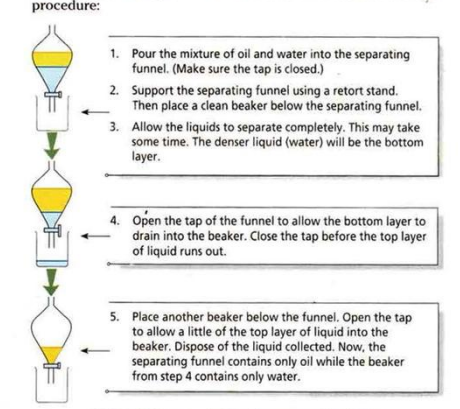
| Separating funnel | Immiscible liquids | Oil and water |
| Fractional distillation | Miscible liquids with different boiling points | Water and ethanol |
| Chromatography | Components that dissolve in the same solvent | Different dyes |
| Centrifugation | Mixtures by spinning | Blood cells from plasma |
| Decanting | Liquid and insoluble particles | Rice from water |
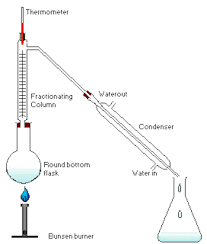
STEPS TAKEN DURING DISTILLATION:
- Thermometer should be placed beside the sidearm of the distillation flask, not inside the liquid.
- Water outlets should be positioned in the correct way with cold water in from the bottom and leaving from the top.
- The condenser slopes downward so the pure solvent runs to the flask/beaker.
- If the distillate is volatile, it has to be kept in a container containing ice so it doesn’t evaporate.
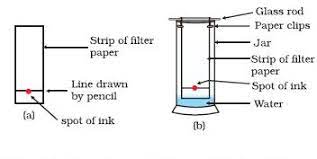
Chromatography
- Different components separated using chromatography have distinct Rf values or Retention factors.
- Rf value is calculated by dividing the distance moved by the solute by the distance moved by the solvent.
- Rf values can be matched by the already known values to identify the component.
- Sometimes, the components are transparent such as amino acids. A locating agent (e.g. Ninhydrin) is sprayed on the chromatogram to make them visible.