Lecture 3 - Cycloalkanes and their Stereochemistry Rev CME 2024
Lecture 3: Cycloalkanes and their Stereochemistry
Naming Cycloalkanes
Cycloalkanes or alicyclic compounds: Saturated cyclic hydrocarbons
Can be represented using skeletal drawings
Follow general rule of naming structures, however, use “cyclo” at the start of the suffix.
Cis-Trans Isomerism in Cycloalkanes
Cycloalkanes are less flexible than open-chain alkanes.
Significantly lesser conformational freedom in cycloalkanes
Cycloalkanes have two faces, when viewed edge-on, owing to their structure (top face and bottom face)
Isomerism is possible in substituted cycloalkanes
Stereoisomerism: Compounds which have their atoms connected in the same order but differ in 3-D orientation
Stereochemistry: Term used to refer to the 3-D aspects of chemical structure and reactivity.
Cis-trans isomers: Stereoisomers that differ in their stereochemistry about a ring or double bond.
Stability of Cycloalkanes: Ring Strain
Angle Strain:
Induced in a molecule when bond angles are forced to deviate from the ideal 190° tetrahedral value.
Cyclic molecules can assume nonplanar conformations to minimize angle strain and torsional strain by ring-puckering
Torsional Strain
Caused due to eclipsing of bonds between neighboring atoms
Steric Strain
Caused due to repulsive interactions when atoms approach each other too closely
Larger rings have many more possible conformations than smaller rings; more difficult to analyze.
Conformations
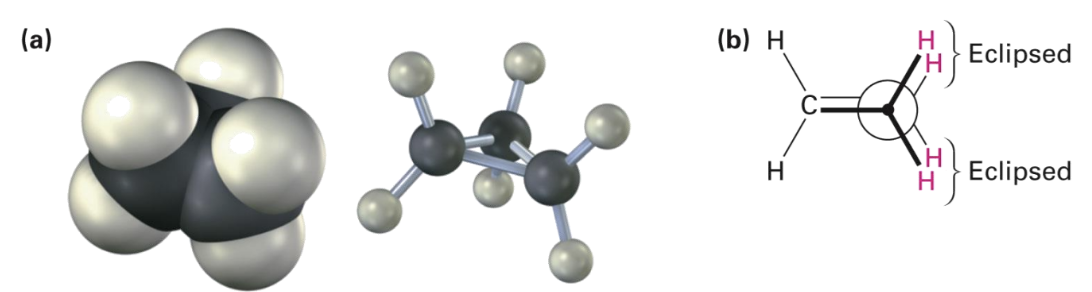
Cyclopropane
Most strained of all rings due to angle strain caused by its C-C-C bond angles of 60°.
Has considerable torsional strain.
Has bent bonds.
C-H bonds are eclipsed.
Weaker and more reactive than typical alkane bonds.
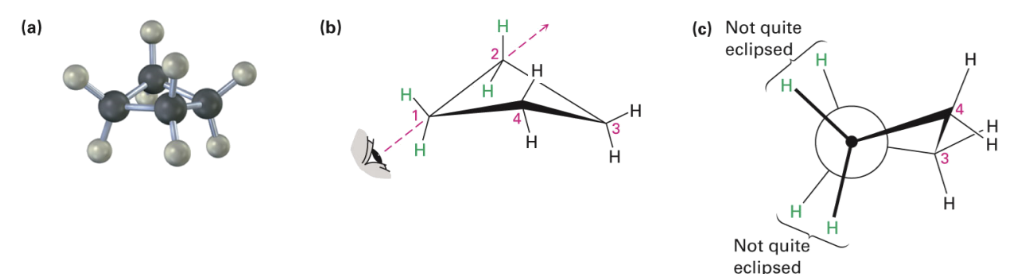
Cyclobutane
Has less angle strain than cyclopropane
More torsional strain because of larger number of ring hydrogens, and their proximity to each other.
Slightly bent out of plane, one carbon atom is about 25° above the plane.
Increases angle strain but decreases torsional strain.
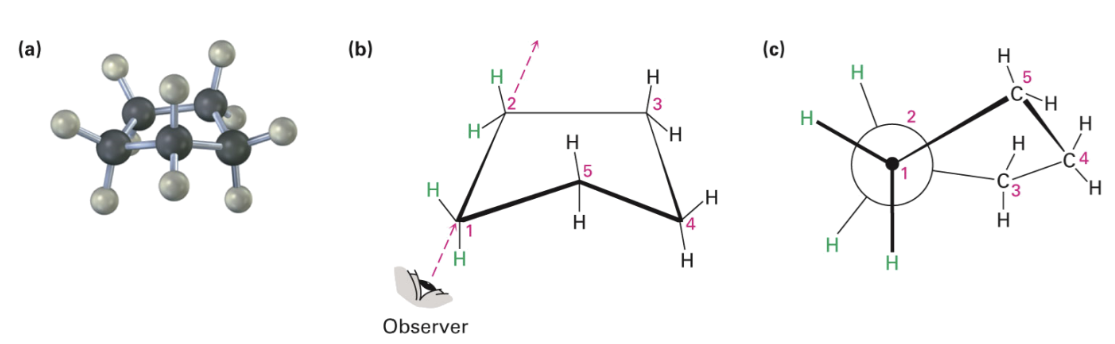
Cyclopentane
No angle strain
Large torsional strain
Non planar conformation strike balance between increased angle strain and decreased torsional strain
Four carbon atoms are approximately in the same plane
Fifth carbon atom is bent out of the plane
Cyclohexane
Cyclic version of hexane, an alicyclic hydrocarbon with a ring of six carbon atoms
a colorless, mobile liquid with a mild, sweet odor
slightly soluble in water and soluble in alcohol, acetone, benzene, ethanol, ethyl ether, olive oil, and carbon tetrachloride; performs the function of a non-polar solvent
commonly used as a chemical intermediate such as utilized as a raw material in the creation of nylon
Melting point: 6.47 C
Boiling point: 80.7 C
Specific gravity: 0.779
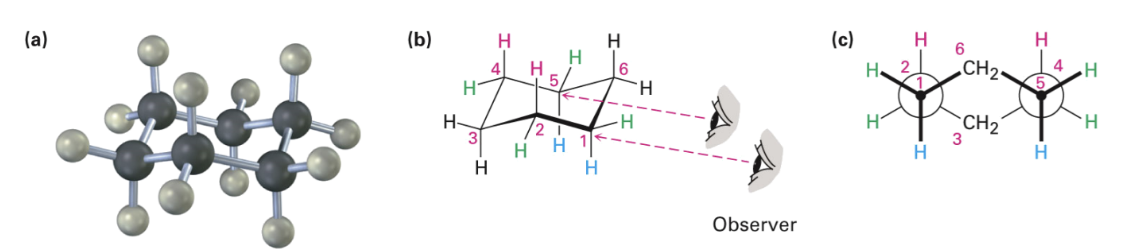
Adopts chair conformation: Strain-free, 3-D shape
Has neither angle strain nor torsional strain
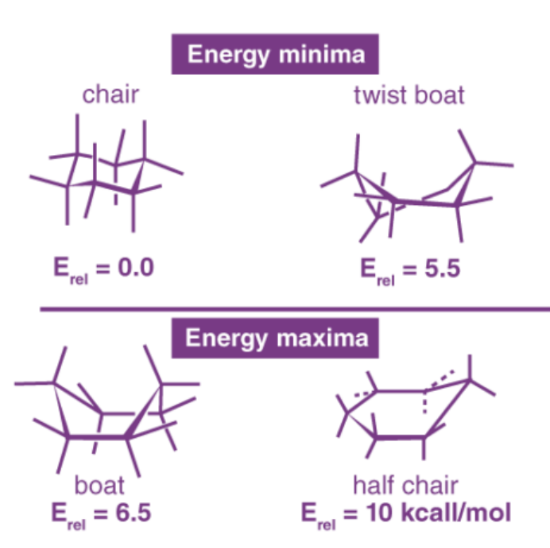
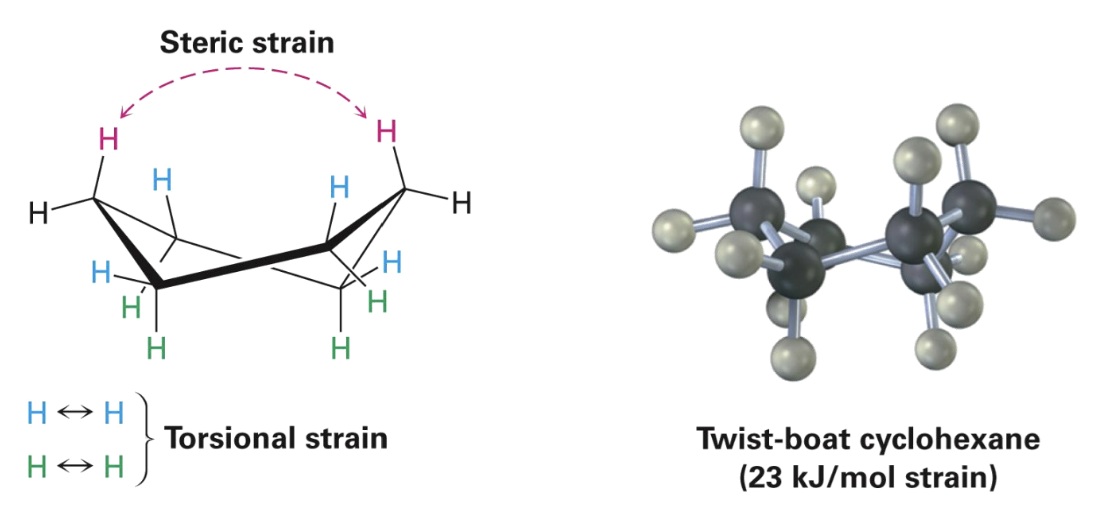
Boat cyclohexane
Alternate conformation of cyclohexane that bears a slight resemblance to a boat
No angle strain
Large number of eclipsing interactions
Twist-boat conformation
Alternate conformation of cyclohexane that is somewhat more stable than a pure boat conformation
Nearly free of angle strain
Axial and Equatorial Bonds
Cyclohexane
Chair conformation positions for substituents on the ring
Axial positions
Equatorial positions
Chair cyclohexane has six:
Axial hydrogens perpendicular to the ring
Equatorial hydrogens near the plane of the ring
Each carbon atom has one axial and one equatorial hydrogen
Each face of the ring has three axial and three equatorial hydrogens in an alternating arrangement.
Conformational Mobility of Cyclohexane
Ring-flip
Interconversion of chain conformations, resulting in the exchange of axial and equatorial positions
Conformations of Disubstituted Cyclohexane
Steric effects of both substituents is taken into account in both conformations
Isomers of 1,2-dimethylcyclohexane
Cis-1,2-dimethylcyclohexane
Trans-1,2-dimethylcyclohexane
Cis isomer
Both methyl groups are on the same face of the ring
Compound can exist in two chair conformations
Trans isomer
Methyl groups are on opposite faces of the ring
One trans conformation has both methyl groups equatorial and only a gauche butane interaction between methyl groups.
Ring-flipped conformation has both methyl groups axial with four 1,3-diaxial interactions
Will exist almost exclusively in di-equatorial conformation
1,3-Diaxial Interactions
Causes Steric Strain
difference between axial and equatorial conformers
Hydrogen atoms of the axial methyl group on C1 are too close to the axial hydrogen three carbons away on C3 and C5
Results in 7.6 kJ/mol of steric strain
Relationship to Gauche Butane Interactions
Gauche butane is less stable than anti butane by 3.8 kJ/mol due to steric interference between hydrogen atoms on the two methyl groups
Four-carbon fragment of axial methylcyclohexane and gauche butane have the same steric interaction
Equatorial methylcyclohexane has no such interactions
More stable