AP Bio
Unit 1
Stems
an-: not
co-:together
valent: strength
electro-: electricity
iso-: equal
neutr-: neither
pro-: before
kilo-: a thousand
hydro-: water
philos: loving
phobos: fearing
hydro-: water
enanti-: opposite
carb-: coal
sulf-: sulfur
thio-: sulfur
con-: together
di-: two
glyco-: sweet
hydro-: water
lyse: break
macro-: large
meros-: part
mono-: single
sacchar: sugar
poly-: many
tri-: three
In Class Notes
kinesis: non-directional response
organisms moving around quickly when they’re unhappy, and slowly when they’re happy
taxis: directional response
-response to specific stimuli, more sophisticated and advanced than kinesis
ethology: the study of animal behavior
the number of bonds an element can form (ex: Nitrogen=3 bonds) determines the molecular shape
living organisms recognize every molecule by shape; shape determines function
cations: metals that have undergone an ionic bond and lost electrons
anions: nonmetals that have undergone an ionic bond and gained electrons
arrows are used in diagrams of ionic bonds to show the transfer of electrons from the metal to the nonmetal
ionic bonds work when the elements are far away on the period table
electronegativity is what really determines what types of bonds will form between elements
electronegativity: how bad an element wants to gain electrons
determining bond type: determine electronegativities and subtract them
Fluorine has the highest electronegativity and the smallest atomic radius, Francium is the opposite
polar covalent bond: when the electronegativities don’t allow the stronger element to fully take the weaker one’s valence electron(s), so it spends more time around the stronger one but hasn’t fully taken it (ex: Hydrogen and Oxygen)
polar covalent symbols: partial positive (weaker element) and partial negative (stronger element)
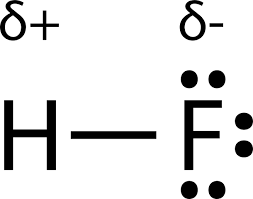
water’s partial covalent bonds cause it to stick to itself and give it a lot of weird properties
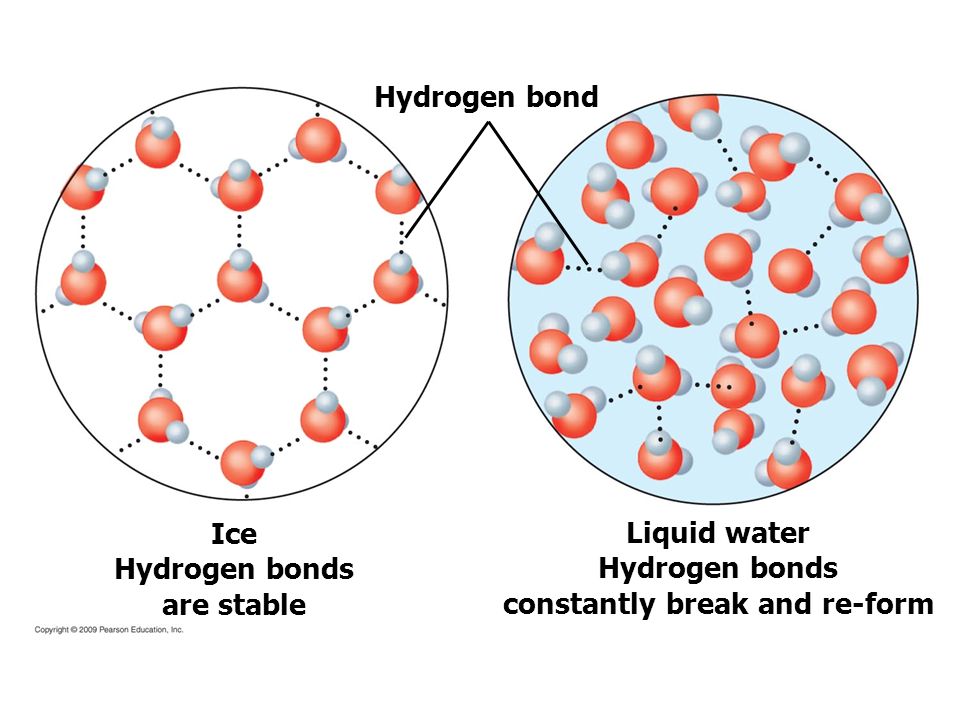
hydrogen bond: the attraction between a partial positive atom and a partial negative atom between separate molecules that are already formed
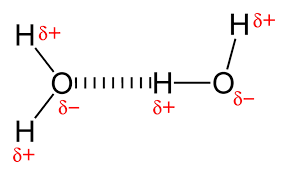
a hydrogen bond is a relatively weak connection, but a lot of them together are really strong
it takes a lot of energy to change the states of water because you have to break some of the hydrogen bonds to get from a solid to a liquid, and all of the hydrogen bonds to get from a liquid to a gas
the symmetry of a molecule determines if it’s polar or nonpolar
polar molecule: asymmetrical (ex: water)
polar molecules will dissolve in other polar molecules
solute: the substance being dissolved in a solution
solvent: the solution a substance is being dissolved in
non-polar molecule: symmetrical (ex: most hydrocarbons)
hydrophilic: substances that like water
anything with a charge is hydrophilic (ex: ions, polar molecules)
hydrophobic: substances
non-polar substances are hydrophobic
pH stands for “potential hydrogen” and is determined by Hydrogen ion concentration
distilled water is a pH 7 because 1.0×10^-7H^+ (the power is 7)
acids have a pH of less than 7 because those substances have more hydrogen ions than distilled water
every number on the pH scale is 10x stronger/weaker than its adjacent numbers (ex: pH 6 is 10x stronger than pH 7, and pH 7 is 100x weaker than pH 5)
bases have a pH of more than 7 because they have less Hydrogen ions than distilled water
a hydrogen ion is 1 proton
an acid is a substance that donates hydrogen ions (protons)
functional groups get added to carbon skeletons because it makes them hydrophilic and therefore able to be processed by water-based living organisms
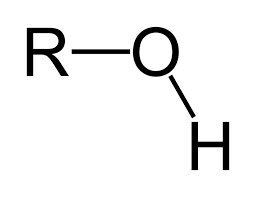
hydroxyl group: OH
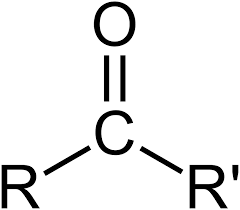
carbonyl group: C=O
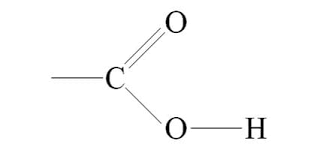
carboxyl group: COOH
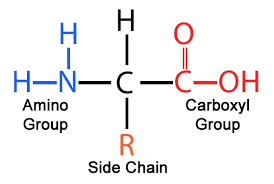
amino group: NH2
sulfhydryl group: SH
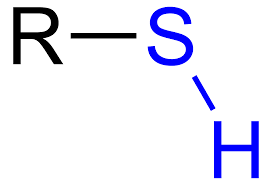
-ane is used for a single-bonded carbon chain, -ene is used for double-bonded carbon chains, and -ine is used for a triple-bonded carbon chain
if there are oxygens somewhere in the molecule it will most likely be polar
macromolecules are relatively large in size and have complex structure
most macromolecules are polymers made from monomers
dehydration synthesis removes a hydroxyl group (OH) from one monomer and an (H) from another monomer to make an open bond site to they can bond
hydrolysis breaks up polymers by adding water (an OH and an H)
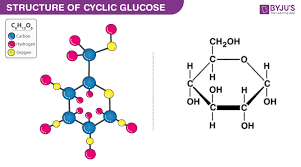
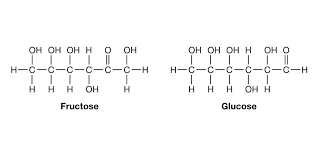
when carbohydrates (sugars) are placed in water they take a ring shape
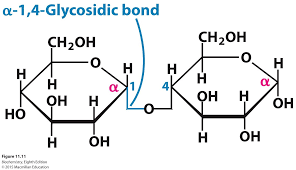
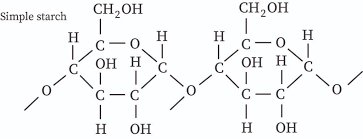
starch: C6H10O5
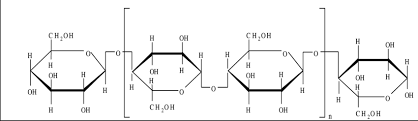
cellulose: C6H10O5
glycogen: C6H10O5
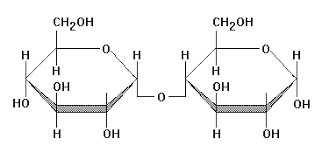
glycogen is normally stored in the liver to help regulate blood sugar levels
starch comes from photosynthesis
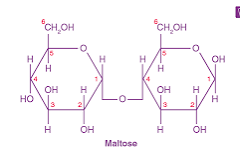
starch can be digested because of the thumb up-thumb up connection in a long chain, not thumb up-thumb down connection
when cellulose chains are lined up next to each other, they form hydrogen bonds between the hydroxyl groups, making a stronger material
excess glucose is stored in the liver and muscle cells
glycogen comes from starch that has been broken into glucose molecules through hydrolysis
glycogen is where glucose is stored after starch is digested
glycogen is stored in the liver and the muscle cells
glycogen branches, starch is just a chain
you can train your body to use other energy sources first before sugar (ex: distance runners use fat for a race and then glycogen for the kick at the end)
excess glycogen becomes fat, which is long-term storage
fats are large molecules and have 3x as many calories as carbohydrates do
unsaturated fats are known as oils
unsaturated fats are from plants
saturated plants are from animals
saturated fats are straight, unsaturated fats are kinked
phospholipids are what make soap
the kink in the unsaturated fat allows them to regulate the rigidity of their cell membranes
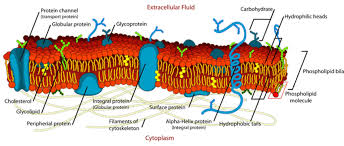
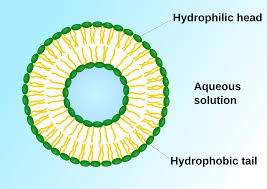
phospholipids are made of a hydrophilic phosphate head and 2 hydrophobic fatty acid tails (hydrocarbon chains)
phospholipid bilayer because the hydrophilic phosphate heads are on the outside and the inside, and then the larger hydrophobic fatty acid tails are facing each other on the inside
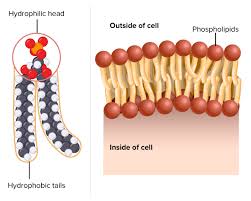
cholesterol helps cell membranes stay flexible
charged and hydrophobic molecules are unable to pass through the fatty acids that make the phospholipid bilayer
every membrane is made from the phospholipid bilayer
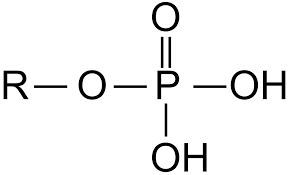
phosphate group: PO4H2
glucose molecule: C6H12O6
maltose molecule (thumb up thumb up): C12H22O11
glycerol molecule: C3H8O3
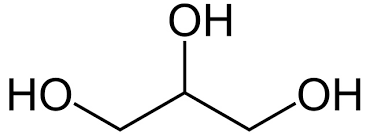
glycerol is a short 3-carbon chain that is often used to bind different fatty acids together and form lipid structures
proteins are the most important macromolecules that are discussed
phospholipids and nucleotides are important
should be able to tell if an amino acids are hydrophobic or hydrophilic
hydrophobic things are usually just C and H
hydrophilic things usually contain O, especially on the end
proteins are chains of amino acid monomers that are connected by covalent peptide bonds in a dehydration synthesis reaction
most living organisms have similar reactions, which is why many living organisms have similar DNA
putting 2 amino acids together in an intramolecular covalent bond is called a peptide bond
protein function ex: immune system, transport, muscle contraction, signaling, production of enzymes
a polypeptide is the same thing as a protein
some hormones are steriods and some are proteins
only living organisms can make protein because they are the only things with DNA
acids donated H and become charged, bases received H and become charged
the shape of a protein determines its function
peptide bonds form between carboxyl and amino groups after dehydration synthesis with nothing else in between
the backbone of amino acids is carboxyl and amino groups, the only things that change are the R groups
4 separate proteins are used to make hemoglobin
women are more likely to be anemic than males because they lose blood from menstruation and iron is part of hemoglobin
4 levels of protein structure:
-primary structure: putting the amino acids in a unique order in a row
-secondary level: folding or coiling of the polypeptide into a repeating configuration resulting from hydrogen bonding of amino and carboxyl groups (nothing with the R groups) and making alpha helixes and beta pleated sheets
-tertiary level: most proteins need to get to this level to do their jobs, results from the interaction between amino acids and R groups and can be any possible type of bond (ex: hydrophobic-hydrophobic bonding, H bonding, sulfide bridges, etc.)
-quaternary level: final protein structure that results from the aggregation of 2 or more polypeptide subunits
if one amino acid is wrong in DNA, it causes sickle cell anemia
it’s possible to change the shape of a protein by changing its environment which causes it to change its shape and therefore its job
when proteins change shape, its irreversible (ex: a fever causes proteins to denature and it’s irreversible)
denaturing:
spiderwebs are secondary level and bulletproof vests are made from a substance that mimics the shape that a spider produces for its web
DNA determines protein shape
an enzyme is made up of proteins and acts as a catalyst that orientates substances in certain ways which allows them to bond up to 10 billion times faster than they usually would
every enzyme has only 1 shape it can accept
proteins serve as structural support, biochemical catalysts, hormones, enzymes, building blocks, and initiators of cellular death
Review Notes:
21 MC, 1 Chi-Square, 1 FRQ about macromolecules
hydrogen bonding: water, cellulose, secondary protein structure
proteins
tertiary level is between R groups and quatrenary level is between differerent peptide chains
hydrophobic R groups are usually carbon and hydrogen
every carbon that gets added onto a carbon chain with an oxygen on it makes it a less polar molecule
do you need to consume starch for it to turn into glucose and then turn into glycogen?
what do we have to know about triglyceride and glycerol
what R groups should we know
what proteins should we know
what is the difference between primary and secondary level
Chapter 5 Note Packet
monomers bind to form polymers through dehydration synthesis
macromolecules are large and complex
polymers decompose into monomers through hydrolysis
carbohydrates: sugars and their polymers
the carbohydrate monomer is monosaccharide
monosaccharides contain only C, H, and O, one hydroxyl group is attached to each carbon, one carbon contains a carbonyl group
polysaccharides are polymers of many monosaccharides that store energy or are structural support
glycogen is an animal’s short-term storage form of energy
lipids are mostly hydrophobic and make membrane structures
lipids are long-term storage of energy (particularly in animals)
lipids regulate cell activities by hormone actions
fats: 1 glycerol and 3 fatty acid tails
glycerol: alcohol
fatty acid: long hydrocarbon chain with a carboxyl group at the end
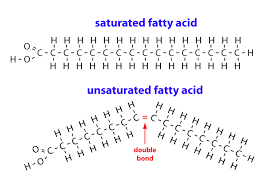
unsaturated fats: one or more double bonds between carbons which allows for kinks in the tails
-liquids at room temp
-mostly from plants
saturated fats: no double bonds on fatty acid tails
-solid at room temp
-mostly from animals
phospholipids: glycerol + 2 fatty acids + phosphate groups
-main structural component of cell membrane (phospholipid bilayer)
waxes: lipids that serve as coatings
steroids: 4 carbon rings without fatty acid tails
-component of cell membranes
proteins: chains of amino acid monomers connected by covalent peptide bonds
-have 3D globular shape
amino acids: monomers of polypeptides, molecules with carboxyl and amino groups
-differ in properties due to differing side chains (R groups)
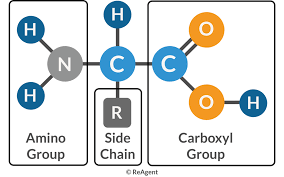
peptide (covalent) bonds connect amino acids to form polypeptide chains one or more polypeptide chains make up protein
nucleic acids: store and transmit hereditary info
genes program the amino acid sequence of proteins and are made of nucleic acids
DNA: deoxyribonucleic acid
-double-stranded
-can self-replicate
-makes up genes that code for proteins
RNA: ribonucleic acid
-single-stranded
-functions in the actual synthesis of proteins coded for by DNA
-is made from the DNA template molecule
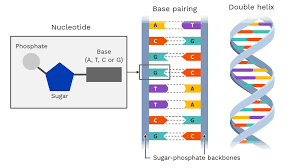
DNA and RNA are composed of nucleotide monomers
nucleotide: pentose (5-C) sugar, phosphate, and a nitrogen base
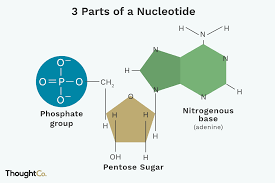
to build a polymer a phosphate group of one of the nucleotides forms a strong covalent bond with the #3 carbon of the sugar of the other nucleotide
peptide bonds: dehydration synthesis between amino acids that takes place in the primary level
hydrogen bonds are most important at secondary level
r group interaction are most important at tertiary level
draw the unbonded pairs
Chapter 2 Hardcover Textbook Notes: Basic Chemistry
carbon, hydrogen, nitrogen, oxygen, phosphorous, and sulfur make up 95% of the body weight of organisms
potassium, calcium, iron, magnesium, and zinc are also important to life
John Dalton developed the atomic theory, which states that elements consist of tiny particles called atoms
atom: the smallest part of an element that displays the properties of the element
AMU: atomic mass unit
protons and neutrons are each assigned one AMU
isotopes: atoms of the same element that differ in the number of neutrons
atomic mass: the average mass for all the isotopes of that atom
the chemical behavior of a radioactive isotope is essentially the same as that of the stable isotopes of an element
molecule: exists when 2 or more elements bond together
compound: a molecule containing at least 2 different elements
organisms obtain energy when electrons inside of them shift in their relationship to one another
single covalent bonds are strong, but double and triple are stronger
calorie: the amount of heat energy needed to raise the temperature of 1g of water 1 degree Celcius
water takes more energy than most other covalently bonded liquids to change temperature because of its hydrogen bonds
water has a high heat capacity
water has a high heat of vaporization because all its hydrogen bonds must be broken to change it to a vapor
when NaCl is put into water, the negative ends of the water molecules are attracted to the sodium ions, and the positive ends of the water molecules are attracted to the chloride ions; this attraction causes the sodium ions and the chloride ions to separate in water
cohesion: the ability of water molecules to cling to each other due to hydrogen bonding
the strong cohesion of water is what allows water to flow freely, yet water molecules don’t separate from each other
adhesion: the ability of water molecules to cling to other polar surfaces as a result of their polarity
surface tension: the force between molecules in a liquid
ice is less dense than water because its hydrogen bonds become more rigid and form a lattice structure at low temperatures, which is also why ice expands more than liquid water
when water ionizes, it releases an equal number of hydrogen ions (protons) and hydroxide ions
only a few molecules dissociate at a time, and the number of hydrogen ions (protons) and hydroxide ions is 1×10-7 mol/L
acids: substances that dissociate in water, releasing hydrogen ions
the acidity of a substance depends on how fully it dissociates in water
bases: substances that either take up hydrogen ions or release hydroxide ions
a pH above 7 is basic because there are more hydroxide ions than hydrogen ions
pH 5 is 100x more acidic than pH 7 and 100x more basic than pH 3
buffer: a chemical or combination of chemicals that keep pH within normal limits by taking up excess hydrogen ions or hydroxide ions
organic: molecules and compounds that contain both carbon and hydrogen atoms
there are only four classes of organic molecules in any living organism: carbohydrates, lipids, proteins, and nucleic acids
biomolecules: carbohydrates, lipids, proteins, and nucleic acids
diversity of life is possible because of the diversity of organic molecules
variety of organic molecules is based on the unique chemical properties of the carbon atom
carbon atoms almost always form covalent bonds
carbon usually bonds with other carbon atoms, or H, N, O, P, S
carbon-carbon bonds are very stable and allow the formation of long carbon chains
hydrocarbons: chains of C atoms that have additional bonds exclusively with H atoms
carbon can form double bonds with itself and other atoms
double bonds are less flexible than single bonds and restrict the movement of bonded atoms
double bonds affect molecular shape and therefore influence function
the presence of double bonds is one way to distinguish between saturated and unsaturated fats
carbon can form triple bonds with itself
branches may form at any carbon atom, making it the ideal building block for biomolecules because it can form long, complex carbon chains
carbon skeleton/backbone: carbon chain of an organic molecule
the carbon skeleton of an organic molecule accounts for its shape
the diversity of organic molecules comes from the attachment of different functional groups to the carbon skeleton
functional group: a specific combination of bonded atoms that always has the same chemical properties and therefore always reacts in the same way, regardless of the carbon skeleton to which it’s attached
the carbon skeleton usually acts as a framework for the positioning of functional groups
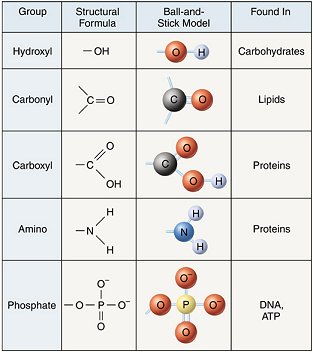
the configuration of functional groups determines the properties of the biomolecule
attached functional groups determine polarity and the types of reactions the molecule will undergo
isomers: organic molecules that have identical molecular formulas but functional groups
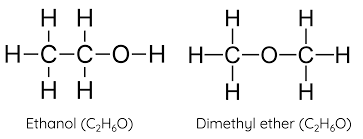
macromolecules: molecules that contain smaller subunits joined together
polymers are constructed by linking together a large number of the same type of subunit called a monomer
carbohydrates, lipids, proteins, and nucleic acids are macromolecules
carbohydrates, proteins, and nucleic acids are polymers
lipids aren’t polymers because they contain 2 different types of subunits
polymers can vary considerably in length
synthesis: building a macromolecule using a dehydration reaction
dehydration reaction: removing the equivalent of a water molecule (an -OH and an -H) as subunits are joined
water molecules are formed as biomolecules are synthesized
biomolecules break down by adding water to them (hydrolysis reaction)
hydrolysis reaction: when an -OH group from water attaches to one subunit and an -H from water attaches to the other subunit
hydrolysis reactions rarely occur spontaneously
usually enzymes act as catalysts that allow hydrolysis reactions to occur or speed up the rate of the reaction
Unit 2
In Class Notes
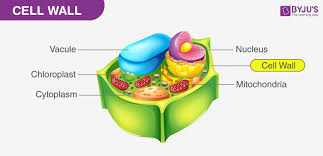
plants have a cell wall, vacuole, and chloroplasts that animal cells don’t have
animal cells have a centriole, which plant cells don’t have
starch is stored in the vacuole
all eukaryotic cells have mitochondria because they have to make ATP
Prokaryotes: a single strand of DNA not inside a nucleus, few simple organelles, found in bacteria and archaea, have a single circular chromosome
eukaryotes: have a nucleus and a variety of organelles, found in plants, animals, fungi, protists
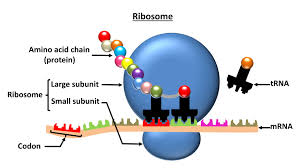
ribosomes make proteins
ribosomes are made of amino acids which are organized by DNA
eukaryotes have membrane-bound organelles (they have membrane on the outside), which allows them to compartmentalize and function without interference
if something is visible with the naked eye it’s eukaryotic
cells are small because it allows for things to get into and out of the cells efficiently
cells have to have a high surface area/volume ratio so they can diffuse things in and out very quickly, which is also why most cells are circular
all cells need a phospholipid bilayer as a cell membrane
cells need a large surface area to a small volume
cells have different shapes that have different functions
the larger the surface area to the volume, the better you are at moving things in and out
photosynthesis is how plants get glucose
almost all bacteria and fungi have cell walls
plants have cell walls made of cellulose
most plant cells are rectangular
any membrane is a phospholipid bilayer
phospholipid bilayer: selectively permeable, small hydrophilic head, large hydrophobic tales
cell membrane: phospholipid bilayer
unless you have an identical twin, no one has an identical carbohydrate chain attached to the cell membrane
white blood cells consume things they believe to be foreign which is what causes organ rejection
auto-immune problems: white blood cells get rid of parts of the body that are supposed to be there
nuclear pores allow certain things to enter and exit the nucleus(ex: RNA can leave to tell ribosomes what to do, but DNA can’t leave)
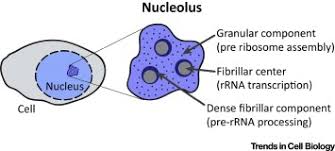
nucleolus: inside the nuclear, makes ribosomal RNA and ribosomal subunits
ribosomes make proteins
where the ribosomes are located determines what proteins they produce, but there is only one kind of ribosome
free ribosomes: suspended in cytosol, make proteins used in cytosol
bound ribosomes: attached to the ER, builds secretory proteins destined for the cell membrane and/or exportation
the dehydration synthesis reaction between amino acids happens in the ribosome
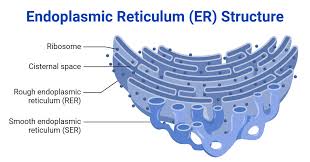
smooth ER doesn’t have ribosomes on it, rough ER has bound ribosomes attached
smooth ER: synthesis of lipids (steroids and sex hormones (hydrophobic)), storage of Ca ions that aid in muscle contraction, detoxification of drugs and poisons, metabolism of carbohydrates (ex: liver, add hydroxyl groups to the drug, making them polar and more soluble and therefore “flushable”)
“drugs” include anything foreign the body needs to get rid of
take vitamin D
the body makes more smooth ER when the body is placed under stress to a drug (ex: people becoming immune to ibuprofen because there is more smooth ER to process it quickly)
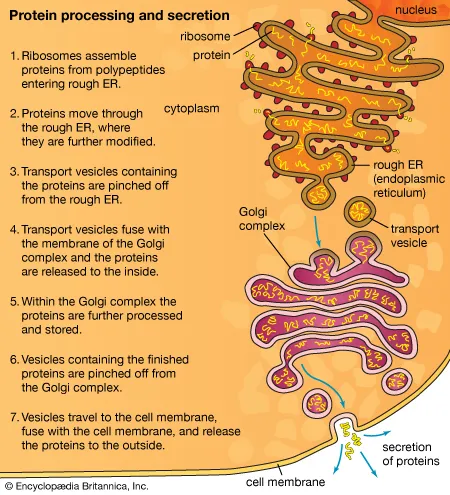
rough ER: rough due to bound ribosomes, assembly of secretory proteins (due to bound ribosomes)
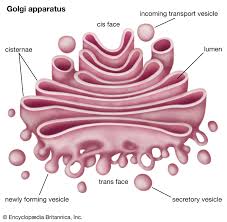
Golgi apparatus: consists of flattened membranous sacs called cisternae, modifies, sorts, packages, transports, and stores proteins that were made in the rough ER
proteins are made inside the ER with the help of messenger RNA so the cell can’t get at it, then they are sent in a membrane to the Golgi body to make finishing touches, then it gets sent in a membrane through the cell membrane and be released out of the cell
transport vesicle: what transports protein to the Golgi body
rough ER (attached to nucleus exterior) to transport vesicle to Golgi body to secretory vesicle to cell membrane (either stays in the cell membrane or goes outside of the cell
because all membranes are made from phospholipid bilayers, they can meld into each other
microtubules are what lead the transport vesicles
golgi makes the secretory protein into the correct shape
the cytoplasm of the cell is a pretty harsh environment, which is why things that are being transported (Ex: proteins going to the Golgi body, proteins going to the cell membrane, messenger RNA) must be protected by membranes or something else
the cytoskeleton gives the cells its shape
vesicle: small pouches or sacks that transport proteins and other cellular materials, they arise from the Golgi, they fuse within the cell membrane
endocytosis: taking in materials from outside the cell
exocytosis: releasing materials out of the cell
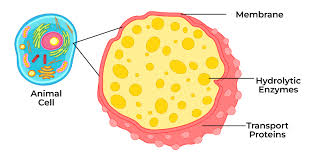
lysosome: membranous sac of hydrolytic enzymes, used to digest macromolecules, found in animals cells, acidic environment aids in digestion,
lysosomes break open and digest cells
letting lysosomes break open will tenderize meat, but when too many break open it leads to rot
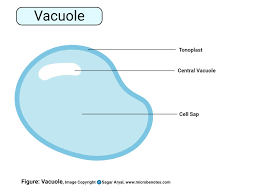
vacuoles: found in plants and fungi, perform hydrolysis on various materials, may also pump excess water out of the cell, may provide storage and structural support
pumping takes energy because it’s working against gravity
pumps require ATP or some other sort of energy
starch is stored in a central vacuole in plant cells
mitochondria: outer and inner membrane, found in all eukaryotes, site of cellular respiration and ATP production, results in transferring energy from glucose to ATP, oxygen is needed (reactant) and water and carbon dioxide are waste (product)
every cell in your body needs oxygen
every cell needs to be 3 cells away from a capillary to ensure that they will receive oxygen
the inner membrane of mitochondria (cristae) is folded to increase surface area, which increases productivity
mitochondria are bean-shaped and have a maze-like folded inner membrane
have to have a membrane to produce ATP efficiently
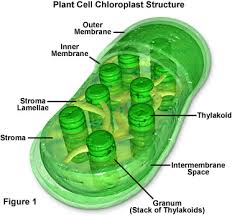
chloroplast: site of photosynthesis, light energy, CO2, and H2O are used to make glucose and oxygen, found only in the cells of leaves and some stems
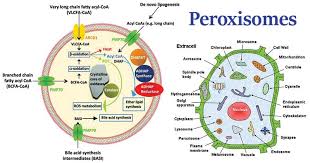
peroxisome: specialized metabolic compartment, containing enzymes that transfer H for various substrates to O, which produces H peroxide
microtubules: help with motility (movement), maintain cell shape, move chromosomes during cell division, move organelles, create a path for vesicles and other things being transported
a larger surface area to volume area makes getting things in and out of the cell more efficient (ex: deer in Flordia are a lot smaller so they can efficiently get rid of heat, deer in Wisconsin are bigger so they can retain heat for longer)
protein channels are created to let large or polar molecules through the cell membrane more quickly
Review:
be able to tell the difference between plant and animal cells
know the organelles in order that are needed to secrete a protein
study root words for quiz
look at book chapters 4-5
be able to recognize organelles
know what can and can’t pass through the cell membrane
general example for last FRQ, make it obvious where that protein is being used
higher surface area/volume is more efficient
if you have 2 cubes, the smaller one will have higher surface area ot volume ratio
exocytosis when something si being secreted out of the cell membrane
protein secretion is active transport
endosymbiotic theory: the idea that mitochondria and chloroplasts were originallyprokaryotes on their own that were engulfed by a larger cell and eventually became codependent
-this happened first with chlorplasts because Earths atmop=sphere had very little oxygen before phorosynthesis
secretory proteins: inside the membrane our outside of it
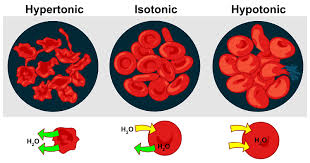
hypertonic solution will have a lower water potenial because the higher c value makes a more negaitve number
no root words on test
study more organeeleS: lysosomes, golgi body, nuclear membrane, nucleus, nocleoulis, cell wall, cell membrane
higher osmolarity = more hypertonic
Stems:
centro-: the center
soma: a body
chloro-: green
cili-: hair
-ell: small
endo-: inner
eu-: true
extra-:
flaggel-: whip
glyco-: sweet
lamin-: sheet/layer
lyso-: loosen
micro-: small
tubul: a little pipe
cytos: vessel
pro-: before
karyo-: nucleus
pseudo-: false
trans-: across
vacu-: empty
aqua-: water
co-:together
trans-: across
endo-: inner
cyto-: cell
exo-: outer
hyper-: exceeding
hypo-: lower
iso-: same
phago-: eat
pino-: drink
liga-: bound or tied
trans-: across
Organelles and Parts Packet
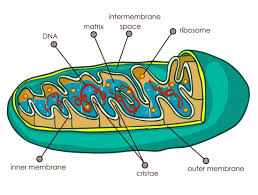
mitochondria:
carries out cellular respiration and produces ATP molecules
10-50 cm long, 2.5-5 cm thick
consists of an inner portion and an outer portion. The inner mitochondrial membrane forms folds known as cristae and separates the innermost area (the matrix) from the inter-membrane space. The outer membrane separates the inter-membrane space from the cytoplasm
inner and outer membranes both made of phospholipid bilayers
found in eukaryotes
found in both plant and animal cells
chloroplast:
carries out photosynthesis and produces sugars
25 cm long and 2.5-5cm thick
grana: lamellae or thylakoids, chlorophyll pigments
stroma: grana, enzymes, DNA, ribosomes, etc
in eukaryotic cells
found in plant cells
peroxisomes:
vesicle that is involved in fatty acid metabolism
2.5-6 cm round
phospholipid bilayer with many membrane-bound proteins
found in eukaryotic cells
found in plant and animal cells
ribosomes:
particles that carry out protein synthesis
.15 cm round
made of nucleotides (ACGU)
found in eukaryotic and prokaryotic cells
found in plant and animal cells
cell membrane:
outer surface of cell that regulates the entrance and exit of molecules
.02 cm thick
phospholipid bilayer
found in eukaryotic and prokaryotic cells
found in plant and animal cells
cell wall:
outer surface that shapes, supports, and protects cell
-.5cm-50cm thick
-cellulose microfibrils, cross-linking glycans, pectin, polysaccharides
-found in eukaryotic and prokaryotic cells
-found in plant cells
endoplasmic reticulum (rough and smooth):
rough ER: studded with ribosomes that synthesize proteins
smooth ER: synthesizes lipid molecules
-25-50cm (around nucleus)
-nuclear envelope, peripheral tubular ER, peripheral cisternae, plasma membrane, mitochondira, Golgi, endosomes, peroxisomes
-found in eukaryotic cells
-found in plant and manmial cells
golgi bodies:
processes, packages, and secretes modified proteins
-2.505cm by 5-10cm
-cisternae, associated vesicles
-found in eukaryotic cells
-found in plant and animal cells
lysosomes:
vesicle that digests macromolecules and cell parts
-1.25cm-5cm round
-lipids, proteins, single membrane
-found in eukaryotic cells
-found in plant and animal cells
vacuole:
sac used for storage of waste products
-120 cm (80% of cell)
-water containing inorganic and organic molecules including enzymes in solution, sometimes solids
-found in prokaryotes and eukaryotes
-found in plant and animal cells
vesicle:
-1.25cm-5cm round
-fluid or gas surrounded by a phospholipid bilayer
-found in eukaryotic cells
-found animal and plant cells
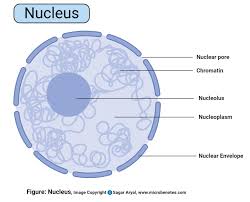
nucleus:
command center of cell
-20cm-50-cm round
most of the cell’s DNA, surrounding the nuclear matric and enveloped in the nuclear envelope
found in eukaryotic cells
found in plant and animal cells
DNA/chromosome/chromatin:
10,000,000cm long
nucleotides (AGTC)
Each chromosome consists of a single, enormously long linear DNA molecule associated with proteins that fold and pack the fine DNA thread into a more compact structure. The complex of DNA and protein is called chromatin
prokaryotic DNA is found in the cytoplasm, whereas eukaryotic DNA is found in the cell's nucleus
found in plant and animal cells
nuclear membrane (envelope)/pores:
2 phospholipid bilayers with nuclear pores that enclose the nucleus
pores-.5cm
two phospholipid bilayers. The membrane facing the cytoplasm is termed the outer nuclear membrane (ONM), and the membrane facing the nucleoplasm is termed the INM.
found in eukaryotic cells
found in plant and animal cells
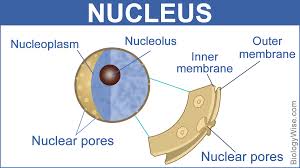
nucleolus:
center of nucleus that produces subunits of ribosomes
5-10cm round
a ribosome factory, composed of deoxyribonucleic acid (DNA), ribonucleic acid (RNA), and protein
found in eukaryotic cells
found in plant and animal cells
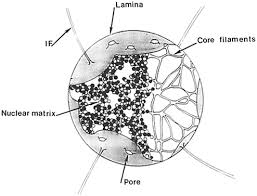
nuclear matrix and nuclear lamina:
lamina is 30-100nm thick
The nuclear lamina is essential for nuclear integrity and is composed of intermediate filaments made up of lamins and lamin-associated proteins. These proteins include the lamin-associated protein (LAP) 1 and 2 families, the lamin B receptor (LBR), and Emerin [24,21]
“nuclear matrix” (NM)1, 2 implies a structure consisting mainly of nonhistone proteins which remain after extraction of chromatin, casually lipids and residual DNA from isolated cell nuclei. Morphologically the NM consists of extracted nucleoli, nuclear envelope (lamina) and intranuclear fibrogranular network
found in eukaryotic cells
found in plant cells
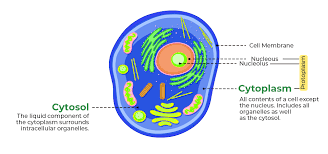
cytosol:
8-11nm diameter
consists mostly of water, dissolved ions, small molecules, and large water-soluble molecules (such as proteins)
found in both prokaryotes and eukaryotes
found in both plant and animal cells
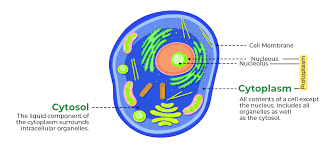
cytoplasm:
semifluid matrix outside nucleus that contains organelles
100–300 mg/mL
composed of water, salts, and various organic molecules
both prokaryotic and eukaryotic cells and functions to house and maintain an optimal environment for the cellular organelles
found in both plant and animal cells
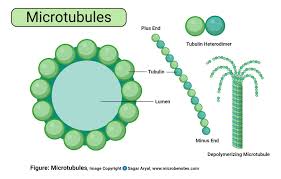
microtubules:
protein cylinders that move organelles
.125 cm thick by 1-125cm long
composed of a single type of globular protein, called tubulin
found throughout the cytoplasm of all eukaryotic cells
found in plant and animal cells
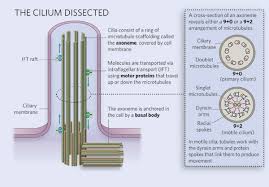
cilia:
move water relative to the cell in a regular movement of the cilia
1.25cm diameter 10-50 cm long
made up of microtubules coated by the plasma membrane
present in eukaryotes
found in most animal cells but only in some plant cells
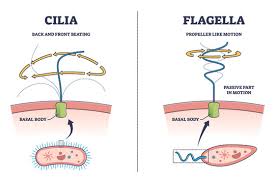
flagella:
help an organism in movement
1.25cm diameter 50-1000 cm long
composed of a single protein, flagellin
exists on both eukaryotic and prokaryotic cells and serves the purpose of moving the cell through the fluid environment in which that cell is found in. However, the structure, composition and even the mechanism by which the flagellum functions in these two different cells differs greatly
most microorganisms and animals, but not in higher plants
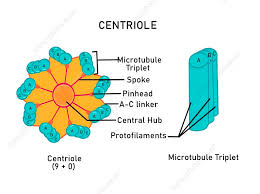
centrioles:
short cylinders of microtubules
.5cm by 2.5cm long
cylindrical structures that are made up of protein tubes called microtubules. Specifically, nine groups of three microtubules, known as triplet microtubules, are linked together to make the walls of the cylinder
Only eukaryotic cells have centrioles. Centrioles make up the centrosome, which is important for organizing spindle fibers during cell division in eukaryotes
Found only in animal cells
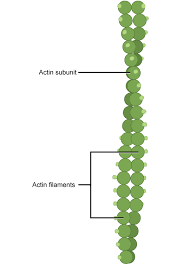
microfilaments/actin filaments:
protein fibers that play a role in cell division and shape
.035cm diameter
Each microfilament is made up of two helical, interlaced strands of subunits. Much like microtubules, actin filaments are polarized
Actin exists in the cytoplasm of eukaryotic cells
actin filaments and microtubules form different cytoskeletal arrays with distinctive functions in plant and animal cells
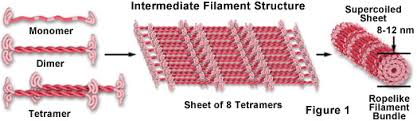
intermediate filaments:
protein fibers that provide stability of shape
.04-.06cm diameter
Intermediate filament proteins contain a central α-helical rod domain of approximately 310 amino acids
found in most eukaryotic cells
Intermediate filaments (IFs) constitute a major structural element of animal cells. They build two distinct systems, one in the nucleus and one in the cytoplasm.
Water Potential:
water potential (ψ): measures the potential for water to leave a cell
diffusion: things other than water moving across a cell membrane
-if things diffuse from a cell membrane, then the concentration of that substance will go from high to low
molarity (M): how much stuff is dissolved in a solution
diffusion doesn’t take energy
diffusion stops when the concentration of both sides of the membrane is equal
osmosis: the diffusion of water
-generally moves from high water potential to low water potential
water can’t have a concentration because it’s the solvent
water potential=pressure potential+solute potential
the pressure potential of a solution in an open container is 0
water potential decreases as solute potential increases (the more solute added, the less chance the substance will have to lose water)
diffusion doesn’t stop, the rates of importing and exporting the substance into the cell become equal
water potential can never be positive
solute potential=-iCRT
the number that you get as the solute potential will be the same as the water potential because the pressure potential will be 0 because there is an open system
isotonic: the amount of solute and the water concentration is the same on both sides of the membrane
the point of doing a water potential calculation is to compare it to something else
adding more solute gives a larger C value and makes the solute potential smaller (more negative) and makes the water potential smaller (more negative)
plant cells can only absorb so much water due to the cell wall
plant cells can build up pressure which can have an effect on the water potential
the cell is in an isotonic solution when the line crosses the X-axis
hypertonic: more solute is dissolved on one side of the membrane than on the other
hypotonic: less solute is dissolved on one side of the membrane than the other
osmosis: water travels from hypotonic to hypertonic
diffusion: solute travels from hypertonic to hypotonic
water can move through the membrane because it’s so small
passive transport: no energy needed to go from high to low concentration (ex: osmosis, diffusion, facilitated diffusion)
facilitated diffusion: when a protein channel is needed to pass through the cell membrane
active transport: energy is needed to move something from low to high concentration (movement)
water potential calculation ex:
- T= 298K
- C=.37M
- solute potential = -(1.0)(.37M)(.0831Lbars/Kmol)(298K) = -9.163 bars
- water potential = solute potential + pressure potential = -9.163 + 0 = -9.163 bars
-if the water potential inside the cell is -9.163 bars, and the water potential outside the cell is -11 bars, then the cell is hypotonic to the outside solution
-water will move out of the cell
Membrane Notes:
membranes move laterally
-kinks in unsaturated fatty acid tails keep plant cell membranes fluid
-cholesterol keeps animal cell membranes fluid
integral proteins go from edge to edge of the membrane
peripheral proteins are attached to the surface of a membrane
bound ribosomes make any proteins used in the cell membrane (integral and peripheral)
transport proteins: (type of integral protein) provide a hydrophilic tunnel for ions specifically for the substances they transport
concentration gradient determines the direction of a molecule that can move freely through the phospholipid bilayer
water can pass through the phospholipid bilayer, but aquaporins are channel proteins that make the process more efficient
water will always move where there is a high concentration of solute
water always moves from hypotonic to hypertonic
facilitated diffusion: passive transport where solutes need a channel protein to pass through the phospholipid bilayer because they are either too big or too polar to fit through the membrane on their own
active transport: pumps molecules across the cell membrane against their concentration gradients, requires ATP
there must be a reason for active transport to take place because otherwise, the cells wouldn’t use their energy on it
secreting a protein takes energy
cotransport: 2 substances can only enter the cell when they’re together
exocytosis and endocytosis are how substances too large for protein channels pass through the cell membrane
endocytosis: the cell forms vesicles to take in substances
phagocytosis: endocytosis of large particulate substances, cell eating
pinocytosis: endocytosis of fluid and dissolved solutes, cell drinking
exocytosis: vesicles fuse with the cell membrane to release substances from the cell
endocytosis and exocytosis are active transport
many channel proteins are only in the secondary phase so the r groups can stick off and create the hydrophobic environment for polar molecules to be able to pass through
nonpolar hydrophobic substances can make it through the cell membrane on their own
Book Chapter 4-5 Notes
bulk transport: molecules too large to be transported by carrier proteins so they are brought in and out of the cell by vesicles
exocytosis: intracellular vesicle fuses with the plasma membrane and secretion occurs
-vesicle membrane becomes a part of the cell membrane because both are nonpolar
endocytosis: cells intake substances by forming vesicles around the material
-a portion of the cell membrane pinches develops the substance and pinches off to form a vesicle
Unit 3
In Class Notes
metablosim: any movement that
making peptide bonds in anabolic, breaking peptide bonds it catabolic
free energy: energy that is available to do something
catabolic reactions are generally exergonic, anabolic reactions are generally endergonic
the energy recieved from the sun is stored as ATP so it can be used elsewhere within the cell
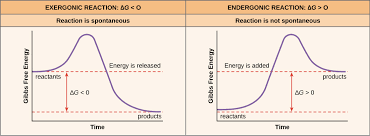
the energy given off suring the exergonic reaction is turned into ATP and then used in endergonic reactions
an enzyme lowers the activation energy needed for a reactoin to take place
enzymes recognize everything by shape
there always has to be an input of energy into the cell otherwise it dies
cells can’t let energy build up otherwise energy will not flow from high to lower concentration inside the cell
genetics determines enzyme shape
adding a phosphate from ATP will change the shape and make it have a function
ATP loses a phosphate and becomes ADP, then goes back to the mitochondria and eventually regains the phosphate and becomes ATP again
ATP to ADP to AMP
AMP is a nucleotide and is used to send messages in the cell
the cell provides more ideal conditions for exergonic reactions than a lab
ATP is the link between the exergonic reaction of respiration and all the other endergonic reactions needed
the more phosphates you have, the more unstable a molecule is
adding a phosphate group changes its shape
cellular respiration: adding another phosphate group on an ADP that had been an ATP and lost a phosphate, this happens in the mitochondria
less energy stored in products than in reactants means the reaction is exergonic
most enzymes end in “ase”
free ribosomes make enzymes that are used inside the cell, bound ribosomes make enzymes that are used outside the cell
eukaryotes have very similar metabolic reactions
catabolic: complex to simple
enzymes are at least at tertiary level proteins (R group interactions)
active site: groove or space on an enzyme that a specific substrate fits into
substrate: the molecule the enzyme is acting upon, fits into the enzyme’s active site
lock and key hypothesis: active sites are made to fit a specific substrate shape, so most enzymes only have one substrate they can act upon
the enzyme makes the orientation of the substrate so ideal that it is easy to break it apart through hydrolysis or synthesize it
induced fit hypothesis: enzyme and substrate somewhat fit together, and the enzyme slightly changes shape to better fit the substrate, implying that enzymes might have more than one substrate they can act upon
we call coenzymes vitamins, and they make enzymes the correct shape
comepetitive inhibition: usually some type of medication or toxin; it blocks the enzyme’s actuive site from the correct substrate to prevent it form functioning; used to control the rate at which an enzyme functions
noncompetitive inhibition: inhibitor binds to a enzyme’s inhbitor site which then changes the shape of the active site and makes the enzyme stop functioning
allosteric inhibitor: type of noncompetive inhibitior, allosteric site is somehwere distant from the active site
alosteric site is usually far away from the active site ad can either be filled by an allosteric activator, or by an allosteric inhibitor that changes the shape of the avctive stie to prevent the enzyme from funcitoning
allosteric inhibition is reffered to as negative feedback because the product that it’s making shuts the process off
allosteric activation: a product makes the active site the correct shape or better than it was before and therefore more efficient; an example of positive feedback
Temperature and pH graphs are bar graphs
Enzymes are usually proteins at tertiary level with R-group attractions
High temperatures denature most enzymes because the protein is vibrating so fast that the R-group attractions break apart
Average healthy body temperatures are determined by optimum temperature for enzyme collisions, which is why a very high fever is dangerous because it will denature proteins
Cold temperatures will reduce the number of substrate-active site collisions, but it doesn’t denature proteins
The enzymes inside the stomach have to be in an acidic environment otherwise they are denatured
Most enzymes are most active around pH 7
Acidic environment: lots of H ions
A lot of R groups have negative charges, because the H ions bond to the R groups which prevents the R groups to bond with each other and causes the protein to denature
Basic environment: lots of OH ions
The OH ions in basic environments are attracted to R-groups with positive charges and prevent R-groups from bonding with each other, which denatures the protein
Allosteric inhibitor: the product goes back and changes the shape of the active site
Non-competitive inhibitors are common toxins
Cell differences are all about what protein the cell is told to make
Proteins are made in ribosomes
DNA makes RNA that tells ribosomes what proteins need to be made
How Are Enzymes Controlled In Living Organisms:
Environment:
-temperature: increased temperature increases reaction rate up to a certain point; increased temperature increases molecular movement which increases substrate-active site collisions, which increases the number of successful collisions
-pH: in the correct pH, the enzyme is turned on
Cofactors: “turn on” an enzyme by making the active site the correct shape
-some cofactors fit into the active site to make it the correct shape, and some join somewhere else to make the active site the correct shape
Inhibitors: “turn off” the enzyme by making the active site the wrong shape
-competitive inhibitor: competes with the substrate to fill the active site
-non-competitive inhibitor: changes the shape of the active site (ex: allosteric inhibitor)
Coenzymes: make the active site the correct shape
Controlling transcription and translation: controlling if the protein/enzyme is produced
chloroplasts are in stacks to increase surface area
green is not used in photosynthesis since it is reflected, so it must be red or blue
plants that live in water utilize blue light because it has the shortest wavelength and therefore the highest energy so they can penetrate the water deeper
most protists are unicellular and can be separated into animal and plant
prokaryotes (bacteria) don’t have mitochondria and don’t have chloroplasts, but protists have both
almost all chloroplasts are in the leaves of plants
chloroplast are usually in the top part of a leaf so they have more direct access to sunlight
plants want to fit as many chloroplasts are possible per cell so be able to fit more chloroplasts that can make more macromolecules
prokaryotes fold their outer membrane to function as a primitive chloroplast ad undergo phtoynthesis
endosymbiotic theory: chloroplaats and mitochondria were most likely one were morst likely pro kaaryotes that were absorbed by another cell to form eukaryotes due to the fact that mitochondria and chloroplast shave their own DNA
granum: a stack of thylakoids
stroma: fluid surrounding granum stacks
the light reaction takes place in the membrane of the thykaloids, while the
photosynthesis reaction: CO2 2 6 12 6 2
Review:
how to control enzyme activity
draw a picture and label the parts of a chloroplast (chloroplast, 1 circular chromosome, ribosomes, thykaloid, granum, and stroma)
stems are on quiz
Cells, Energy, and Metabolism Notes
ATP: Adenosine triphosphate Adenine-ribose-3 phosphate groups that store and transport chemical energy within cells:
-role in the synthesis of nucleic acids
-store the usable energy that plants convert in cellular respiration
-provides the phosphate for the protein
-kinase reactions in signal transduction pathways
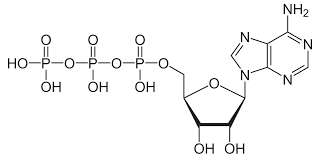
Δ G of ATP into ADP and an inorganic phosphate:
-12 kCal/mole inside of a living cell
-7.3 kCal/mole in laboratory conditions.
-Exergonic
-Hydrolysis reaction (needs water)
Human body cells:
-Need the hydrolysis of 200 to 300 moles of ATP daily.
-Each ATP molecule is recycled 2000 to 3000 times during a single day.
Enzymes:
-Protein that catalyzes (speeds up) a spontaneous chemical reaction.
-Can couple reactions so a thermodynamically favorable reaction can drive an unfavorable one.
-Lowers the activation energy for a reaction -over 5000 known enzymes, most end in “ase”
Active site: very small
-only about 10 amino acids
-where the substrate binds
Lock and Key Hypothesis -Emil Fischer, 1890
Induced Fit Hypothesis -Daniel Koshland, 1958
Enzyme Co-factors
-Some enzymes require the addition of a non-protein part to become active.
Three types:
-Activators (bound ions such as calcium in blood clotting process)
-Coenzymes (non-bound organic molecules that transfer atoms such as NAD)
-Prosthetic groups (bound organic molecules such as the heme group that carries iron in blood)
Inhibition of Enzymes and Reactions:
-Competitive Inhibition
-Noncompetitive Inhibition
Allosteric Enzymes: Enzymes that are regulated by the end products of the reactions that they catalyze.
Active site and an allosteric site (activator or inhibitor)
Why are enzymes important:
-Mutations in the gene for an enzyme can lead to a disease. Example: PKU-phenylketonuria caused by a problem with the enzyme phenylalanine hydroxylase (catalyses the first step in the breakdown of phenylalanine)-build up of amino acid caused mental retardation.
-Many drugs are enzyme inhibitors. Example: Aspirin inhibits an enzyme that produces prostaglandin (causes inflammation) therefore stopping pain/swelling response.
-In everyday products to speed up reactions. Example: Detergents, beer making, dairy/cheese making, rubber and paper making process.
Kinase enzyme that:
-Removes a phosphate group from ATP
-Adds phosphate groups to a protein (phosphorylation)
-Can only attach phosphate to a protein with a free OH group (serine, threonine, or tyrosine)
-Activates the protein
-Over 2% of our genes (500 genes) code for kinases!
Phosphatase enzyme that:
-Removes a phosphate group from a protein
-Inactivates the protein
Photosynthesis Chapter 10 Notes
What things do photosynthesis:
-plants,
-protists: unicellular (euglena) and mutlicellular (algae),
-prokaryotes: cyanobacteria
Chloroplast: organelle found in leaf cells of mesophyll
-30-40 mesophyll per cell
Prokaryotes don’t have have chloroplasts, but interfolding of the plasma membrane function as primitive chloroplasts
Net equation for photosynthesis: 6CO2 +6H2O C6H12O6 + 6O2
-the othe oxygen comes from the water not the carbon dioxide
Stages of photosynthesis:
light reactions:
-thykaloid membrane
-convert solar energy into chemical energy
Calvin cycle/dark reactions:
-stroma
-carbon fixation (C into an organic molecule)
Light reactions:
Pigments needed:
-chlorophyll a (blue-green)
-chlorophyll b (yellow-green)
-carotenoids (yellow and oranges)
-chlorophyll a: required for light reactions, other pigments absorb light and transfer it to chlorophyll a
Light energy (in the form of a photon) is absorbed by pigment
Electron in pigment is boosted to a higher energy level
Photosystems: in thylakoid membrane, includes chlorophyll, other pigments, and proteins
-2 types: photosystems 1 and photosystem 2
transfers energy to reaction center
-reaction center: contains primary electron acceptor
-oxidation-reduction reaction: chlorophyll a loses an electron (oxidized) and electron receptor gains electron (reduced)
Two ways electrons can flow:
-cyclic: uses photosystem 1 only, no NADPH, no O2, makes ATP
-noncyclic: most used method, produces ATP and NADPH (to be used in Calvin Cycle) and O2
What Happens in Light Reactions:
Why 2 methods of electron flow:
-Calvin Cycle uses more ATP (then NADPH)
-cyclic electron flow produces ATP to make up the difference
How is the ATP made: chemiosmosis
-Protons pumped from stroma into thylakoid space (H+ reservoir)
-H+ diffuses from thylakoid space to stroma via ATP synthase complex
-Powers the synthesis of ATP (from ADP and P)
What happens in the Calvin Cycle
Starts with:
-carbon dioxide (from air)
-ATP (from light reactions)
-NADPH (from light reactions)
Ends with:
-3 carbon sugar-G3P
3 phases:
-carbon fixation
-reduction
-regeneration of carbon dioxide acceptor
How is this process different in dry climate plants
Stomata open to let carbon dioxide in, but lose water.
• Dry, hot days the stomata stay closed-no carbon dioxide is being let into the plant
• Photorespiration occurs
– Calvin cycle stops and oxygen builds up (from light reactions)
– Oxygen is used in Calvin cycle instead of CO2
– A two C compound is made in Calvin cycle and is broken down by cell in CO2
C3 Plants (most plants)
– Rice, wheat, beans
– Uses Calvin cycle to make 3 carbon sugar
C4 Plants
– Corn, grasses, sugarcane
– Fixes CO2 into a 4 carbon sugar (PEP) before Calvin cycle
CAM Plants
– Cacti, succulents
– Open stomata at night and close during day
– Opposite of most plants
– Store CO2 as an organic acid
d
Unit 4
In Class Notes:
Bound ribosomes make signal molecules
Ligand: a smaller piece that fits into a larger one
The pituitary gland tells other cells what signals to send out
Apoptosis: programmed cell death
An infected cell is signaled to break open so antibodies can get at a virus or whatever is infected
Ligands usually travel through the bloodstream because they are used outside of the cells they are made in
The signal is only received by cells with the correct receptor protein shape
Ligands are either protein hormones or steroid hormones
Extracellular reception: If ligands are polar they need a receptor protein to be received because they can’t pass through the phospholipid bilayer
Bound ribosomes also make receptor proteins
Once the logan is received, the receptor protein changes shape to give the cell interior a sign that the signal has entered the cell
A protein hormone reception usually occurs quickly but is short-lived
A protein hormone can result in multiple different responses
Intracellular reception: Steroid hormone ligands are usually nonpolar and can pass through the cell membrane without assistance
Intracellular reception is usually slow but long-lasting
Steroid hormone ligands were mostlikely made by smoot ER
Intracellular reception is almost always to tell the cell to turn on a gene to make a new protein
Steps for extracellular reception: reception, transduction, response
Kinase (enzyme): takes a phosphate from ATP and adds it to a protein to activate it
Amplification: many proteins can be turned on by one (or few) reception(s)
Some of the ATP made during respiration is used during protein reception
The signal that happens for growth and repair happens during sleep
Review:
What is a kinase: know that it takes a phosphate from ATP and adds it to protein
What is a cell’s response to an extracellular reception: a
The main focus is long-distance cell communication
What happens when there is an error during cell signaling
list the stages of the cell cycle in order and briefly summarize what is happening in each stage
explain what conditions might cause a cell to halt the cell cycle and state briefly where in the cycle this would occur
discuss how apoptosis represents a regulatory event of the cell cycle
list the stages of interphase and describe the major events that occur during each stage in preparation for cell division
list the checkpoints that regulate the progression of cells through the cell cycle
explain the mechanisms within the G1 cell cycle checkpoint that evaluates growth signals, determines nutrient availability, and assesses DNA integrity
what is the potential effect of an abnormally high level of growth hormone n the regulation of the cell cycle
why might some cancers be associated with a mutation in the gene encding the p53 protein
explain how DNA becomessufficiently compacted to fit inside a nucleus
distinguish between euchromatin and heterochromatin
list the stages of chromosome compacting, starting with a singl DNA strand
explain how the cell prepares the chromosomes and centrosomes prior to nuclear diivsion
summarize the major evens that occur during mitotsis and cytokinesis
discuss whyh human ste cell continuously conduct mitosis
dummarize the differences bteween cytokinesis in animal and plant cell nd explain why the differences are necessary
discuss the importance of stemm cells in the human ody
ddescribe the basic characteristics of cancer ccells
explain the differene betweeen a benign and malignant tumor
distinguishbetweenthe roles f the tumor suppresor genes and proto-oncogenes in the rergulation of the cell cycle
compar and contrast the effect on the cell cycle of a mutation in a proto-oncogene and a mutation in a tumor suppressor gene
Cell Communication Slides:
Short distance: local regulators: paracrine signaling:
-secreting cell ratgets nearby cells through the extracellular release
-synaptic signaling: nerve cell releases neurotransmitter into the synapse
-takes place when damaged cells send out a signal to the other cells nearby to either fix or replace it
Long distance hormones: hormone signaling, hormones released into bloodstream for targeted cell (cell with the correct receptor protein)
Most signals sent out in plant cells are in the form of a gas that can be sent out to nearby leaves
Cell junctions: cells joining together so they can get signals to each other more quickly
-gap junctions for plant cells
-plasmodesmata for animal cells
Signal transduction pathway: reception, transduction, response
Ligand: a small molecule that binds to a larger one
-the binding causes a receptor protein to chain conformation (shape)
The receptor protein changing shape or a steroid hormone being accepted intracellularly is what begins the cascade of event
Intracellular receptors: non-polar molecules, receptors found in cytoplasm, enters the nucleus to turn a gene on or off (make a new protein)
Kinases add a phosphate from ATP to a different protein to activate it
Protein kinases regulate most cellular activity
Abnormal protein kinase activity can contribute to cancer
Protein phosphatases: enzymes that turn off a signal pathway by removing phosphate groups from activated proteins
Battle between kinases and phosphatases: kinases are turned on when the ligand is accepted, when the receptor protein changes shape phosphatases are turned on
Second messenger: small non-protein polar molecules or ions
-Ca+2 and cAMP
Its very common to store Ca in the smoooth ER
all cells have chromosomes
Hardcover Book Notes (pg 147-160):
Cell cycle: an orderly set of stages that takes place between the time a eukaryotic cell divides and the time the resulting daughter cells also divide
When a cell is going to divide, it grows larger, the number of organelles doubles, and the amount of DNA doubles as DNA replication occurs
2 portions of the cell cycle are interphase and the mitotic stage where mitosis and cytokinesis occur
Interphase:
Most of the cell cycle is spent in interphase, where the cell performs its usual functions (depending on its location in the body)
The amount of time a cell takes for interphase varies widely
Interphase consists of 3 stages: G1, S, and G2
“G” stands for growth
G1 Stage:
G1is the stage where the cell recovers from the previous division: the cell grows in size, increases the number of organelles (such as mitochondria and ribosomes), and accumulates material that will be used for DNA synthesis
Cells are constantly performing their normal daily functions during this stage, including communicating with other cells, secreting substances, and carrying out cellular respiration
Some cells like nerve and muscle cells exit interphase during G1 and enter a phase called G0
When cells are in G0 they continue to perform normal, everyday functions but no preparations are being made for cell division
Cells can’t leave G0 until they have received a signal from other cells or other parts of the body to do so
The completion of the cell cycle is very tightly monitored
S Stage:
DNA synthesis (replication) occurs in S stage
At the beginning of the S stage, each chromosome is composed of one DNA double helix
Following DNA replication, each chromosome is composed of 2 identical DNA double-helix molecules
Each double helix is called a chromatid, and the 2 identical chromatids are referred to as sister chromatids
The sister chromatids remain attached until they are separated during mitosis
G2 Stage:
G2 is the stage after DNA replication
G2 is the stage from the completion of DNA synthesis to the onset of mitosis
The cell synthesizes proteins that will assist in cell division during this stage
This is the stage where microtubules are made
Microtubules are used during the miotic stage to form the mitotic spindle which is critical during the mitotic stage
M (Mitotic) Stage:
The mitotic stage includes mitosis (nuclear division) and cytokinesis (division of the cytoplasm)
During mitosis, daughter chromosomes are distributed by the mitotic spindle to 2 daughter nuclei
When cytokinesis is complete, 2 daughter cells are present
Control of the Cell Cycle:
Signal: an agent that influences the activities of a cell
Growth factors: signaling proteins received at the plasma membrane
Even cells arrested in G0 will finish the cell cycle if stimulated to do so by growth factors
In general, signals ensure that the cell cycle stages follow one another in the normal sequence
Cell Cycle Checkpoints:
The cell must pass through 3 main checkpoints before it proceeds to the next stage of the cell cycle
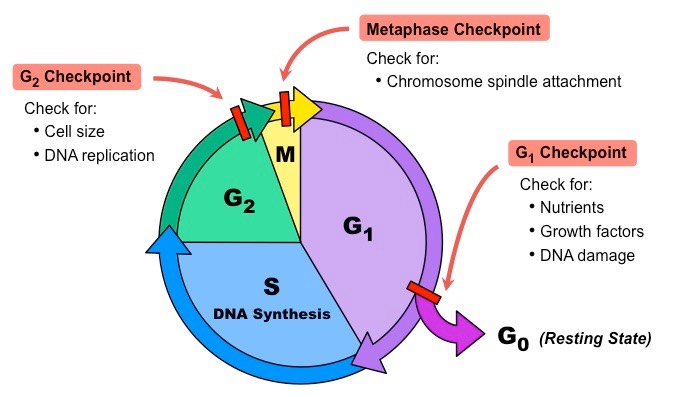
the cell either stops at the checkpoint or continues to the next phase of the cell cycle, depending on the internal signals received
cyclins: a family of internal signaling proteins that increase and decrease as the cell cycle continues
specific cyclins must be present for the cell to proceed from G1 to S and from G2 to M
the G1 checkpoint is the primary checkpoint of the cell cycle
in mammalian cells, signaling protein p53 stops the cycle at the G1 checkpoint when DNA is damaged
first p53 tries to repair the DNA, but rising levels of p53 can trigger apoptosis
apoptosis: programmed cell death
the cell cycle may also stop at the G2 checkpoint if DNA has not finished replicating
the G2 checkpoint prevents the initiation of the M stage before the completion of the S stage
the G2 checkpoint also offers the opportunity for DNA to be repaired
the M checkpoint stops the cell cycle if the chromosomes are not properly attached to the mitotic spindle
normally, the mitotic spindle ensures that the chromosomes are distributed accurately to the daughter cells
Apoptosis:
during apoptosis, the cell rounds up, causing it to lose contact with its neighbors
the nucleus fragments and the plasma membrane develops blisters
finally, the cell fragments are engulfed by white blood cells and/or neighboring cells
caspases: the enzymes that bring about apoptosis
caspases are always present in the cell and are held back by inhibitors until either internal or external signals unleash them
Apoptosis and Cell Division:
in living systems, opposing events keep the body in balance and maintain homeostasis (ex: cell division and apoptosis)
somatic cells: body cells
apoptosis prevents a tumor from developing and helps limit the spread of viruses
The G1 Checkpoint:
the G1 checkpoint ensures that conditions are right before committing to divide by evaluating the meaning of growth signals, determining the availability of nutrients, and assessing the integrity of DNA
failure to meet any of these criteria results in the cell entering G,0, or undergoing apoptosis if problems are severe
Evaluating Growth Signals:
signaling molecules such as hormones may be sent from nearby cells or distant tissues to encourage or discourage cells from entering the cell cycle
suchsignals may tell a cell to enter G0 o comlete G1 and enter the S stage
groth signals that promote cell division cause a cyclin-dependent-kinase (CDK) to add a phosphate group to the RB protein, a major regulator of the G1 checkpoint
usualy and EF2 protein is bound to RB, but hen RB is phosphhorylated, it’s shap changes and releases EF2
EF2 then beinds to DNA, actuvating certan genes whose products are needed to complete the cell cycle
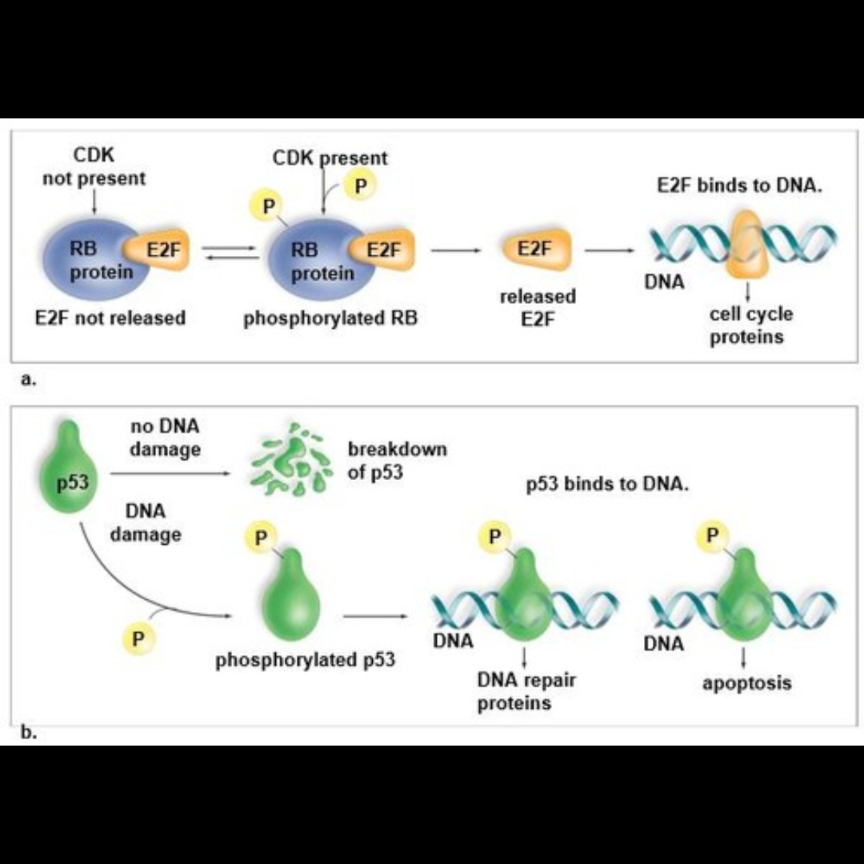
growth signals promote cells that are in G0 to reenter the G1 stage, complete it, and enter the S stage
if growth signals are sufficient, the cell passes through the G1 checckpoint and cell division occurs
Determining Nutrient Availability:
a cel ensures that nutrientlevelsare sufficient before commtting to cell division
starving cells in culture enter G0because of this
at that time, phosphate grops are reoved from RB (see revers arrows on part a. in the miage avoe) o RB doesnt release E2F and the proteins needed to complete the cell cycle aren;t produced
when nutrietnts become avaiabe, CDKs bring aobut the phosphorylation o RB ,which thne releases E2F (see forward arrows on parta. in the image above)
after E2F brind to DNa, the proteins needed to compete the ell cycle are prodced
Assessing DNA Integrity:
for cell division to occur, DNA must be free of errors and damage
the p53 protein is involved in this quality control funcion
usually p53 is broken down because it has no job
in response to damage, CDk phosphorylates p53 (see part b on image above) which prevents the breakdown of p53 and instead triggers a rise in p53 levels in the nucleus
phosphorylated p53 bids to DNA, gertain genes are activiates, and DNA repair protens are produced
if th eDNA damage can’t be repaired, p53 levels continue to rise and apoptosisis trigered
if the repair is successful ,p53 levels fall and the cell is allowed to complete G1 as long as growth signals and nutrients are present
many criteria must be met for a cell to commit to cecll divsion,and the failure to meet any one of them my cause the cll to be halted and.or apoptosis to be initiated'
a better understanding of the G1 checkpoint may lead to curing cancer
The Eukaryotic Chromosome
a eukaryotic chromosome contains a single double-helix DNA molecule, but it is composed of more than 50% protein
some of these proteins are concerned wth DNA and RNA synthesis, but a large majroity of these proteins (histones) play a primariy structural role
5 primary types of histones: H1 H2A, H2B, H3, and H4
the amino acid sequences of H3 and H4 vary little between organisms, suggesting that few mutations in the histone proteins have occurred during the course of evolution and that histones have essential functions for survival
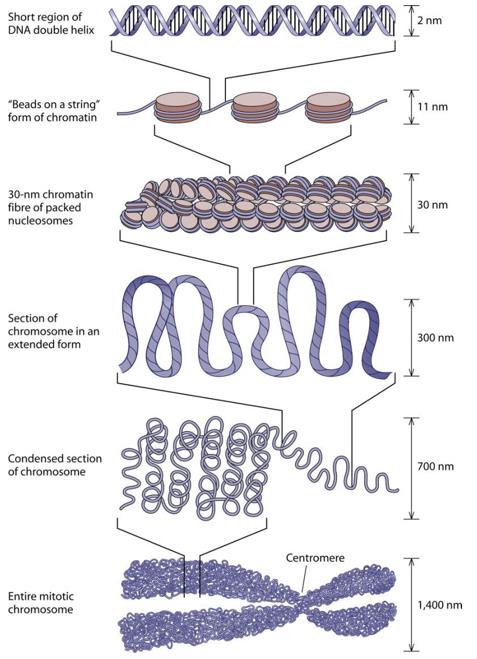
a human cell contains atleast 2m of DNA, yet all of the DNA is packed into a nucleus that is about 6 micrometers in diameter
histones are responsible for packaging the DNA so it will fit into such a small space
first, the DNA double helix is wound at intervals around a core of eight histone molecules (two copies each of H2A, H2B, H3, and H4) giving the appearance of a string of beads

each “bead” is a nucleosome, and the nucleosomes are joined together by linker DNA
the string is compacted by folding into a zigzag structure, further shortening the DNA strand
histone H1 appears to bring about this coiling process
the fiber then loops back and forth into radial loops
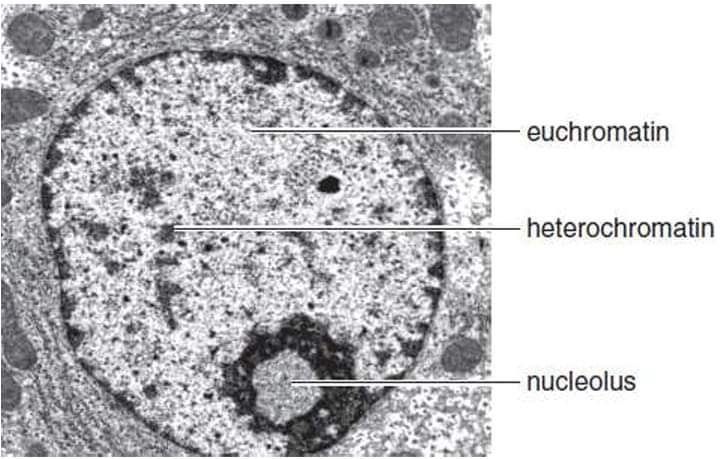
this loosely coiled euchromatin represents the active chromatin containing genes that are being transcribed
the DNA of euchromatin may be acessed by RNA polymerase and other factorsthat are neededto promote transcription
researc indicate that regultin g the level of compaction of the DNA is an importnt method of controling gene expression in the cell
heterochromatin: more highly compacted form of the chromsome
most chromosomes exhibit both levels of compaction in a living cell, depending on which portions of the chromosomes are being used more frequently
heterochromatin is considered inactive chromatin because the genes contained on it are infrequently transcribed, if at all
prior to cell division, a protein scaffold helps further condense the chromosome into a form that is characteristic of metaphase chromosomes because compact chromosomes are easier to move about than extended chromatin
during mitosis, the sister chromatids are separated and distributed to 2 daugther cells
Chromosome Number
when a eukaryotic cell is not ndergoing cell division, the DNA and associated protein are located withing chromatin
chromatin has the appearance of a tangled mass of thin threads
before mitosis begins, chromatin becomes highly coiled and condensed, and it is easy to see the individual chromosomes
each species has a characteristic chromosome number
diploid (2n): the full number of chromosomes found in all cells of the individual
most somatic animal cells are diploid)
haploid(n): half the diploid number, contains only one chromosome of each kind
the gametes of animals (eggs and sperm) are haploid cells
Preparations for Mitosis:
during interphase a cell replicates chromosomes and duplicates most other organelles, including the centrosome
the centrosome organizes the spindle apparatus necessary for the movement of chromosomes
during mitosis, a 2n (diploid) nucleus divides to produce daughter nuclei that are also 2n
by the end og the S phase DNA has replicated and each chromosomehas 2 identicaldouble helical molecules
each odube helix is a chromatid
sister chromatids are constricted and attached to each other at the centromere
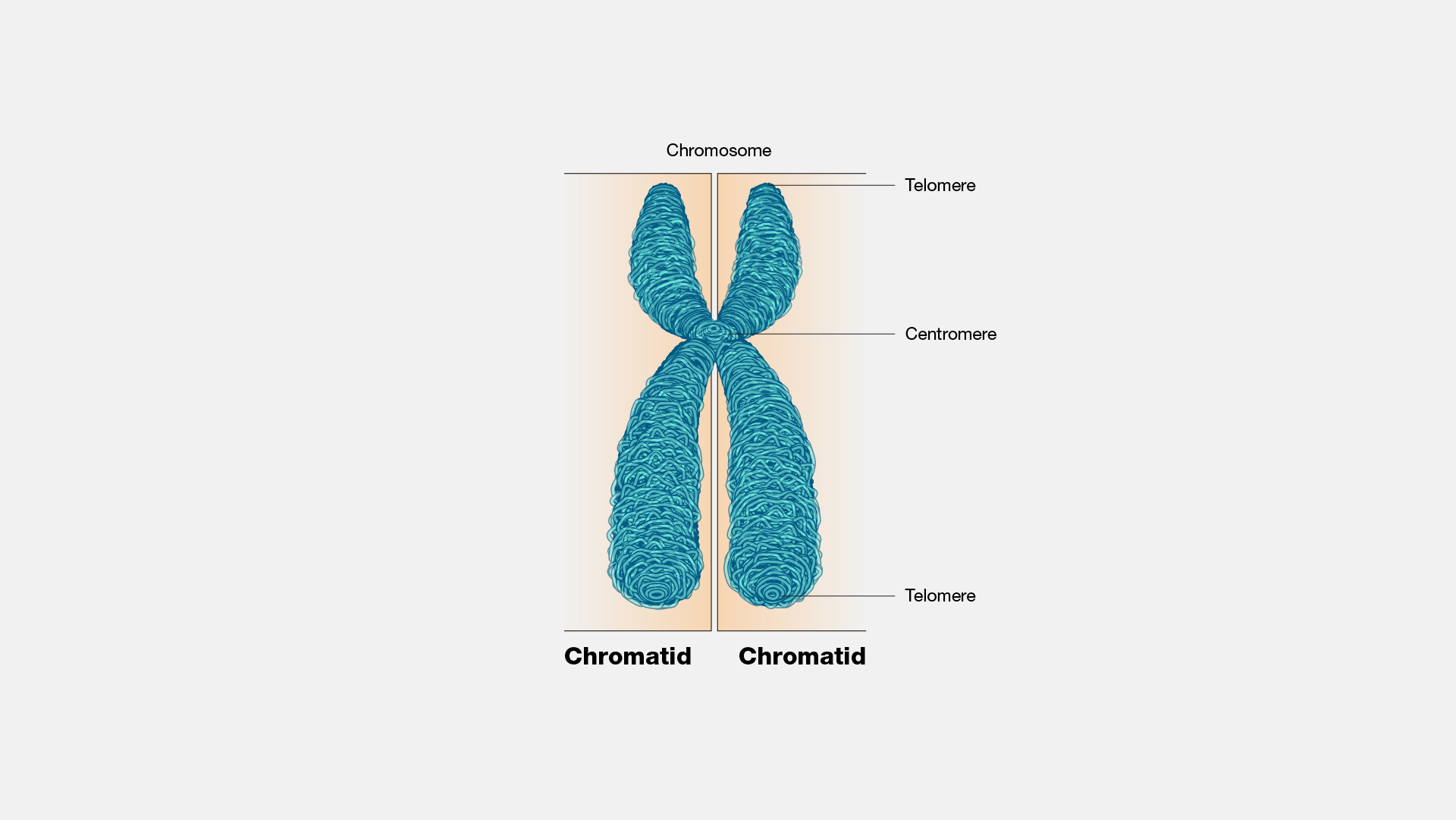
kinetochores: protein complexes that develop on either side of the centromere during cell division
the 2 sister chromosomes separate at the centromere during nuclear division
each daughter chromosome has only one double-helix molecule
an equal amount of daughter chromatids are distributed to each daughter cell so each daughter nucleus gets a copy of each chromosome that was in the parent cell
Division of the Centrosome:
centrosome: the main microtubule-organizing center of the cell, divides before mitosis begins
each centrosome in an animal contains a pair of barrel-shaped organelles called centrioles
centrioles aren’t in plant cells
centrosomes organize the mitotic spindle, which contains many fibers made of bundles of microtubules
microtubules: hollow cylinders made of tubulin protein
microtubulin assembles when tubulin subunits join, and when they disassemble tubulin subunits become free again
microtubules of the cytoskeleton disassemble when spindle fibers begin forming
Phases of Mitosis:
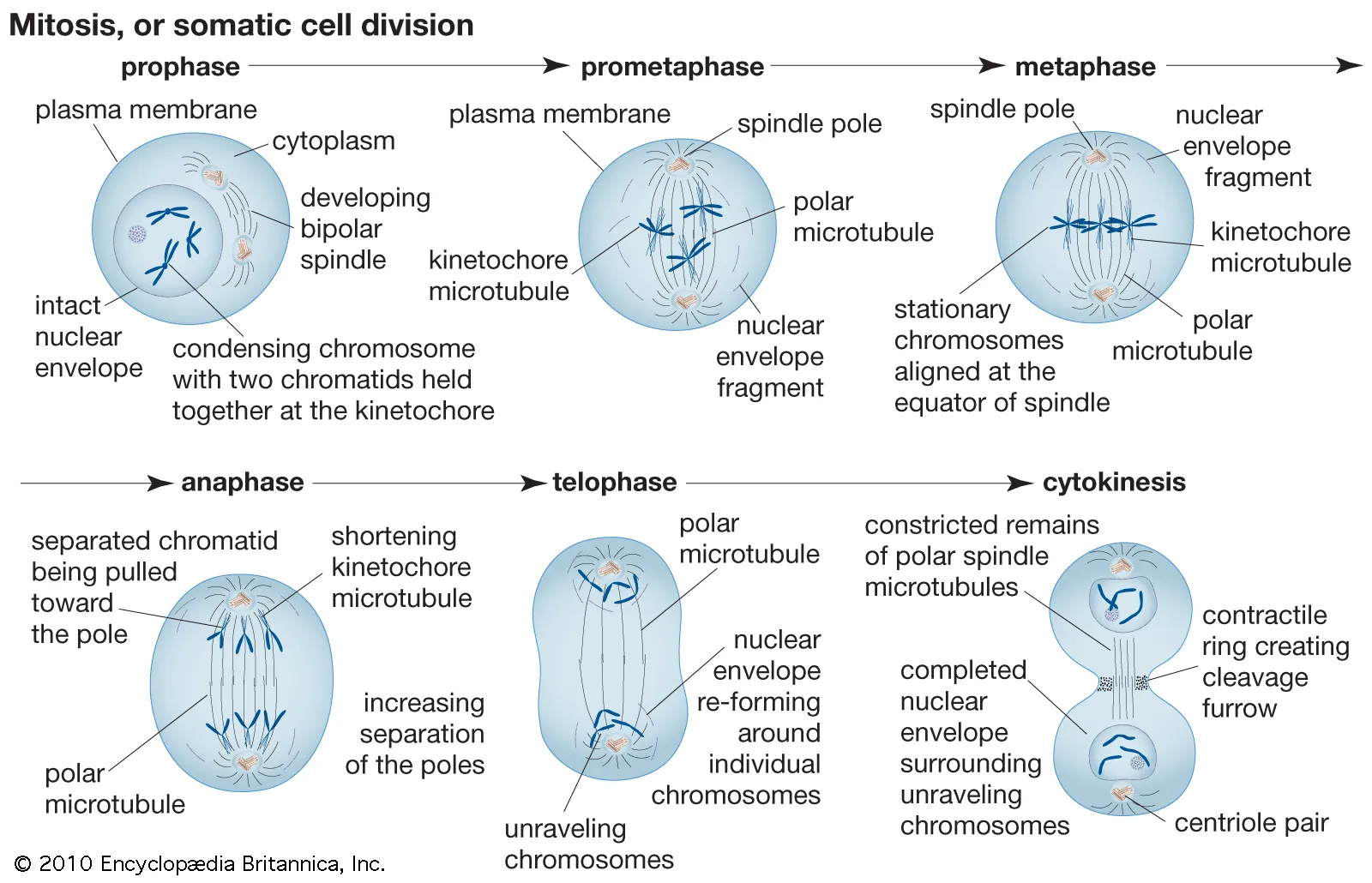
Prophase
prophase is when it becomes apparent that mitosis is going to occur because chromatin has condensed and the chromosomes are visible
DNA replication already occurred in interphase so parental chromosomes are already duplicated and composed of 2 sister chromatids held together at a centromere
counting the number of centromeres in a diagram gives the number of chromosomes for the cell depicted
the nucleolus disappears and the nuclear envelope fragments during prophase
the spindle begins to assemble as the 2 centrosomes migrate away from each other
in animal cells a structure of microtubules called asters radiates toward the plasma membrane from the centrosomes
asters brace the centrioles during the later stages of cell division
the chromosomes have no particular orientation in prophase because the spindle has not yet formed
Metaphase:
the centromeres of chromosomes are aligned in a single plane at the center of the cell
metaphase plate: the imaginary line that passes through the aligned chromosomes and indicates where the cell will split during the later stages of mitosis
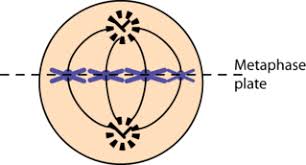
polar spindle fibers: sever nonattached spindle fibers that reach beyond the metaphase plate and overlap
the M checkpoint delays the start of anaphase until the kinetochores of each chromosome are attached properly to spindle fibers and the chromosomes are properly aligned along the metaphase plate
Anaphase:
at he start of aaphase the two sister chromaties o each duplicated chromosome separate at the centromere, giving rise to two daughter chromosomes
daughter chromosomes each have a centromere and a singe chromaid composed of a single double helix
the daughter chromosomes are pulled towards opposite poles of the cellas the kinetochore spindle fibers disassemble at the region of the kinetochores
s the daughter chromosomes move towards the spindle poles, the poles themseevs are sliding apart bease the polar spinde fibers are fliding past one another
microtubule-associated proteins such as motor molecules kinesin and dynein are involved in the sliding process
anaphase is the shortest phase of mitosis
Telophase:
the spindle disappears as new nuclear envelopes form around the daughter chromosomes
each daughter nucleus contains the same number and kinds of chromosomes as the original parent cell
remnants of the polar spindle fibers are still visible between the two nuclei
the chromosomes become the less compact chromatin again and a nucleolus appears in each daughter nucleus
Cytokinesis in Animal and Plant Cells:
cytokinesis: division ofthecytoplasm
cytokinesis accompanies mitosis is most cells but not all
if mitosis occurs without cytokinesis, the result is multi-nucleated cell
ctokinesis begins in anaphase but doesn’t reach completion until the beginning of the next interphase
by the end of mitotis each daughter cell has recieved a share of the cytoplasmic oganelles that were duplicted during interphase
cytokinesis proceeds differently in plantand anima cell s because of differences in cell structure
Cytokinesis in Animal Cells:
cleavage furrow: an indentation of the membrane between the two daughter nuclei that forms in animal cells as anaphase ends
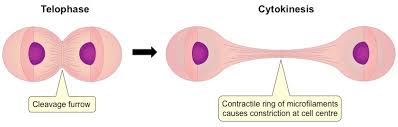
by the time the cleavage furrow forms, each daughter cell has recieved its share of duplicated cytoplasmic organelles
the cleavage furrow deepens when a band of actin filaments called the contractile ring slowly forms a circular constriction between the 2 daughter cells
after telophase the contractile ring tightens more and completely separates the cytoplasm, creating 2 daughter cells
Cytokinesis in Plant Cells:
the cell wall in plants is too rigid to allow cytokinesis by furrowing
cytokinesis in plant cells involves building new cell walls between the daughter cells
cytokinesis is apparent when a small flattened disk appears between the 2 daughter plant cells near the site where the metaphase plate once was
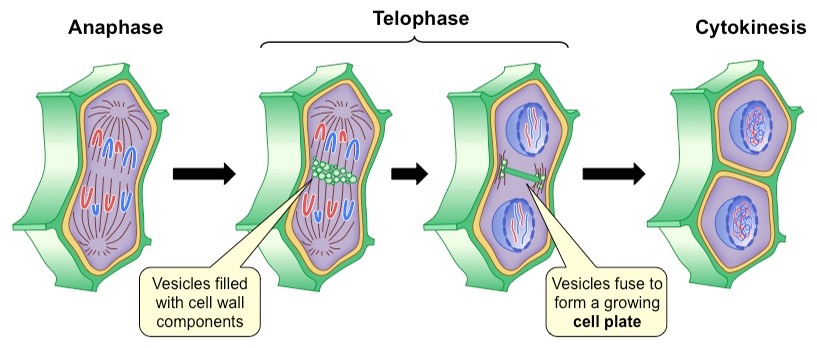
the disk is at right angles to a set of microtubules that radiate outward from the forming nuclei
the Gogi apparatus produces vesicles that move along the microtubules to the region of the disk
as ore vesicles arrive and fuse, a cell plate becomes visible
cel plate: a newly formed plasma membrane that expands outward until it reaches the old plasma membrane and then fuses with the old membrane
thenew membrane releases molecules that form new plat cell walls
the alls (known as primary cell wals) are later strengtheed bythe ddition of celulose firbils
the space between the daughter cells becomes filled with middle lamella, which cements the primary cell walls together
The Functions of Mitosis:
mitosis permits growth and repair
in flowering plants meristematic tissue retains the ability to divide throughout the life of the plant; meristematic tissue at the shoot tip is what allows for a flowering plant to keep growing taller as long as it lives
lateral meristem accounts for the ability of trees to increase their width each growing season
Stem Cells:
most cells in adulthood are permanently arrested in G0
many mammilian organs contain stem cells (often called adult stem cells) that retain the ability to divide
theraputic cloning: used to produce human tissues; can begin with either adult stem cells or embryonic stem cells
embryonic stem cells can also be used for reproductive cloning; the production of a new individual
The Cell Cycle and Cancer:
cancer: a cellular rwoth disorder that occurs when cells divide uncontrollably
most cancers are the result of accumulating mtations that ultimately cause a lss of control of the cell cycle
most cancers begin as an abnormal cell growth that is benign (not cancerous)
when additional mutations occur the abnormal benign eclls fail to respond to nhibitin g signals that control the cell cycle and become malignant (cancerous and possessing the ability to spread)
Characteristics of Cancer Cells:
the deveopment of cancer is gradual
a utation in a cell may cause it to ecome preancerous, bt many other regulatory processess within the body prevent it from becoming cancerous
cancers vary greatly, but cells that possess the following haracteristics are generally seen as canceous:
-cells lacking differentiation: cancer cells aren’t specialized and don’t contribute to the functioning of a tissue. although cancer cells may still posses many of the chracteristics of surrounding normal cells, they sually look distinctly abnormal nrmal cells can enter the cell cycle about 50 times before the are incapable of dividing again. cancer ells can center the cell cycle an infinite number of times and seem almost immortal
-abnormal nuclei: the nuclei of cancer cells are enlarged and may contain an abnormal amount of chromosomes. extra copies of one or more chromosomes may be present.oftern there are duplicatedportions of chroosomes present, resulting in gene amplification (extra copies of specfic genes). some chromosomes may also possess deleted portions
-no apoptosis: usually cells with dmaged DNA undergo apoptosis because the immune system recognizes them as abnormal an can prevent tumors from forming. cancer cells fail to undergo apoptosis een though they are abnormal cells
-formation of tumors: normal cells anchor themselves to a substratum an/or adhhere to neighbr cells. they ehibit contact inhibition (when the come i contact with other cells, they stop divising). cancer cells dont exhibit contact inhibition and the abnormal cancer cells pile ontop of each other, grwing in multiple layers and forming a tumor. during carcinogenesis, the most aggressive cell becomes the dominant cell of the tumor
-cancer cells undergo metastasis and angiogenesis: additional mutations may cause a beneign tumor, which is usually contained within a capsule and cn’t invade adjacent tissue,tpbecome malignant and spread thrughout the body, forming ew tumors distant from theprimary one. these cells now produce enzymes that they don’t normally express, allowing tumor cells to invade underlyng tisues. then they travel through the blood and lymph to start stumors elsewhere inthe body. this process s known as metastasis.
the blood vessels supplying nutrients to the tumor cells become insufficient to supportthe rapd grwth of an actively growing tumor, and additional mtations occuring in the tumor cells allow the formationof new blood vessels flowing into the tumor
angiogenesis: additional mutations in tumor cels that alow for the formation og new bood vessels flowing directly into the tumor
Origin of Cancer:
cancer is usually caused by mutations affecting genes that directly or indirectly affect the balance of promotion and inhibition of cell division like proto-oncogenes or tumor suppressor genes
proto-oncogenes: genes that code for proteins that promote the cell cycle and prevent apoptosis, cause the cell cycle to speed up
tumor supressor genes: genes that code for proteins that inhibit the cell cycle and promote apoptosis, they cause the cell cycle to go more slowly or to stop completely
Proto-Oncogenes Become Oncogenes:
proto-oncogenes are often at the end of a stimulatory pathway extending frfom the plasma membrane to the nucleus
a stimulus such as an injury results in he reease of a growth factor that bnds to a receptor protein in the plasma membrnae, which sets in motion a series of enzymatic reactions leading to the activation of genes that promote the cell cycle both direcly and indirectly
proto-oncogees indluce the receptors and signal moecules that make up thesepathways
when mutatinos occur in proto-ncogenes, they become oncogenes (cacer casing genes)
oncogenes are under constant stimultion and keep on promoting the cell cycle regardess of circumstances (ex: an oncogene mght code for a faulty receptor i n the stimulatory pathway such that the cell cycle is stimulated even when no growth factor is present)
Tumor Suppressor Genes Become Inactive:
it is timated that over half of human cancers involved an abnormal or deletd p53 gene (see G1 checkpoin)
Other Causes of Cancer:
a mutation affecting the cell’s DNA repair system leads to much higher chance of a mmutation ccurring ithin a proto-oncogene or a tumr suppressor gene
in some cancer cells, mutation of telomerase enzyme that regulates the length of telomeres (ends of chromosomes) auses the telomeres to remain at a constannt length, allowing the cacer cells to continue dividing repeatedly
Semester 1 Exam
Unit 1:
water is a polar molecule
hydrogen bonds form between spatially negative oxygen ends of a waste molecule and a partially positive enhydrogen end of a different water mnolecule
hydrogen bonds (intermolecular) are much weaker than ionic/covalent bonds (intra molecular)
hydrogen bonds are no just in water
cohesion: hydrogen bonds between water molecules
-responsible for water’s: high heat of vaporization, high specific heat, high surface tension
adhesion: hydrogen bonds between water and other polar molecules
surface tension: force exerted by the water molecules on the+ surface of a body of water that creates a kind of web or net upontthe surface
acidic solutions: excess of H+ (pH0-pH7)
basic solution: excess of OH- (pH7-pH14)
carbon: the central element that makes up living things
H: often used in energy exchange (NAD= vs NADH), H= is used to create energy gradients, basis of acidity and alkalinity
P: in phosphate groups (which are in ATP)
monomers: small building blocks that build carbohydrates, proteins, and nucleic acids
polymers: combined monomers that allow living thins to build macromolecules with 3-D shapes and functions
monomers are like lego bricks, polymers are like a finished lego set
dehydration synthesis allows monomers to come together and build polymers
dehydration synthesis: enzymes pull a hydroxyl (OH) off of one monomer (or a group of already connected monomers) and an H off of another monomer (or group of monomers); resulting in the formation of a polymer and a water molecule
hydrolysis: enzymes insert a water molecule between 2 monomers (that are connected in a polymer) to break them apart
phosphate groups: key for energy exchange
hydroxyl group: makes a molecule hydrophilic
carbonyl group: makes a molecule hydrophilic
carboxyl group: essential in amino acids
amino group: essential in amino acids
sulfhydryl group: important in protein structure
carbohydrates include monosaccharides, disaccharides, and polysaccharides
monosaccharides: energy sources and building blocks (ex: simple sugars)
disaccharides: energy transfer, composed of 2 linked monosaccharides (ex: sucrose, lactose)
polysaccharides: energy storage (ex: starch, glycogen), cell structure (ex: cellulose), composed of multiple monosaccharides
most animals can’t hydrolyze the bonds that connect the glucose monomers in cellulose, but some animals (termites and ruminants) evolved symbiotic relationships with microorganisms that can hydrolyze cellulose into glucose
lipids: wholly or partly nonpolar, not composed of repeating monomers, used for energy storage (fats, oils), waterproofing (waxes), membrane formation (phospholipids), signaling (steroids)
phospholipids have a hydrophobic fatty acid tail connected to a hydrophilic phosphate head via a glycerol molecule
the monomer version of a protein is an amino acid
amino acids: contains a central carbon with a connected amine group (basic) and a connected carboxyl group (acidic) on the other side, an H atom is also connected to the central carbon as well as a variable R group
all proteins are built from the same 20 amino acids
primary structure of proteins: linear sequence of amino acids
secondary structure of proteins: interactions involving polypeptide backbone (alpha helixes and beta pleated sheets)
tertiary structure: interaction between R groups
quaternary structure: interactions between multiple folded tertiary peptides
polypeptide: multiple amino acids linked together
amino acids are connected by peptide bonds (N-C-C)
secondary protein structure: involves interactions (formation of H bonds) between carbonyl groups and amino groups in the polypeptide bbackbone creatin alpa helixes or beta pleated sheets
tertiary protein structure: interactions (H bonds, ionic bonds, covalent bonds, hyrdophoic clustering) bteween amino acid side chains (R grousp)
hydrophobic clustering: nonpolar side chains cluster together avoiding water
quaternery protein sturctue ;involves multiple polypeptides that interact (h bonds, ionic bonds, hydrophobicinteractions) with one another to create the final form of the protein
nucleic acids: molecules of genetic information, includes DNA, RNA, and ATP
DNA: molecule of heridit
RNA: key role is infromation transfer, not a double helix so it can take many forms, can also act as an enzyme, catalyzing reactions (seen in ribosomes)
ATP: nucleotide and RNA monomer, life’s key energy transfer molecle, powering most cellular work
nucleotide: nucleic acid monomer, contains a 5-carbon sugar, a phosphate group (connected to #5 carbon), and one of 4 nitrogenous bases (connected to #1 carbon)
the sugar in DNA is deoxyribose and the bases are adenine, thymine, cytosine, and guanine
the sugar in RNA is ribose and the bases are adenine, uracil, cytosine, and guanine
DNA consists of 2 nucleotide strands that connect to each other via H bonds between nitrogenous bases with complimentary shapes (A-T and G-C)
within each strand the nucleotides connect to one naother by sugar-phosphate bonds
nuclear function: stores and protected genetic info/DNA
DNA is wrapped around proteins fto form chromosomes with sister chromatids during meiosis or mitosis, otherwise DNA is found in the from of chromatin which is more spread out
nucleolus: assembles ribosomes
nuclear membrane: separates chromosomes from cytoplasm
nuclear pore: allows molecules to enter/leave the nucleus (messenger RNA, and transcp)
robsomes:
Unit 5:
Meiosis Note Sheet
Vocab:
gene:
gametes:
somatic cells:
locus:
asexual reproduction:
clone:
sexual reproduction:
homologous chromosomes:
female genotype
male genotype:
sex chromosomes:
autosomes:
diploid (2n) cell: refers to the total number of chromosomes, which exists in 2 sets
haploid (n) cell: half of the diploid number, or a single set of chromosomes
zygote:
meiosis: the type of nuclear division that reduces the chromosome number from the diploid (2n) number to the haploid (n) number (in humans, meiosis reduces the diploid number of 46 chromosomes to the haploid number of 23 chromosomes)
Stages of Meiosis:
khkbjhb
Compare and CONtrast Mitosis and Meisosis:
jhvjhvjhvjh
How does meiosis contribute to genetic variation: