2.Classification
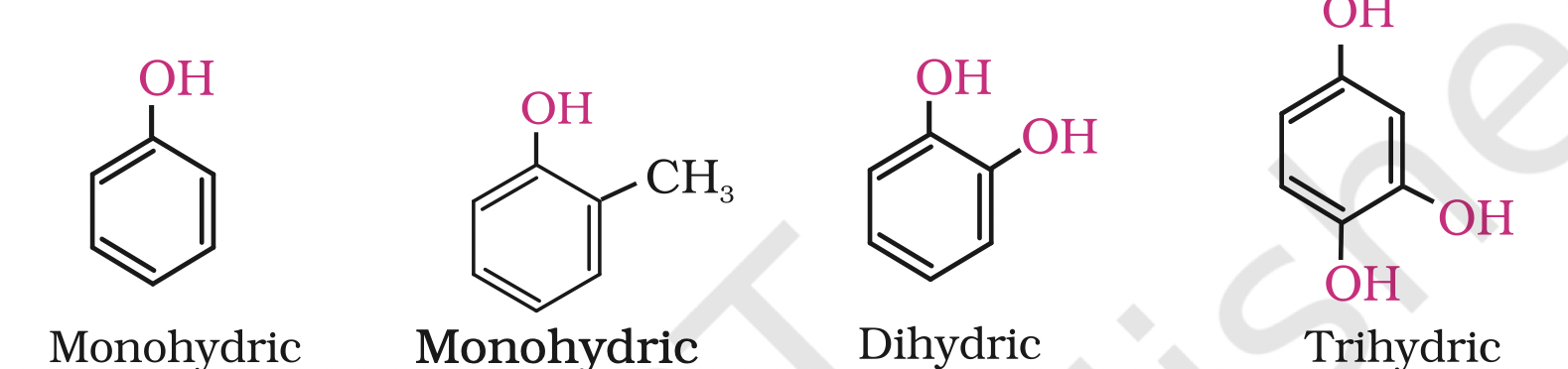
Alcohols and phenols may be classified as mono–, di–, tri- or polyhydric compounds depending on whether they contain one, two, three or many hydroxyl groups respectively in their structures as given below
Monohydric alcohols may be further classified according to the hybridisation of the carbon atom to which the hydroxyl group is attached.
(i) Compounds containing C sp3 - OH bond: In this class of alcohols, sp
the –OH group is attached to an sp3 hybridised carbon atom of an alkyl group. They are further classified as follows:
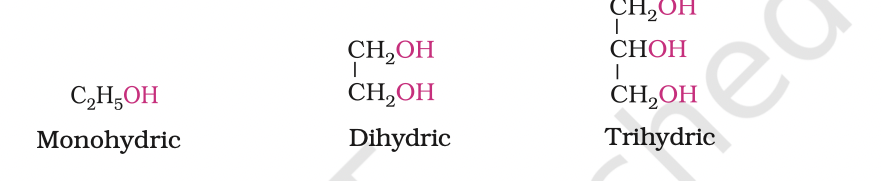
Primary, secondary and tertiary alcohols:
In these three types of alcohols, the –OH group is attached to primary, secondary and tertiary carbon atom, respectively
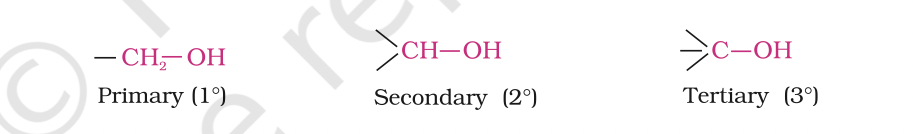
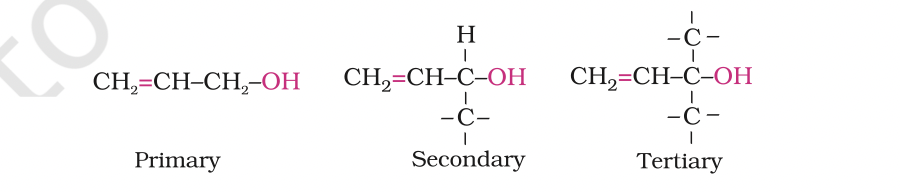
Allylic alcohols: In these alcohols, the —OH group is attached to a sp3 hybridised carbon adjacent to the carbon-carbon double bond, that is to an allylic carbon. For example
Benzylic alcohols: In these alcohols, the —OH group is attached to a sp3—hybridised carbon atom next to an aromatic ring.
For example
Allylic and benzylic alcohols may be primary, secondary or tertiary.
(ii) Compounds containing C sp2 OH bond:
These alcohols contain—OH group bonded to a carbon-carbon double bond, i.e., to a vinylic carbon or to an aryl carbon. These alcohols are also known as vinylic alcohols.
Vinylic alcohol: CH2 = CH – OH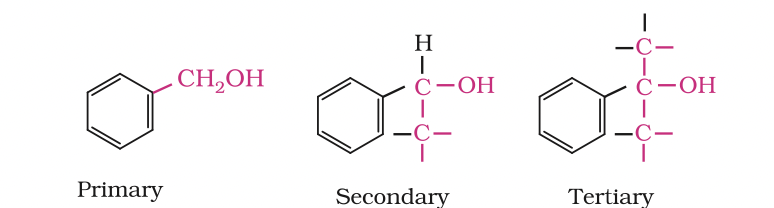
Phenols— Mono, Di and trihydric phenols:
Ethers are classified as simple or symmetrical
if the alkyl or aryl groups attached to the oxygen atom are the same, and mixed or unsymmetrical, if the two groups are different.
Diethyl ether, C2H5OC2H5, is a symmetrical ether whereas C2H5OCH3 and C2H5OC6H5 are unsymmetrical ethers.