2.4.1 - VSEPR Theory
Introduction
- ^^VSEPR^^ - Valence Shell Electron Pair Repulsion.
- VSEPR is only for covalent bonds
- Refers to the distribution of electrons so that they have the least amount of repulsion.
- This means that bonding regions will be as far away from each other as possible (repulsion)
- VSEPR results in the creation of five different shapes, depending on the number of bonding regions and lone pairs.
- Linear
- V-Shaped
- Trigonal planar
- Trigonal pyramidal
- Tetrahedral
- ^^Bonding regions^^ - electrons shared between two atoms
- ^^Lone pairs^^ - electrons that are not bonded to any other atom
- Central atoms have the lowest electronegativity of the entire molecule.
- Molecular shape (VSEPR) can be determined from number of electron regions and lone pairs.
- Structure affects polarity.
VSEPR Shapes
Linear Molecules

- Have either one or two bonding regions that are around a central atom.
- e.g - Cl2, CO2, HCl
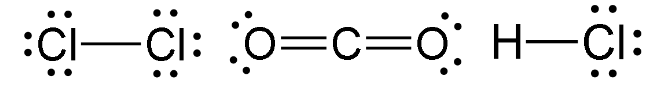
- Note that it refers to bonding regions, specifically - double and triple bonds (see above: CO2) can still be linear molecules.
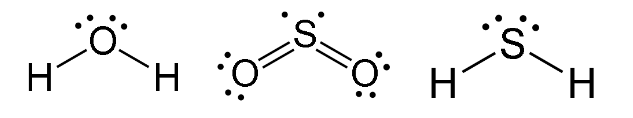
V-Shaped Molecules
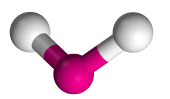
- Have two bonding regions and one/two lone pairs around a central atom.
- Will always be
- e.g H2O, SO2, H2S
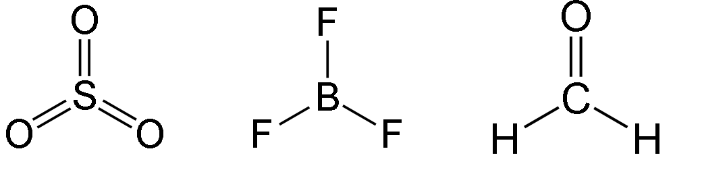
Trigonal Planar
- Have three bonding regions around the central atom.
- Molecule itself is flat.
- Can be - if molecule is symmetrical, it is . Else, .
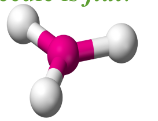
- e.g SO3, BF3, H2CO
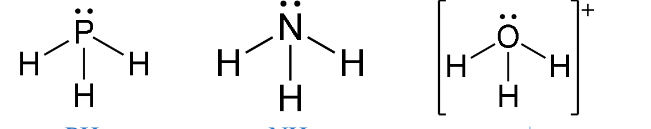
Trigonal Pyramidal
- Have three bonding regions and one lone pair of electrons.
- Will always be
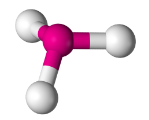
- e.g PH3, NH3, H3O+
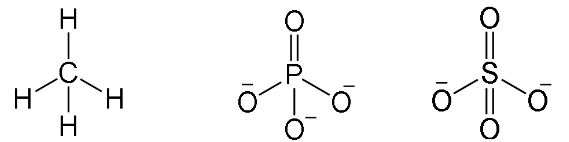
Tetrahedral
- Have four bonding regions.
- Can be - if molecule is symmetrical, it is . Else, .
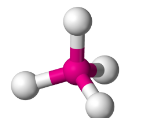
- e.g CH4, PO4(3-), SO4(2-)