Unit 3 - Elements and the Periodic Table
Elements, Compounds, and Mixtures
- Changes * Physical Changes - Don’t produce a new substance * Chemical Changes - Produces a new substance
- Matter Classification * Pure Substances * Can’t be broken into simpler compounds without going through a chemical change * Made of atoms that are chemically bonded to each other * Elements - Pure substances made of only 1 type of atom * Compounds - Pure substances made of 2+ types of atoms * Have fixed ratios between components * Mixtures * Mixing 2+ substances that are NOT chemically combined * Can be separated through physical means * Distillation - Separating components in a mixture through the use of their differing boiling points * Chromatography - Separating components using differences in their ability to pass through substrates * Don’t have fixed ratios between components * Types of Mixtures * Homogeneous Mixture - The components combined are indistinguishable * Heterogeneous Mixture - The components combined are distinguishable
\
Atomic Numbers and Electron Configurations
- Quantum Orbitals
* Orbitals - Location in an atom where an electron could be
* An atom can have any number of orbitals depending on the number of electrons they have
* Each orbital can hold 2 electrons
* Quantum Number - Describes the location of an electron / describes the orbital
* Three main quantum numbers used to describe orbitals: “N“, “L“, and “M“
* N - Principal quantum number; describes the size of the orbital
* Must be >0
* You can think of this as what ring in the Bohr model the orbital coincides with
* L - Angular momentum quantum number; describes the orbital shape
* Can be spherical, dumbbell/peanut, clover, etc shaped
* Can be between 0 → N-1
* M - Magnetic quantum number; describes orientation of the orbital
* Can be between -L → +L
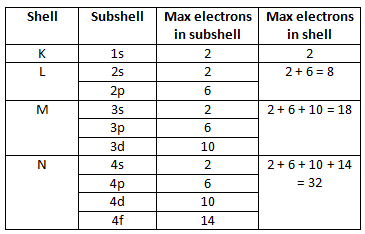
* Shells & Subshells
* Electron shell - A group of orbitals with the same principle quantum number (N)
* Shells are filled consecutively from the center/lowest energy orbitals outward
* Different shells can hold different numbers of electrons
* Full shells are the most stable
* Electron subshells - A group of orbitals with the same principle quantum number (N) AND angular momentum quantum number (L)
* Subshell Classifications
* L = 0 → S Orbital
* L = 1 → P Orbital
* L = 2 → D Orbital
* L = 3 → F Orbital
*
 * The number of different values the magnetic quantum number (M) can be is equal to the number of subshells of a certain classification
* The number of orbitals is equal to the number of different combinations of N, L, and M (Can be calculated with N^2)
* To calculate the number of electrons a shell can hold, you just double this number, since each orbital can hold 2 electrons
* This can also be calculated with the formula 2N^2
* Electron Configuration - How electrons are positioned in an atom
* Orbital Notation - A diagram that shows shells, subshells, and orbitals using lines & arrows
* Lines represent orbitals
* Numbers & letters at the bottom represent the name of the orbital
* Arrows represent electrons
* Upward and downward arrows represent a M subscript s value of either 1/2 or -1/2
* Pauli Exclusion Principle - No 2 electrons can have identical quantum numbers
* A fourth quantum number, M subscript s represents the quantum spin of a number
* Can have a value of either -1/2 and 1/2
* Only 2 values → only 2 electrons can be in a orbital, otherwise at least 1 pair of electrons will have identical quantum numbers
* Hund’s Rule - Electrons are placed in individual orbitals before being paired
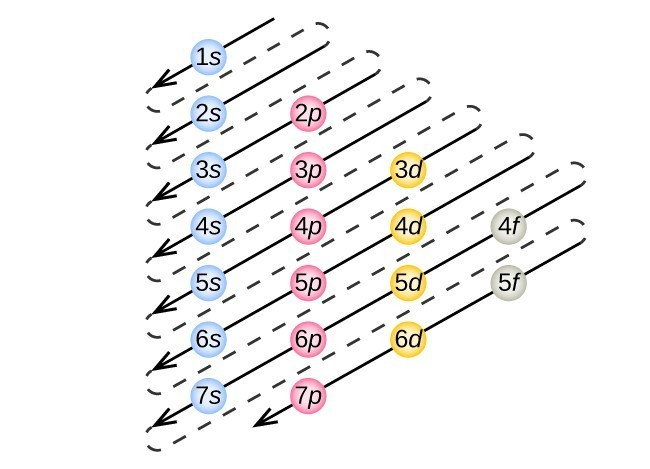
* Aufbau Principle - Electrons fill orbitals from lowest energy → highest energy
* This means electrons fill from lower N to higher N
* D and F are the exception; 3d has higher energy than 4s, so 4s will fill before 3d.
* Follow the diagonal rule to determine order in which orbitals are filled
*
* The number of different values the magnetic quantum number (M) can be is equal to the number of subshells of a certain classification
* The number of orbitals is equal to the number of different combinations of N, L, and M (Can be calculated with N^2)
* To calculate the number of electrons a shell can hold, you just double this number, since each orbital can hold 2 electrons
* This can also be calculated with the formula 2N^2
* Electron Configuration - How electrons are positioned in an atom
* Orbital Notation - A diagram that shows shells, subshells, and orbitals using lines & arrows
* Lines represent orbitals
* Numbers & letters at the bottom represent the name of the orbital
* Arrows represent electrons
* Upward and downward arrows represent a M subscript s value of either 1/2 or -1/2
* Pauli Exclusion Principle - No 2 electrons can have identical quantum numbers
* A fourth quantum number, M subscript s represents the quantum spin of a number
* Can have a value of either -1/2 and 1/2
* Only 2 values → only 2 electrons can be in a orbital, otherwise at least 1 pair of electrons will have identical quantum numbers
* Hund’s Rule - Electrons are placed in individual orbitals before being paired
* Aufbau Principle - Electrons fill orbitals from lowest energy → highest energy
* This means electrons fill from lower N to higher N
* D and F are the exception; 3d has higher energy than 4s, so 4s will fill before 3d.
* Follow the diagonal rule to determine order in which orbitals are filled
* 
\
The History and Arrangement of the Periodic Table
- Antoine Lavosier * Wrote “Elementary Treatise of Chemistry“ in 1789 * Considered the world’s first modern chemistry textbook * Classified elements into 4 groups: * Acid-making * Gas-like * Wrongly classified light & heat as elements * Metallic * Earthy * Almost entirely made up of compounds
- John Dobereiner * Arranged elements w/ similar properties into triads (groups of 3) * Difference between mass of elements 1 & 2 is about equal to difference in mass between 2 & 3
- John Newlands * Arranged elements by atomic mass * Established “law of octaves“ * Repeating pattern of similar properties every 8 elements
- Dimitri Mendeleev * Created the first iteration of the modern periodic table * Arranged elements by atomic mass * Organized table rows/columns by chemical properties
- Henry Moseley * Arranged elements by atomic number * Account for variation in natural isotopes
- Periodic Table * Organized by atomic number (number of protons) * Columns have similar chemical properties due to having the same number of valence (outer) electrons * Each row is a new shell * Periods - A row on the periodic table * Atomic number increases from left to right * Chemical properties systematically change * Groups/Families - A column on the periodic table * All elements in groups have similar chemical properties * Cells - Give information about an element * Atomic number * Atomic mass * Atomic symbol * Element name
- Elements * Natural: Elements 1-94 * Man-Made: Elements 95-118 * Metals: Left of the “staircase“ except hydrogen * Malleable * Ductile * Conduct heat & electricity * Mostly solids * Semi-Metals/Metalloids: The “staircase” * Properties of both groups * Non-Metals: Right of the “staircase“ plus hydrogen * Brittle * Poor Conductors * Can be any state * Main Group Elements * Alkali Metals - Group 1 * Silver colored * Soft * Highly reactive with water/oxygen * Oxidizes in air * Alkaline Earth Metals - Group 2 * Silver colored * More brittle than alkaline metals * Somewhat reactive * Low density, melting, and boiling points * Halogens - Group 17 * Highly reactive w/ metals * Form salts * Toxic to most organisms * Mostly occur as diatomic molecules * Noble Gases - Group 18 * Stable; don’t bond w/ other atoms * Non-flammable * Extremely low boiling points * Used in lights, produces colors when excited * Transition Metals * Form colored compounds * Some have unique properties * Some are magnetic * Some are very reactive * Inner Transition Metals * Can be radioactive * Lanthanides * Actinides
\
Electrons and the Periodic Table
- Noble Gas Notation * Using noble gases to represent filled shells in longhand electron configuration * Separates valence and non-valence(core) electrons in an atom
- Valence Electrons * The number of electrons on the outer shell of an atom * Determines the chemical properties of the atom * Correlated with the groups that the element is in in the periodic table * Group number = number of valence electrons * Determining Valence Electrons * Periods 1-3 * Group number / highest S and P orbitals * Periods 4+ * Highest S and P orbitals + partially filled d and f orbitals
- Periodic Table & Orbitals * S-Block Elements - Elements in groups 1 & 2 + Helium * Has valence electrons in the S orbital * P-Block Elements - Elements in groups 13 → 18 - Helium * Rows 1-3 * Has valence electrons in the S and P orbitals, with the last added electron being in the P orbital * Rows 4+ * Has valence electrons in the S, D, and P orbitals, with “N“ of the D subshell being 1 less than the N of the S & P subshells and the last added electron being in the P orbital * D-Block Elements - Elements in groups 3 → 12 + Lutetium and Lawrencium * Has valence electrons in the S and D orbitals, with “N“ of the D subshell being 1 less than the N of the S subshell and the last added electron being in the D orbital * F-Block Elements - Lanthanides & Actinides - Lutetium and Lawrencium * Has valence electrons in the S and F orbitals, with “N“ of the F subshell being 2 less than the N of the S subshell and the last added electron being in the F orbital * Exceptions * Chromium * Predicted - [Ar] 4s^2 3d^4 * Actual - [Ar] 4s^1 3d^5 * Often D & F block elements that are transition metals * Happens because electrons fill lowest energy shell
\
Periodic Trends
- Atomic Radius - 1/2 the distance between two identical atoms in a diatomic molecule * Increases down a group * Decreases across a row * More protons → electrons are pulled slightly closer together
- Ionic Radius - Measure of the size of an ion * Anion - Negative ions (atoms that gain electrons) * Larger; More electrons cause more electron repulsion * Cation - Positive ions (atoms that lose electrons) * Smaller; less electrons cause less electron repulsion * Increase down a group * Decrease for cations across a row * Decrease for anions across a row * Increase when switching from cations to anions across a period
- Ionization Energy - The energy required to remove an electron from an atom in a gas phase * Changes based * Nuclear charge * Distance from nucleus * The number of already removed electrons * First Ionization Energy - Energy needed to remove 1 electron from an atom * Second ionization energy - amount of energy to remove another electron after the first one is removed, etc * Main Group Elements * Increases across periods * Decreases down groups * Decreases between groups 2 & 13 and groups 15 & 16
- Electron Affinity - Energy required to add an electron to a neutral atom in a gas phase * Decreases across a period * Increases down a group
- Electronegativity - How much an atom attracts other electrons from other atoms * Increase across a period * Decrease down a group
\