Carbs
carbohydrates:
- these include sugars, starch, glycogen, and cellulose and they only contain carbon, oxygen, and hydrogen
| Type of carbohydrates | features |
|---|---|
| Monosaccharides | Simple sugars that only contain one molecule. E.g triodes, pentodes, hexoses |
| disaccharides | Two simple sugars chemically linked by glycosidic bonds formed due to a condensation reaction Sucrose = glucose + fructose Lactose = glucose + galactose maltose = glucose + glucose |
| Polysaccharides | Many simple sugars linked by glycosidic bonds Starch = energy store in plants Glycogen = fuel store in animals Cellulose = in plant cell walls |
structure of glucose:
- has the molecular formula of C6H12O6
- the carbon atoms are numbered from 1 on the far right, to 6 at the top
The isotopes of glucose:
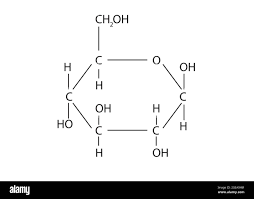
- Alpha glucose, where both the OH groups are ‘below the ring’
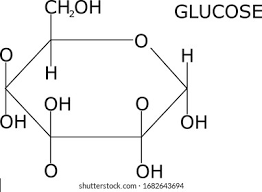
Beta glucose, where one OH is below and the other is above ‘the ring’
Testing for glucose:
- using Benedict’s solution which is actually copper (II) sulphate which has a blue colour
- When a reducing sugar is heated with Benedict’s, the COOH group in the molecule reduces the Cu2+ ion to Cu+ ions which formed a brick red precipitate of copper (I) oxide.
- There is a range of results from blue → green → yellow → brown → red
disaccharides:
Are made of two monosaccharides jointed together by glycosidic bonds that form from condensation reactions and produce water (hydrolysis uses water to break these bonds in the opposite reaction)
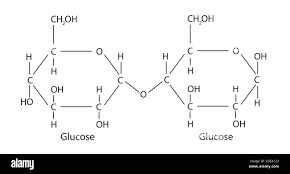
- the glycosidic linkage occurs between C-1 and C-4 of the alpha glucose molecule
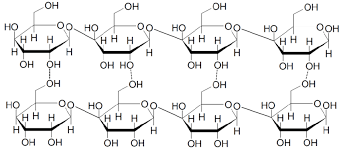
polysaccharides:
Are polymers made up of many monosaccharides linked together by glycosidic bonds formed during condensation reactions.
Polysaccharides are macromolecules as they’re giant molecules.
starch:
- is a mixture of 2 polysaccharides, both of which are polymers of alpha glucose
- Amylose - unbranched chain of alpha glucose
- Amylopectin - highly branched chains of alpha glucose
- The molecule is highly coiled due to the 1-4 glycosidic bonds in amylose and highly branched due to the 1-6 and 1-4 glycosidic bonds in amylopectin
- Starch is a major store of energy in plants and therefore has to be compact.
- It’s useful as it is compact and insoluble so it doesn’t have an osmotic effect
- Starch is fast at being hydrolysed in order to produce energy which is used in respiration to form energy due to the highly branched nature of it
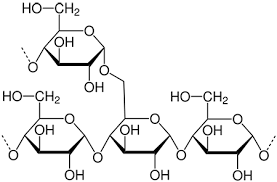
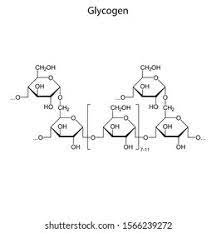
glycogen:
- is a polymer of alpha glucose
- It is very similar to amylopectin, but is larger and has more branches
- Glycogen is found in the liver cells and the muscle cells and is a energy store in humans and animals
- Glycogen is very quickly hydrolysed due to the branching that forms because of the 1-6 and 1-4 glycosidic bonds between 2 molecules of alpha glucose
- Glycogen is branched after every 10 glucose residues where as amylopectin is branched every 30 glucose residues (residues = when monomers are linked together in a polymer)
cellulose:
- is a polymer of beta glucose
- Cellulose is a very straight and unbranched molecule
- This is because every other beta glucose molecule is inverted which allows the 1-4 glycosidic bonds to form in a straight way that produces long straight chains
- The chains naturally become packed into fibres which are held together by hydrogen bonds - these strong fibres that are arranged in different directions are what make plant cell walls strong and able to withstand the hydrostatic pressure