
Exam 1
Changes in the ventricular system in schizophrenia
One of the most consistent findings in schizophrenia is the enlargement of the brain's ventricles

Amigdala → regulating anxiety, aggression, fear conditioning, emotional memory, and social cognition.
Located in the forebrain, diencephalon. part of limbic system
Regulates vital functions including hunger, thirst, temperature, sex
sends strong outputs to:
midbrain/hindbrain (autonomic function)
pituitary (neuroendocrine gland)
Fear reflex
Thalamus to amygdala pathway carries information rapidly to the amygdala
The thalamus to cortex to amygdala pathway is slower but allows the external stimuli to be cognitively appraised
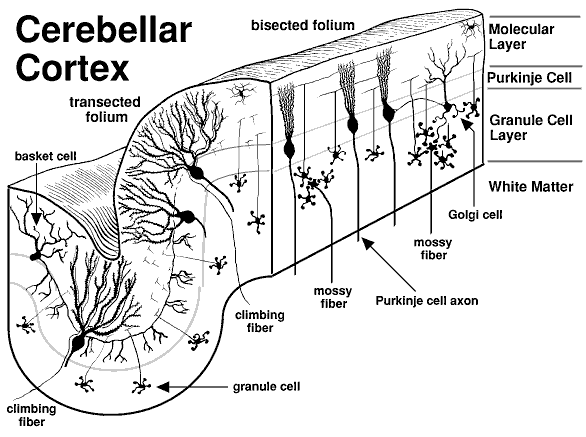
Cerebellum - involved in motor coordination and basic learning
Layers of the cerebellum
Granule Cell layer (innermost layer)→ composed of small neurons
Purkinje cell layer (mid-layer) → a single row formed by large cells (purkinje cells)
Molecular layer (outermost layer) → composed of parallel fibers of granular cells and dendritic trees of Purkinje cells
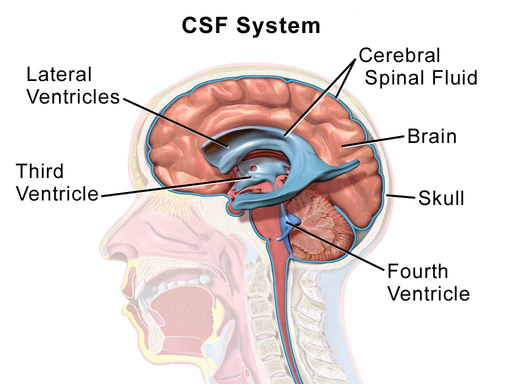
Ventricular system
series of four chambers filled with cerebral spinal fluid (CSF). Lined with choroid plexus, a membrane of cells that produces cerebral spinal fluid.
Two lateral ventricles in telencephalon
one in of each hemisphere, extend s into all four lobes
3rd ventricles in diencephalon
at the midline between the lateral ventricles
4th ventricles in hindbrain
CSF can exit here into the subarachnoid space, connect with with the central canal in the spinal cord
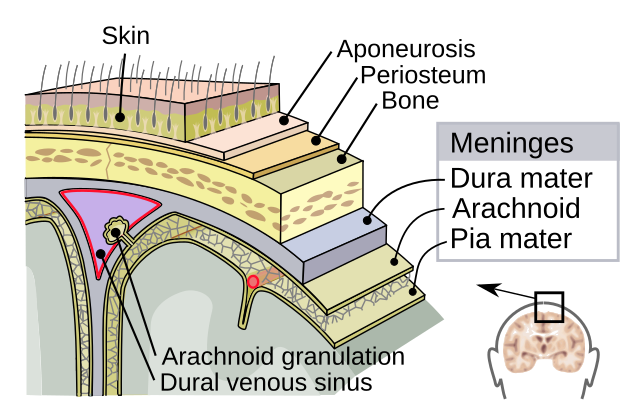
Dura matter - tought outermost sheet
Arachnoid - substance between the dura and pia matter that chushons the brain in cerebrospinal fluid (CSF)
functions of cerebrospinal fluid (CSF)
shock absorber
exchange medium btw blood and brain
Pia Matter - delicate innermost layer
Dynamic physical and metabolic barrier between blood and CSF/brain consisting of specialized endothelial cells that protects the brain from blood-borne compounds and maintain homeostasis in the brain
Composed by
Intercellular pathway → passage of water-soluble molecules
Transcellular lipophilic pathway → passive diffusion of lipid-soluble molecules across the barrier
Transport protein pathway → active diffusion of large molecules across the barrier by specific proteins
Protein pumps → active transport back into the bloodstream of some lipophilic molecules
Major arteries to the brain (three of them)
The anterior, middle & posterior cerebral arteries
The anterior and middle originate form the internal carotid artery
the posterior originates form the basilar artery that itself arises from the vertebral arteries
Action potential (AP) → rapid electrical signal that travels along the axon of a neuron
Neurotransmiter → chemical messenger between neurons.
Neuron at rest = balance of electrochemical forces
Ions → electrically charged molecules.
Dissolved in intracellular fluid, separated from the extracellular fluid by the cell membrane
Anions → negatively charged
Cations → positively charged
Sodium-potassium pump
maintains resting potential
pumps 3 Na+ out for every 2 K+ pumped in
Equilibrium potential → K+ reaches equilibrium when its movement out is equal to the movement in
equilibrium potential of the resting membrane potential is about -65mV (btw -50 & -80mV)
Brief and large change in membrane potential produced by the movement of Na+ ions into the cell
originates in the axon hillock and propagates along the axon towards axon terminals
carries information to postsynaptic targets
unidirectional as a result of the refractory state of the membrane post-depolarization

Vocab
Depolarization → decrease in membrane potential
inside of cell becomes more positive
Repolarization
Hyperpolarization → increase in membrane potential
inside of cell becomes more negative
Membrane potential at any given time depends on how many and which ion channels are open
Refractory period → time when only some stimuli can produce an action potential
there are two phases of it
Absolute refractory period → time where no action potentials can be produced
Relative refractory period → time when only strong stimulation can produce an action potential

K+ creates resting potential (-65 mV)
Open K+ leak channels → reached equilibrium potential
Na+ are closed
Cell become more negative increasing the membrane potential
closed K+ channels → K+ leak channels allow K+ flow in and out → make the cell negative → bring it closer to threshold
Absolute refractory period
At threshold (-40mV) voltage gated Na+ channels open allowing Na+ inside the cell
neurons have an all-or-nothing property that makes it so that a neuron must reach the -40mV threshold for the neuron to fine.
if the threshold is not reach not action potential will occur
the membrane undergoes depolarization until its peak at 40mV
Relative refractory period
At 40mV the Na+ channels close automatically, K+ channels open creating a disbalance that causes afterpotential
membrane undergoes depolarization and then hyperpolarization
Membrane returns to its resting potential
all channels close (except K+ leak channels)
K+ will diffuse in and out the cell while all the anions remain inside the cell allowing it to return to its resting potential.

Conduction velocity → the speed of propagation of AP which varies with the diameter of the axon
the smaller the diameter the faster it goes
Since there is no myelin sheath in the unmyelinated axons the conduction of AP in them is slow (10 m/s)
invertebrates
Rapid conduction (150s/m) thanks to myelin sheath
vertebrates
Saltatory conduction along the myelinated axon
Saltatory conduction → the axon potential travels inside the axon and jumps from node to node
Nodes of Ranvier → small gaps in the insulating myelin sheath
axon is exposed
genetic abnormality of ion channels often causing a disorder (23 currently identified)
Na+ channelopathy → various seizure, muscle and cardiac disorders
Cl- channelopathy → associated with deafness, kidnney problems, movement disorders and epilepsy
Certain animals contain toxins that block specific ion channels
Toxins that block voltage gated Na+ channels
Tetrodoxin (TTX) → produced in the ovaries of puffer fish
Blocks Na+ channels by binding to the outer pore of voltage-gated sodium channels in nerve cells, effectively preventing sodium ions from entering the cell and thus inhibiting the generation of action potentials, leading to rapid weakening and paralysis of muscles, including those of the respiratory tract, which can lead to respiratory arrest and death.
Saxitoxin (STX) → produced by algae
acts similar to TTX
Toxins that force voltage gated Na+ channels to remain open
Batrachotoxin → produced by poison arrow frogs
Binds to and irreversibly opens the sodium channels of nerve cells and prevents them from closing, leading to irreversible depolarization of nerves and muscles, fibrillation, arrhythmias and eventually cardiac failure
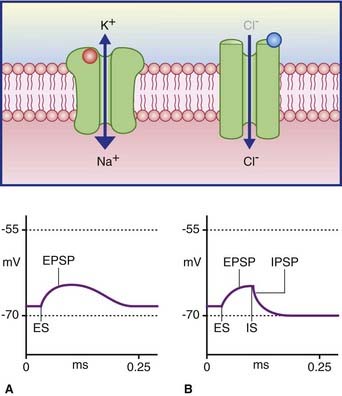
Synapses cause local, graded changes in the postsynaptic membrane potential
Synaptic delay → delay between an action potential
reaching the axon terminal and creating a postsynaptic
potentia
Postsynaptic potential → brief change in resting potential
there are 2 types
Inhibitory postsynaptic potential (IPSP) → produces a small hyper-polarization, pushing the cell further away from threshold (inhibiting the cell’s ability to produce a new AP)
this potential is a result of Cl− ions entering the cell thus making it more negative (below resting potential)
Excitatory postsynaptic potential (EPSP) → produces a small local depolarization, pushing the cell closer to threshold
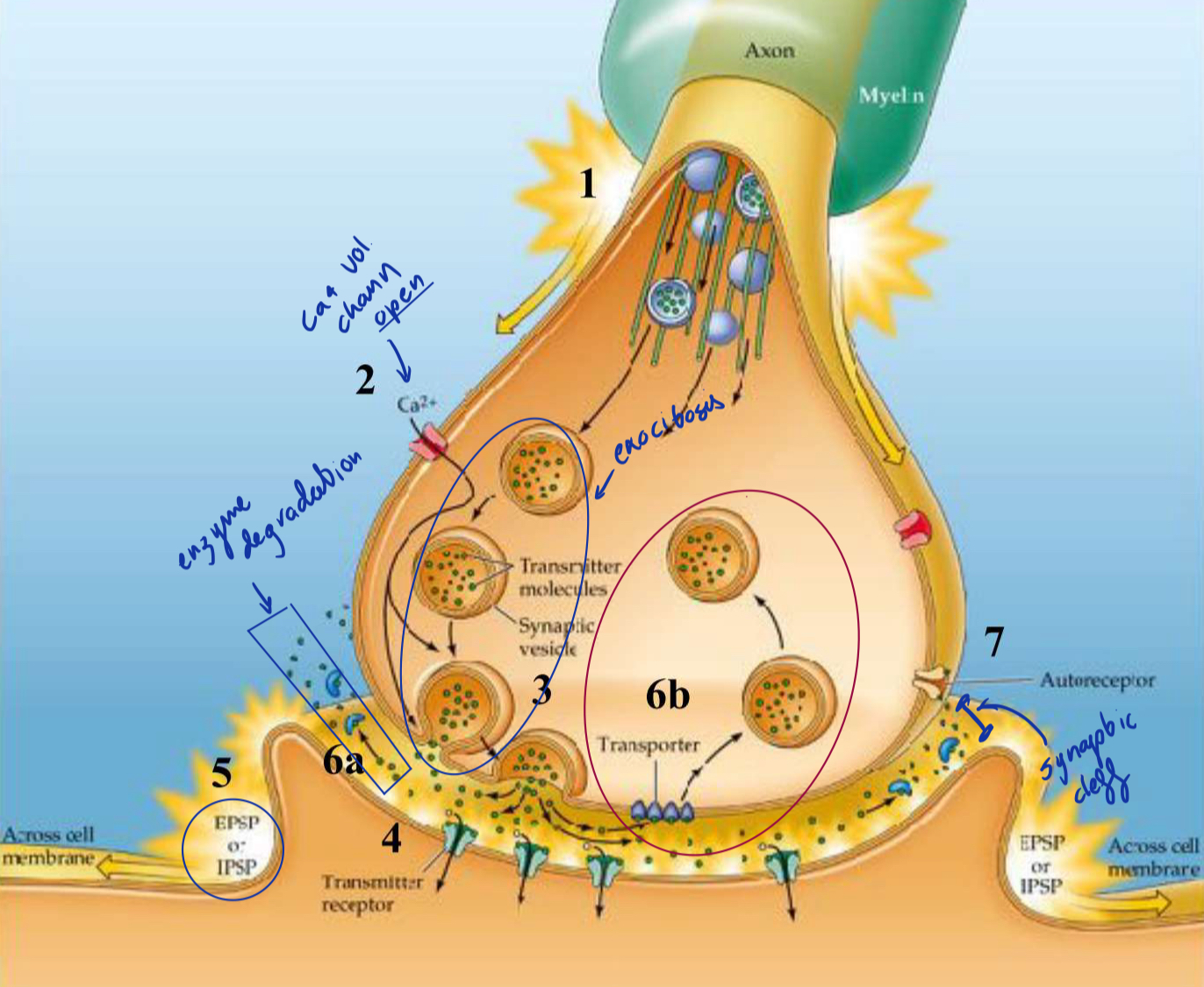
AP travels down the axon to terminal
voltage gated Ca2+ (Calcium) open and Ca2+ enters
Neurotransmitters are released into the synaptic cleft bia exocytosis
Synaptic vesicles fuse with membrane and release the neurotransmitters
Neurotransmitters cross the synaptic cleft and bind to receptors in the postsynaptic membrane, causing either EPSP or IPSP
EPSP or IPSP spread towards the post synaptic axon hillock
Spacial summation → summing of potentials that come from differents parts of the cell
If the EPSP input is stronger than the IPSP and their sum exceeds the -40mV threshold the axon hillock will produce an action potential
Neurotransmitter action is brief it will be either…
Inactivated by enzymatic degradation (6a)…
rapid breakdown and inactivation of transmitter by an enzyme (ex. AchE breaks down ACh and recycles it)
Or removed by transporters for reuptake and recycling (6b)
transmitter is taken up into (endocytosis) the presynaptic cell by specialized transporters (SSRI)
neurotransmitters may activate presynaptic autoreceptors resulting decrease of its own release
Receptors are activated/inhibited by ligands
two types
Endogenous ligands → Produced by the body
neurotransmitters (acetylchloride → ACh)
ACh is a neurotransmitter that can bind to the nicotinic receptor to allow Na+ ions to enter the cell
Hormones
Exogenous ligands → from outside the body
drugs & toxins from outside the body
Nicotinic ACh receptors → ligand-gated ion channel
located…
on muscles
in autonomic ganglia
Muscarinic ACh receptors → G-protein-coupled receptor
located…
in the brain
organs innervated by the parasympathetic division of the autonomic system
activated by…
ACh & muscarine (found in mushrooms)
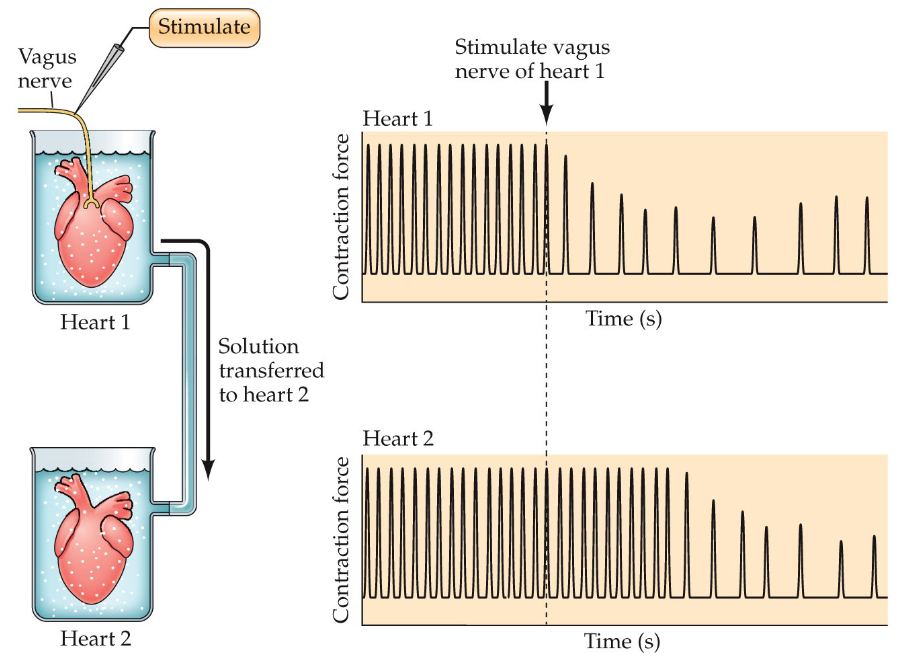
Otto Loewi's demonstration involved electrically stimulating the vagus nerve of a frog heart, causing it to slow down, then transferring the fluid surrounding that heart to another frog heart, which also slowed down, proving that a chemical substance released by the first heart (acetylcholine) acted as a "chemical messenger" to influence the second heart's rate
This experiment provided the first evidence of chemical neurotransmission.
Receptor # in cells are dynamic
this is a result of things such as daily changes in adulthood, changes during development and changes of drug use
Two types of regulaions
Up-regulation → increase receptor #
Down-regulation → decrease receptor #
There are 2 ways in which receptors control ion channels
Ionotropic receptors (ligand-gated ion channel)
activated by neurotransmitters → direct opening of ion channel → ion enters the cell
acts fast → only 2 mol. of neurotransmitters needed for the ion channels to open
Example → Nicotinic ACh receptor

Metabotropic receptors (G-protein-coupled receptors)
activated by neurotransmitters → actvation of G-proteins → activation of 2nd chemical (secondary messenger) or activation of nearby ion channel → ion enters the cell
acts slow → causes an indirect opening of ion channel
80% of ligands (neurotransmitters and hormones) bind to G-protein-coupled receptors
Example → Muscarinic ACh receptors

Chemical synapse
Chemical substance mediates synaptic transmission form pre to post-synapse
Synaptic cleft = 20-40 nm
Electrical synapse (also known as gap junctions)
Ions flow through large channels called connexons into adjacent cells
Synaptic cleft = 2-4 nm
no synaptic delay!
2 types of substances
Exogenous substances → substances from outside our own bodies, used throughout human history to affect our physiology and behavior
Endogenous substances → substances that naturally occur within the body
2 types of receptors
Inotropic (fast)→ when activated by a neurotransmitter binding to it the receptor will change in shape
Metabotropic (slow)→ when activated by a neurotransmitter binding to it the receptor will alter chemical reactions
They are Versatile
a single neurotransmitter can bind to several receptor subtypes
Inotropic (fast)
Metabotropic (slow)
either type of receptor can exite or inhibit a target cell
substance exists in presynaptic axon terminal
is released when AP reach axon terminals
receptors of the substance exist in presynaptic membrane
when experimentally applied, substance induces changes in postsynaptic cells
blocking the release of the substance prevents changes in postsynaptic cell
Glutamate
most prevalent excitatory neurotransmitter
plays a role in → cognition, learning and memory
binds to both ionotropic (NMDA, AMPA, kainate) and metabotropic (mGLUR1-18) receptors
Excitotoxicity → excess glutamate release resulting in damage/loss of neurons
plays a role in → Alzheimer’ disease, brain trauma,
seizure disorders, Parkinson's disease, stroke,
Huntington's disease, autism, schizophrenia
GABA (gamma-Aminobutyric acid)
most prevalent inhibitory neurotransmitter
binds to both ionotropic (GABAA, GABAC) and metabotropic (GABAB) receptors
Drugs based on enhancing GABA functions
Hypnotics, sedatives, tranquilizers, anticonvulsants
most well known → benzodiazepines (Diazepam=Valium)
Alchohol, cannabis
Used to treat pain, seizures, anxiety and migrane
Acetylcholine (ACh)
can be both inhibitory or excitatory
binds to ionotropic (nicotinic) and metabotropic (muscarinic) receptors
plays a role in → arousal, attention, learning & memory, and motivation
damage to cholinergic nerve cell bodies in the brain is associated with Alzheimer’ disease
Catecholamines
Dopamine (DA)
found in neurons in the →
Mesostrial pathway originating in the substantia nigra and projecting to the striatum
important in motor controll
Mesocorticolimbic pathway originating in the ventral tegmental area (VTA) and projecting to the cortex & limbic areas
important for reward and aversion, and learning
Abnormalities associated with schizophrenia and depression
Epinephrine/adrenaline
not in the brain
Norepinephrine (NE)
synthesized in the locus coeruleus (pons) and lateral tegmental system (midbrain)
binds to metabotropic (alpha 1, alpha 2; beta1, beta 2, beta 3) receptors
modulates mood, arousal, attention, behavioral flexibility and sexual behavior
Drugs → beta blocker (proparanolol)
reduces preformance anxiety
Indoleamines
Melatonin
sleep & wakefulness
Seratonin (5-hydroxythryptamine, 5-HT)
synthesized in 7 raphe nuclei, with dorsal raphe nucleus being the largest
role in sleep, mood, sexual behavior, depression and anxiety
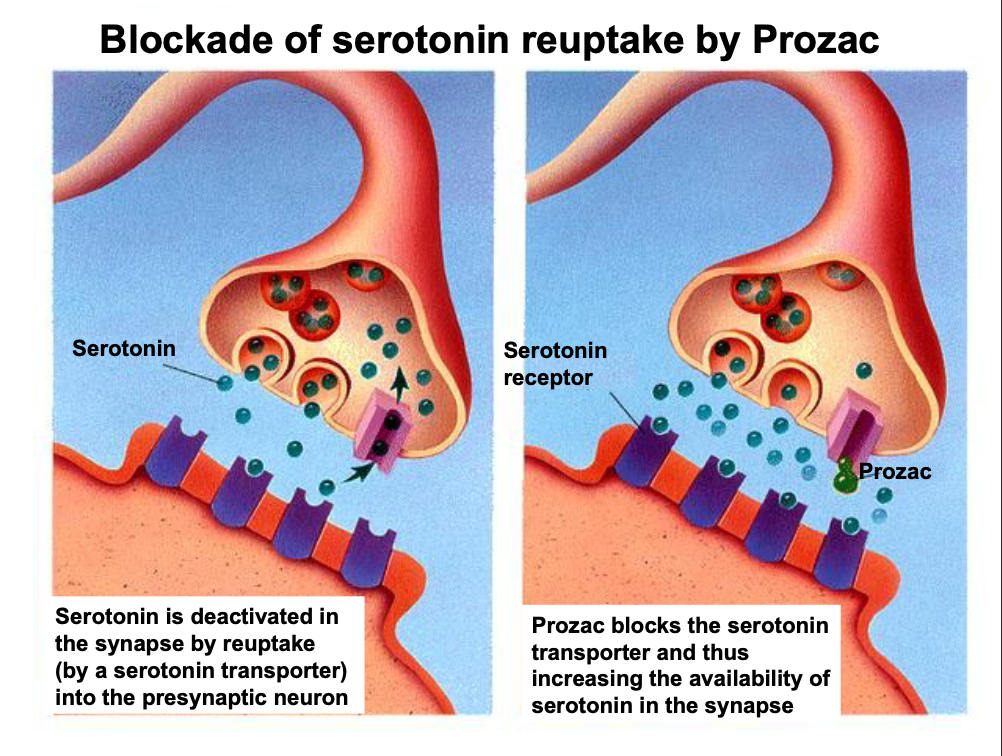
Drugs → selective serotonin reuptake inhibitors (SSRI)
antidepressants (prozac)
a drug can be…
Agonist → mimics effects of usual neurotransmitter
Antagonist → binds receptor without activating it, thereby blocking the receptors from being activated
competitive vs non-competitive antagonist
Competitive antagonist → compounds that compete with agonists for the same receptor, but they do not exert an agonist effect themselves and so reduce the effect of any agonist present
Non-competitive antagonist → a compound that prevents an agonist from having a biological response by binding to a receptor protein
Inverse agonist → binds receptor and initiates opposite effect of usual neurotransmitter
Binding affinity → the degree of chemical attraction btw a ligand and a receptor
the more affinity a drug has for its receptor the lower the doses
neurotransmitters are low-affinity ligands allowing them to dissociate from receptors
Efficacy (intrinsic activity) → ability of a bound ligand to acctivate the receptor
Agonist have high efficacy
antagonists have low efficacy
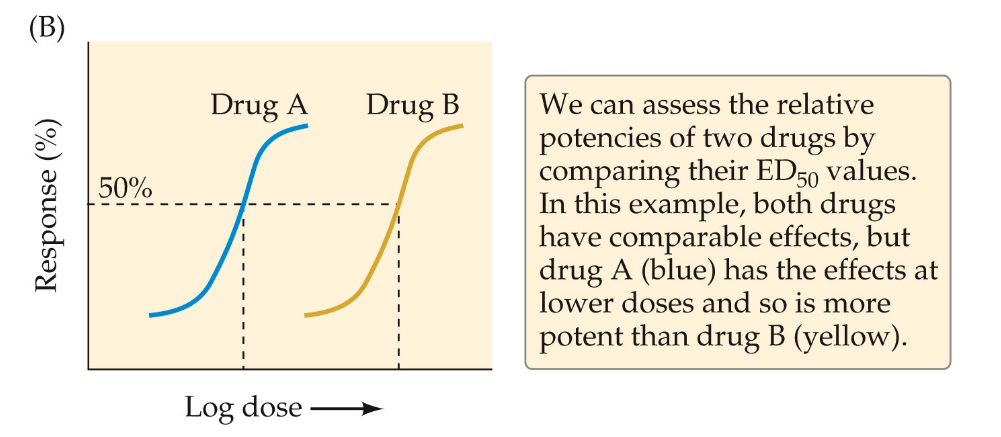
ED50 value
effective dose for 50% of people recive the drug
allows comparison of potency of drugs
the higher the potency the more comparable the effects are at lower doses
Therapeutic index
separation between effective dose and toxic/lethal dose
determined by comparing ED50 with TD50 (toxic dose for 50% of individuals) or LD50 (lethal dose for 50% of individuals)
Antidepressants
Class of drugs used to treat symptoms of depression
Monoamine oxidase (MAO) inhibitors
prevent breakdown of monoamines at synapses
Tricyclic antidepressants
prevent the reuptake of serotonin and norepinephrine into presynaptic axon terminals
Selective serotonin reuptake inhibitors (SSRIs)
same mechanism as tricyclic antidepressants but with fewer side effects
Anxiolytics (tranquilizers)
class of drugs used to trreat anxiety disorders
Benzodiazepine agonists
act on GABAA receptors and enhanse the inhibitory effects of GABA
safe and and effective for short term use but long-term use is discouraged because of dependency and withdrawal effects.
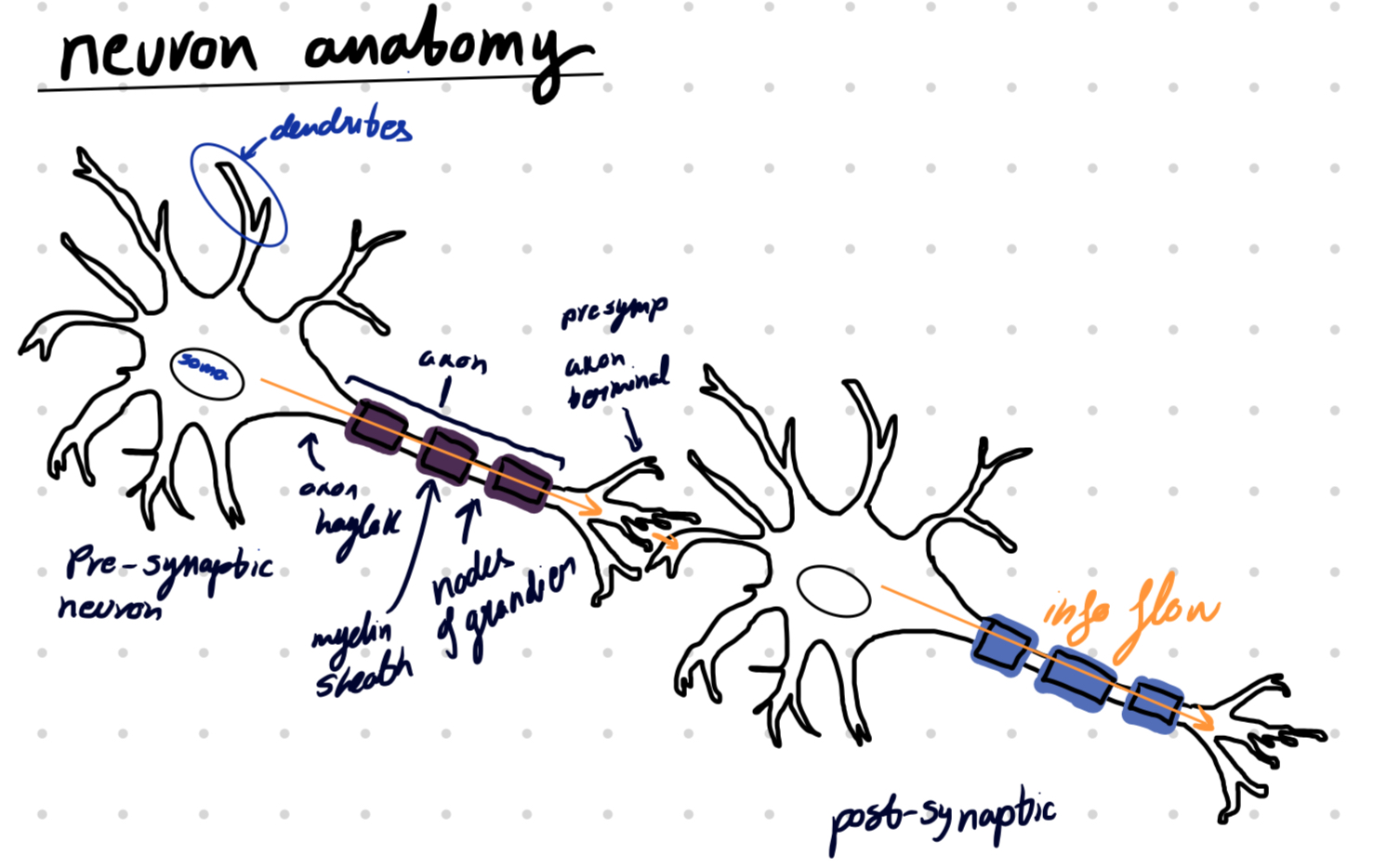
Dendrite → communication with other neurons
Synapse → receiving part of a neuron
Axon → sending part of a neuron
Axon → Receiver
Synapse → Sender
Dendrite → Junction between two neurons
Myelin sheath → Insulating layer
Oligodendrocytes → remove debris from injured cells
Microglia → form myelin sheath around axons in peripheral nervous system
Astrocytes → form myelin sheath around axons in central nervous system
Swann cell → monitor neural activity
a) Olfactory
b) Trigeminal
c) Vestibulocochlear
d) Optic
e) Hypoglossal
lumbar → pelvic
sacral → base of spinal cord
coccyx → lower back
cervical → trunk
thoracic → neck
a) constricts the airways
b) stimulates digestion
c) constricts the blood vessels
d) stimulates salivation
e) accelerates heartbeat
f) constricts the pupils
anterior → tail end
lateral → middle
ventral → sideways
dorsal → front
posterior → back
medial → head end
Hypothalamus → hunger
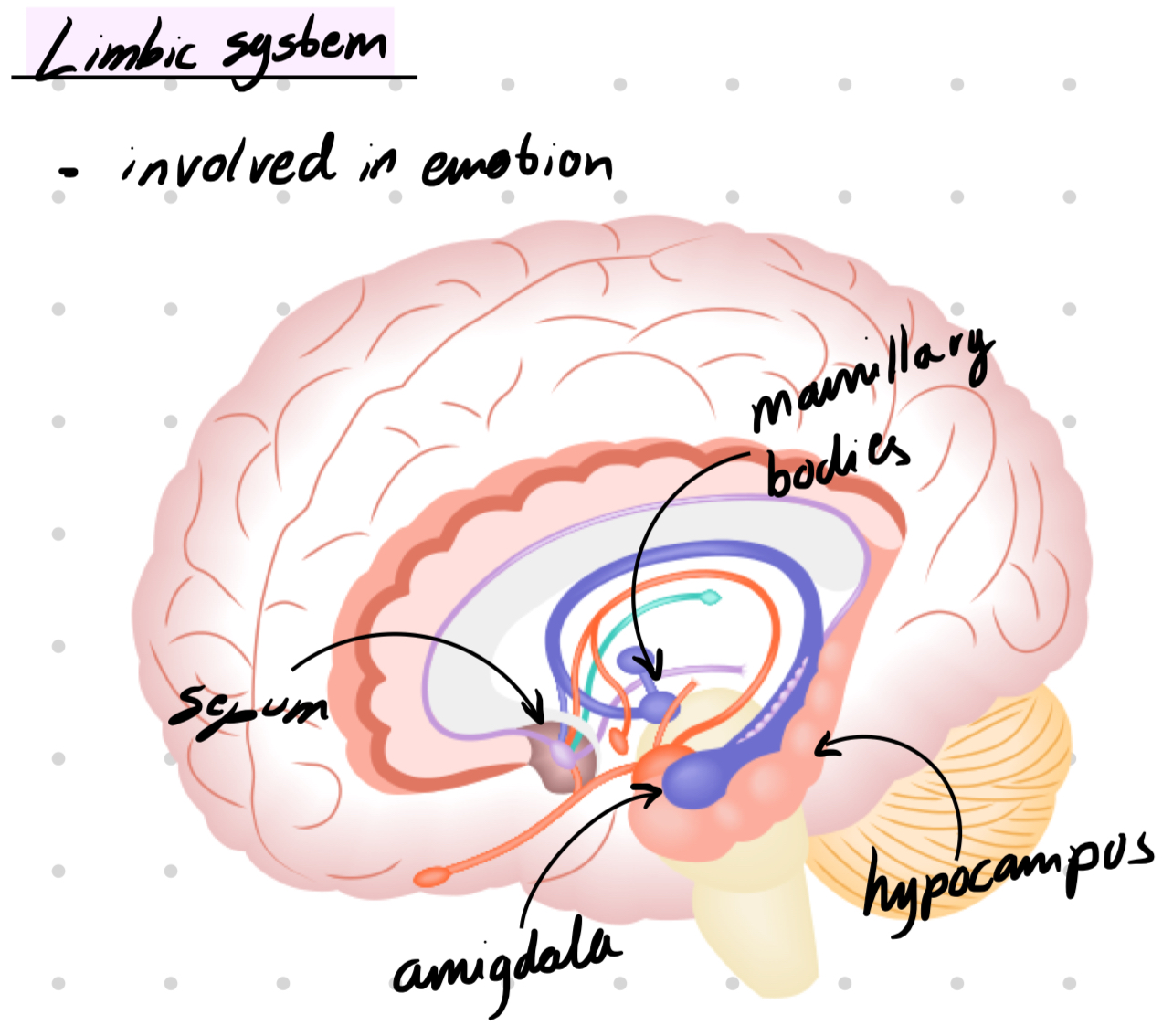
Amygdala → emotion
Septum → emotion
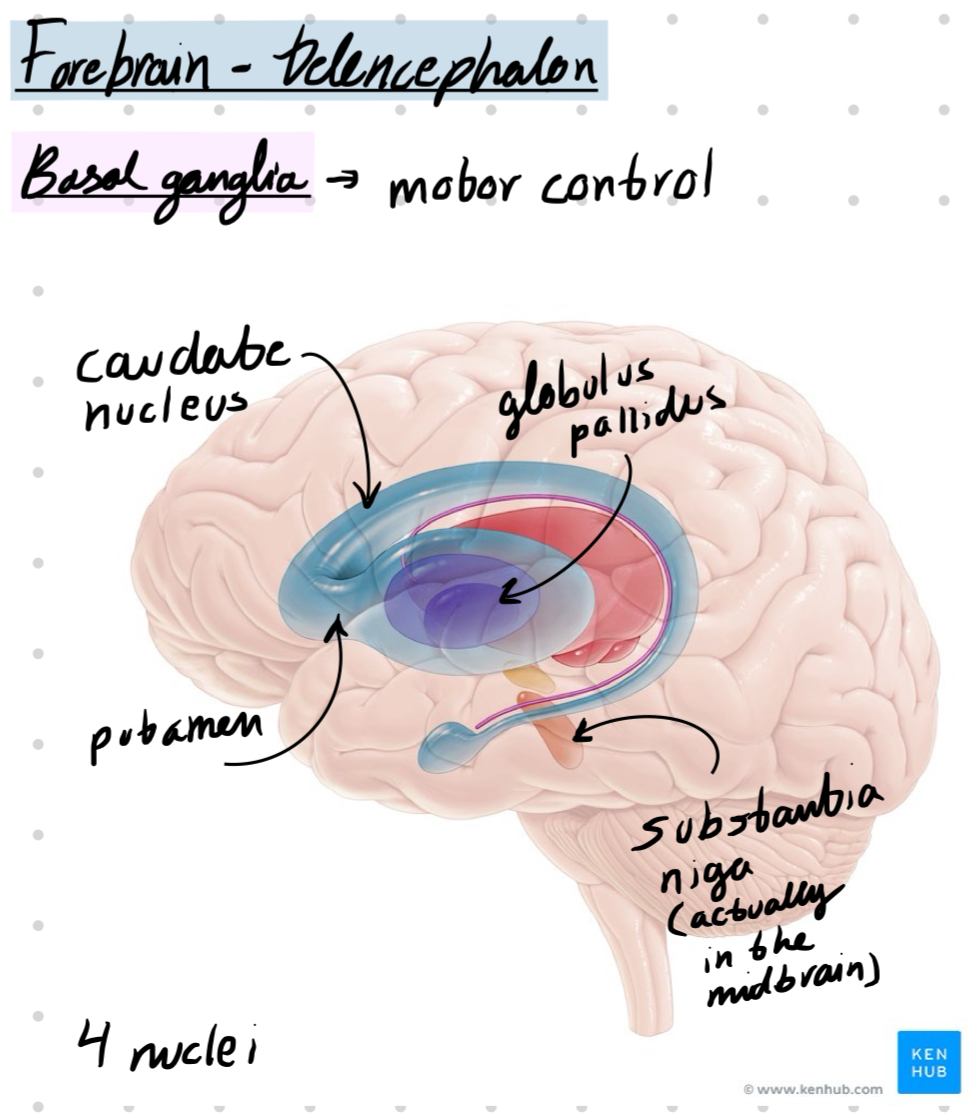
Caudate nucleus → motor control
Putamen → motor control
Thalamus → sensory information
a) Blood-brain-barrier
b) Choroid plexus
c) Meninges
d) Cerebral hemispheres
e) Ventricular system of the brain
f) Carotid arteries
g) Spinal cord
a) pumping potassium and sodium into the cell
b) pumping sodium into the cell and potassium out of the cell
c) pumping potassium into the cell and sodium out of the cell
d) initiating an action potential
e) creating a positive net charge inside the cell
K+ → maintains resting potential
NA+ → prevents action potential
Cl- → required for action potential
1 → Voltage-gated Na+ channels open
2 → Na+ influx (into the cell)
3 → Voltage-gated Na+ channels close and voltage-gated K+ channels open
4 → K+ efflux (out of the cell)
5 → Voltage-gated K+ channels close
a) synapse
b) dendrites
c) axon hillock
d) cell body
a) Electrical conduction across the synaptic cleft
b) Reuptake of neurotransmitters
c) Influx of calcium at the presynaptic membrane
d) Binding to autoreceptors
1 → Postsynaptic receptors are activated
2 → NA+ enters the cell
3 → Neurotransmitters are released in the synapse
4 → The action potential is propagated to the axon terminal
1 → Ionotropic synapse
2 → Electric synapse
3 → Metabotropic synapse
a) Type of ion that flows across membrane
b) Type of ligand
c) Type of target cell
d) Type of presynaptic terminal
a) Type of target cell
b) Type of postsynaptic terminal
c) Type of ion channel modulated by the metabotropic receptor
d) Type of ligand
a) slow
b) changes shape
c) activates G-proteins
d) neurotransmitter binds directly to the ion receptor
e) alters chemical reactions
a) type of receptor it binds to
b) whether it is released in the brain or periphery
c) concentration of ACh
Q5 - Match the neurotransmitter with region of synthesis
serotonin → locus coeruleus
norepinephrine → substantia nigra
dopamine → nucleus basalis
acetylcholine → raphe nuclei
GABA → Benzodiazepines
Serotonin → SSRI (antidepressants)
Norepinephrine → Propranolol
Opioids → Morphine
The higher the affinity of the drug, the - lower - the concentration of the drug.
a) low ED50
b) low affinity
c) high LD50
d) high TD50
a) Stimulate norepinephrine receptors
b) Decrease the synthesis of serotonin
c) Increase serotonin availability
d) Upregulation of postsynaptic serotonin receptors
a) pumping potassium and sodium into the cell
b) pumping sodium into the cell and potassium out of the cell
c) pumping potassium into the cell and sodium out of the cell
d) initiating an action potential
a) synapse
b) dendrites
c) axon hillock
d) cell body
a) impulses
b) axons
c) synapses
d) nerves
a) meninges
b) myelin
c) dura mater
d) pia mater
a) blood-brain-barrier
b) carotid arteries
c) spinal cord
d) ventricular system
e) meninges
Changes in the ventricular system in schizophrenia
One of the most consistent findings in schizophrenia is the enlargement of the brain's ventricles





Amigdala → regulating anxiety, aggression, fear conditioning, emotional memory, and social cognition.
Located in the forebrain, diencephalon. part of limbic system
Regulates vital functions including hunger, thirst, temperature, sex
sends strong outputs to:
midbrain/hindbrain (autonomic function)
pituitary (neuroendocrine gland)
Fear reflex
Thalamus to amygdala pathway carries information rapidly to the amygdala
The thalamus to cortex to amygdala pathway is slower but allows the external stimuli to be cognitively appraised

Cerebellum - involved in motor coordination and basic learning
Layers of the cerebellum
Granule Cell layer (innermost layer)→ composed of small neurons
Purkinje cell layer (mid-layer) → a single row formed by large cells (purkinje cells)
Molecular layer (outermost layer) → composed of parallel fibers of granular cells and dendritic trees of Purkinje cells

Ventricular system
series of four chambers filled with cerebral spinal fluid (CSF). Lined with choroid plexus, a membrane of cells that produces cerebral spinal fluid.
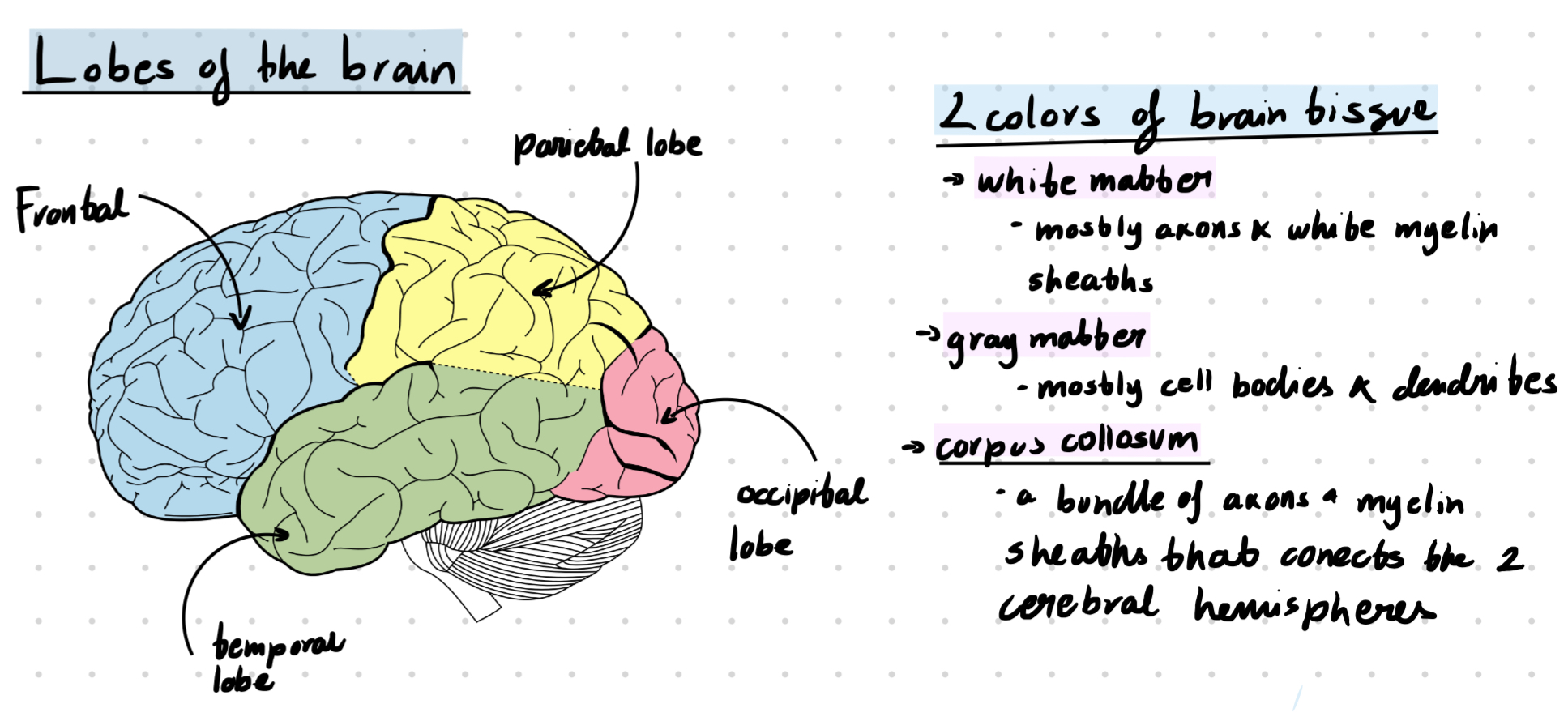
Two lateral ventricles in telencephalon
one in of each hemisphere, extend s into all four lobes
3rd ventricles in diencephalon
at the midline between the lateral ventricles
4th ventricles in hindbrain
CSF can exit here into the subarachnoid space, connect with with the central canal in the spinal cord

Dura matter - tought outermost sheet
Arachnoid - substance between the dura and pia matter that chushons the brain in cerebrospinal fluid (CSF)
functions of cerebrospinal fluid (CSF)
shock absorber
exchange medium btw blood and brain
Pia Matter - delicate innermost layer
Dynamic physical and metabolic barrier between blood and CSF/brain consisting of specialized endothelial cells that protects the brain from blood-borne compounds and maintain homeostasis in the brain
Composed by
Intercellular pathway → passage of water-soluble molecules
Transcellular lipophilic pathway → passive diffusion of lipid-soluble molecules across the barrier
Transport protein pathway → active diffusion of large molecules across the barrier by specific proteins
Protein pumps → active transport back into the bloodstream of some lipophilic molecules
Major arteries to the brain (three of them)
The anterior, middle & posterior cerebral arteries
The anterior and middle originate form the internal carotid artery
the posterior originates form the basilar artery that itself arises from the vertebral arteries
Action potential (AP) → rapid electrical signal that travels along the axon of a neuron
Neurotransmiter → chemical messenger between neurons.
Neuron at rest = balance of electrochemical forces
Ions → electrically charged molecules.
Dissolved in intracellular fluid, separated from the extracellular fluid by the cell membrane
Anions → negatively charged
Cations → positively charged
Sodium-potassium pump
maintains resting potential
pumps 3 Na+ out for every 2 K+ pumped in
Equilibrium potential → K+ reaches equilibrium when its movement out is equal to the movement in
equilibrium potential of the resting membrane potential is about -65mV (btw -50 & -80mV)
Brief and large change in membrane potential produced by the movement of Na+ ions into the cell
originates in the axon hillock and propagates along the axon towards axon terminals
carries information to postsynaptic targets
unidirectional as a result of the refractory state of the membrane post-depolarization

Vocab
Depolarization → decrease in membrane potential
inside of cell becomes more positive
Repolarization
Hyperpolarization → increase in membrane potential
inside of cell becomes more negative
Membrane potential at any given time depends on how many and which ion channels are open
Refractory period → time when only some stimuli can produce an action potential
there are two phases of it
Absolute refractory period → time where no action potentials can be produced
Relative refractory period → time when only strong stimulation can produce an action potential

K+ creates resting potential (-65 mV)
Open K+ leak channels → reached equilibrium potential
Na+ are closed
Cell become more negative increasing the membrane potential
closed K+ channels → K+ leak channels allow K+ flow in and out → make the cell negative → bring it closer to threshold
Absolute refractory period
At threshold (-40mV) voltage gated Na+ channels open allowing Na+ inside the cell
neurons have an all-or-nothing property that makes it so that a neuron must reach the -40mV threshold for the neuron to fine.
if the threshold is not reach not action potential will occur
the membrane undergoes depolarization until its peak at 40mV
Relative refractory period
At 40mV the Na+ channels close automatically, K+ channels open creating a disbalance that causes afterpotential
membrane undergoes depolarization and then hyperpolarization
Membrane returns to its resting potential
all channels close (except K+ leak channels)
K+ will diffuse in and out the cell while all the anions remain inside the cell allowing it to return to its resting potential.

Conduction velocity → the speed of propagation of AP which varies with the diameter of the axon
the smaller the diameter the faster it goes
Since there is no myelin sheath in the unmyelinated axons the conduction of AP in them is slow (10 m/s)
invertebrates
Rapid conduction (150s/m) thanks to myelin sheath
vertebrates
Saltatory conduction along the myelinated axon
Saltatory conduction → the axon potential travels inside the axon and jumps from node to node
Nodes of Ranvier → small gaps in the insulating myelin sheath
axon is exposed
genetic abnormality of ion channels often causing a disorder (23 currently identified)
Na+ channelopathy → various seizure, muscle and cardiac disorders
Cl- channelopathy → associated with deafness, kidnney problems, movement disorders and epilepsy
Certain animals contain toxins that block specific ion channels
Toxins that block voltage gated Na+ channels
Tetrodoxin (TTX) → produced in the ovaries of puffer fish
Blocks Na+ channels by binding to the outer pore of voltage-gated sodium channels in nerve cells, effectively preventing sodium ions from entering the cell and thus inhibiting the generation of action potentials, leading to rapid weakening and paralysis of muscles, including those of the respiratory tract, which can lead to respiratory arrest and death.
Saxitoxin (STX) → produced by algae
acts similar to TTX
Toxins that force voltage gated Na+ channels to remain open
Batrachotoxin → produced by poison arrow frogs
Binds to and irreversibly opens the sodium channels of nerve cells and prevents them from closing, leading to irreversible depolarization of nerves and muscles, fibrillation, arrhythmias and eventually cardiac failure
Synapses cause local, graded changes in the postsynaptic membrane potential
Synaptic delay → delay between an action potential
reaching the axon terminal and creating a postsynaptic
potentia
Postsynaptic potential → brief change in resting potential
there are 2 types
Inhibitory postsynaptic potential (IPSP) → produces a small hyper-polarization, pushing the cell further away from threshold (inhibiting the cell’s ability to produce a new AP)
this potential is a result of Cl− ions entering the cell thus making it more negative (below resting potential)
Excitatory postsynaptic potential (EPSP) → produces a small local depolarization, pushing the cell closer to threshold


AP travels down the axon to terminal
voltage gated Ca2+ (Calcium) open and Ca2+ enters
Neurotransmitters are released into the synaptic cleft bia exocytosis
Synaptic vesicles fuse with membrane and release the neurotransmitters
Neurotransmitters cross the synaptic cleft and bind to receptors in the postsynaptic membrane, causing either EPSP or IPSP
EPSP or IPSP spread towards the post synaptic axon hillock
Spacial summation → summing of potentials that come from differents parts of the cell
If the EPSP input is stronger than the IPSP and their sum exceeds the -40mV threshold the axon hillock will produce an action potential
Neurotransmitter action is brief it will be either…
Inactivated by enzymatic degradation (6a)…
rapid breakdown and inactivation of transmitter by an enzyme (ex. AchE breaks down ACh and recycles it)
Or removed by transporters for reuptake and recycling (6b)
transmitter is taken up into (endocytosis) the presynaptic cell by specialized transporters (SSRI)
neurotransmitters may activate presynaptic autoreceptors resulting decrease of its own release
Receptors are activated/inhibited by ligands
two types
Endogenous ligands → Produced by the body
neurotransmitters (acetylchloride → ACh)
ACh is a neurotransmitter that can bind to the nicotinic receptor to allow Na+ ions to enter the cell
Hormones
Exogenous ligands → from outside the body
drugs & toxins from outside the body
Nicotinic ACh receptors → ligand-gated ion channel
located…
on muscles
in autonomic ganglia
Muscarinic ACh receptors → G-protein-coupled receptor
located…
in the brain
organs innervated by the parasympathetic division of the autonomic system
activated by…
ACh & muscarine (found in mushrooms)

Otto Loewi's demonstration involved electrically stimulating the vagus nerve of a frog heart, causing it to slow down, then transferring the fluid surrounding that heart to another frog heart, which also slowed down, proving that a chemical substance released by the first heart (acetylcholine) acted as a "chemical messenger" to influence the second heart's rate
This experiment provided the first evidence of chemical neurotransmission.
Receptor # in cells are dynamic
this is a result of things such as daily changes in adulthood, changes during development and changes of drug use
Two types of regulaions
Up-regulation → increase receptor #
Down-regulation → decrease receptor #
There are 2 ways in which receptors control ion channels
Ionotropic receptors (ligand-gated ion channel)
activated by neurotransmitters → direct opening of ion channel → ion enters the cell
acts fast → only 2 mol. of neurotransmitters needed for the ion channels to open
Example → Nicotinic ACh receptor

Metabotropic receptors (G-protein-coupled receptors)
activated by neurotransmitters → actvation of G-proteins → activation of 2nd chemical (secondary messenger) or activation of nearby ion channel → ion enters the cell
acts slow → causes an indirect opening of ion channel
80% of ligands (neurotransmitters and hormones) bind to G-protein-coupled receptors
Example → Muscarinic ACh receptors

Chemical synapse
Chemical substance mediates synaptic transmission form pre to post-synapse
Synaptic cleft = 20-40 nm
Electrical synapse (also known as gap junctions)
Ions flow through large channels called connexons into adjacent cells
Synaptic cleft = 2-4 nm
no synaptic delay!
2 types of substances
Exogenous substances → substances from outside our own bodies, used throughout human history to affect our physiology and behavior
Endogenous substances → substances that naturally occur within the body
2 types of receptors
Inotropic (fast)→ when activated by a neurotransmitter binding to it the receptor will change in shape
Metabotropic (slow)→ when activated by a neurotransmitter binding to it the receptor will alter chemical reactions
They are Versatile
a single neurotransmitter can bind to several receptor subtypes
Inotropic (fast)
Metabotropic (slow)
either type of receptor can exite or inhibit a target cell
substance exists in presynaptic axon terminal
is released when AP reach axon terminals
receptors of the substance exist in presynaptic membrane
when experimentally applied, substance induces changes in postsynaptic cells
blocking the release of the substance prevents changes in postsynaptic cell
Glutamate
most prevalent excitatory neurotransmitter
plays a role in → cognition, learning and memory
binds to both ionotropic (NMDA, AMPA, kainate) and metabotropic (mGLUR1-18) receptors
Excitotoxicity → excess glutamate release resulting in damage/loss of neurons
plays a role in → Alzheimer’ disease, brain trauma,
seizure disorders, Parkinson's disease, stroke,
Huntington's disease, autism, schizophrenia
GABA (gamma-Aminobutyric acid)
most prevalent inhibitory neurotransmitter
binds to both ionotropic (GABAA, GABAC) and metabotropic (GABAB) receptors
Drugs based on enhancing GABA functions
Hypnotics, sedatives, tranquilizers, anticonvulsants
most well known → benzodiazepines (Diazepam=Valium)
Alchohol, cannabis
Used to treat pain, seizures, anxiety and migrane
Acetylcholine (ACh)
can be both inhibitory or excitatory
binds to ionotropic (nicotinic) and metabotropic (muscarinic) receptors
plays a role in → arousal, attention, learning & memory, and motivation
damage to cholinergic nerve cell bodies in the brain is associated with Alzheimer’ disease
Catecholamines
Dopamine (DA)
found in neurons in the →
Mesostrial pathway originating in the substantia nigra and projecting to the striatum
important in motor controll
Mesocorticolimbic pathway originating in the ventral tegmental area (VTA) and projecting to the cortex & limbic areas
important for reward and aversion, and learning
Abnormalities associated with schizophrenia and depression
Epinephrine/adrenaline
not in the brain
Norepinephrine (NE)
synthesized in the locus coeruleus (pons) and lateral tegmental system (midbrain)
binds to metabotropic (alpha 1, alpha 2; beta1, beta 2, beta 3) receptors
modulates mood, arousal, attention, behavioral flexibility and sexual behavior
Drugs → beta blocker (proparanolol)
reduces preformance anxiety
Indoleamines
Melatonin
sleep & wakefulness
Seratonin (5-hydroxythryptamine, 5-HT)
synthesized in 7 raphe nuclei, with dorsal raphe nucleus being the largest
role in sleep, mood, sexual behavior, depression and anxiety
Drugs → selective serotonin reuptake inhibitors (SSRI)
antidepressants (prozac)

a drug can be…
Agonist → mimics effects of usual neurotransmitter
Antagonist → binds receptor without activating it, thereby blocking the receptors from being activated
competitive vs non-competitive antagonist
Competitive antagonist → compounds that compete with agonists for the same receptor, but they do not exert an agonist effect themselves and so reduce the effect of any agonist present
Non-competitive antagonist → a compound that prevents an agonist from having a biological response by binding to a receptor protein
Inverse agonist → binds receptor and initiates opposite effect of usual neurotransmitter
Binding affinity → the degree of chemical attraction btw a ligand and a receptor
the more affinity a drug has for its receptor the lower the doses
neurotransmitters are low-affinity ligands allowing them to dissociate from receptors
Efficacy (intrinsic activity) → ability of a bound ligand to acctivate the receptor
Agonist have high efficacy
antagonists have low efficacy
ED50 value
effective dose for 50% of people recive the drug
allows comparison of potency of drugs
the higher the potency the more comparable the effects are at lower doses

Therapeutic index
separation between effective dose and toxic/lethal dose
determined by comparing ED50 with TD50 (toxic dose for 50% of individuals) or LD50 (lethal dose for 50% of individuals)
Antidepressants
Class of drugs used to treat symptoms of depression
Monoamine oxidase (MAO) inhibitors
prevent breakdown of monoamines at synapses
Tricyclic antidepressants
prevent the reuptake of serotonin and norepinephrine into presynaptic axon terminals
Selective serotonin reuptake inhibitors (SSRIs)
same mechanism as tricyclic antidepressants but with fewer side effects
Anxiolytics (tranquilizers)
class of drugs used to trreat anxiety disorders
Benzodiazepine agonists
act on GABAA receptors and enhanse the inhibitory effects of GABA
safe and and effective for short term use but long-term use is discouraged because of dependency and withdrawal effects.
Dendrite → communication with other neurons
Synapse → receiving part of a neuron
Axon → sending part of a neuron
Axon → Receiver
Synapse → Sender
Dendrite → Junction between two neurons
Myelin sheath → Insulating layer
Oligodendrocytes → remove debris from injured cells
Microglia → form myelin sheath around axons in peripheral nervous system
Astrocytes → form myelin sheath around axons in central nervous system
Swann cell → monitor neural activity
a) Olfactory
b) Trigeminal
c) Vestibulocochlear
d) Optic
e) Hypoglossal
lumbar → pelvic
sacral → base of spinal cord
coccyx → lower back
cervical → trunk
thoracic → neck
a) constricts the airways
b) stimulates digestion
c) constricts the blood vessels
d) stimulates salivation
e) accelerates heartbeat
f) constricts the pupils
anterior → tail end
lateral → middle
ventral → sideways
dorsal → front
posterior → back
medial → head end
Hypothalamus → hunger
Amygdala → emotion
Septum → emotion
Caudate nucleus → motor control
Putamen → motor control
Thalamus → sensory information
a) Blood-brain-barrier
b) Choroid plexus
c) Meninges
d) Cerebral hemispheres
e) Ventricular system of the brain
f) Carotid arteries
g) Spinal cord
a) pumping potassium and sodium into the cell
b) pumping sodium into the cell and potassium out of the cell
c) pumping potassium into the cell and sodium out of the cell
d) initiating an action potential
e) creating a positive net charge inside the cell
K+ → maintains resting potential
NA+ → prevents action potential
Cl- → required for action potential
1 → Voltage-gated Na+ channels open
2 → Na+ influx (into the cell)
3 → Voltage-gated Na+ channels close and voltage-gated K+ channels open
4 → K+ efflux (out of the cell)
5 → Voltage-gated K+ channels close
a) synapse
b) dendrites
c) axon hillock
d) cell body
a) Electrical conduction across the synaptic cleft
b) Reuptake of neurotransmitters
c) Influx of calcium at the presynaptic membrane
d) Binding to autoreceptors
1 → Postsynaptic receptors are activated
2 → NA+ enters the cell
3 → Neurotransmitters are released in the synapse
4 → The action potential is propagated to the axon terminal
1 → Ionotropic synapse
2 → Electric synapse
3 → Metabotropic synapse
a) Type of ion that flows across membrane
b) Type of ligand
c) Type of target cell
d) Type of presynaptic terminal
a) Type of target cell
b) Type of postsynaptic terminal
c) Type of ion channel modulated by the metabotropic receptor
d) Type of ligand
a) slow
b) changes shape
c) activates G-proteins
d) neurotransmitter binds directly to the ion receptor
e) alters chemical reactions
a) type of receptor it binds to
b) whether it is released in the brain or periphery
c) concentration of ACh
Q5 - Match the neurotransmitter with region of synthesis
serotonin → locus coeruleus
norepinephrine → substantia nigra
dopamine → nucleus basalis
acetylcholine → raphe nuclei
GABA → Benzodiazepines
Serotonin → SSRI (antidepressants)
Norepinephrine → Propranolol
Opioids → Morphine
The higher the affinity of the drug, the - lower - the concentration of the drug.
a) low ED50
b) low affinity
c) high LD50
d) high TD50
a) Stimulate norepinephrine receptors
b) Decrease the synthesis of serotonin
c) Increase serotonin availability
d) Upregulation of postsynaptic serotonin receptors
a) pumping potassium and sodium into the cell
b) pumping sodium into the cell and potassium out of the cell
c) pumping potassium into the cell and sodium out of the cell
d) initiating an action potential
a) synapse
b) dendrites
c) axon hillock
d) cell body
a) impulses
b) axons
c) synapses
d) nerves
a) meninges
b) myelin
c) dura mater
d) pia mater
a) blood-brain-barrier
b) carotid arteries
c) spinal cord
d) ventricular system
e) meninges