4.2 Electron Transport Chain
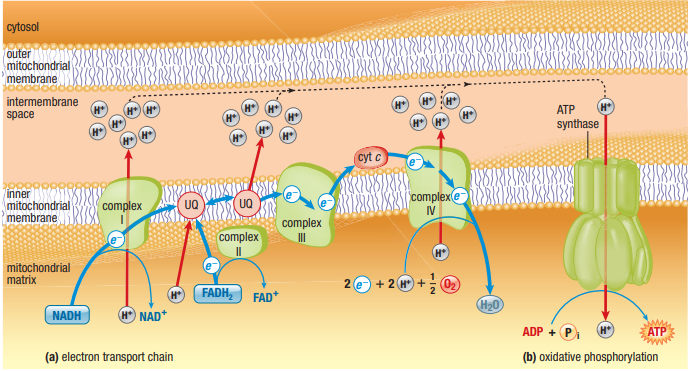
Electrons (with their hydrogens) are passed from one molecule to the next in order of increasing electronegativity
Energy is released with each transfer
O2 is the final electron acceptor
This energy is used to pump H+ across from matrix to intermembrane space, setting up an electrochemical gradient of protons
- Proton gradient
- Proton-motive force (back down to low electronegativity through ATP synthase)
- Will be used to form ATP through OP
IN DETAIL
- 1st Oxidation
- NADH dehydrogenase oxdized NADH to NAD+
- Energy released drives H+ from matrix into intermembrane space
- 2nd & 3rd Oxidation
- Succinate dehydrogenase oxdizes FADH2
- Ubiquinone oxidizes NADH dehydrogenase ans Succinate dehydrogenase
- UQ is hydrophobic, shuttles within bilayer
- uses electrons to move H+ across membrane
- 4th & 5th Oxidation
- Cytochrome complex oxidizes UQ
- Cyt C oxidizes Cytochrome complex
- cyt c is hydrohphillic, shuttles in the intermembrane space
- 6th & 7th Oxidation
- Cytochrome oxidase oxidizes Cyt C
- Oxygen oxidizes cytochrome oxidase
- energy released pumps H+ across the membrane
- 8th Reaction
- Proton gradient sets up proton-motive force
- only way back is with ATP synthase
- Flow of protons allows the energy to synthesize ATP
- Chemiosmosis
- OP