Chapter 5: Reactivity Centers: Functional Groups
Organic Chemistry for Dummies 2nd Edition by Arthur Winter, PhD
Chapter 5: Reactivity Centers: Functional Groups
__Functional groups:__ carbons forming stable bonds to other non-carbon atoms, forming reactive centers.
==Hydrocarbons==
Hydrocarbons: molecules that contain just hydrogen and carbon.
- Includes:
- __Alkanes:__ contain only single bonds and are generally not considered a functional group.
- Inert under most conditions
- __Alkenes:__ molecules containing carbon-carbon double bonds)
- __Alkynes:__ molecules containing carbon-carbon triple bonds
- __Aromatics__: double-bond-containing ring systems.
Simple hydrocarbons: usually cheap and commercially availble because they’re found as components in crude oil.
@@Double the fun: The alkenes@@
General form of an alkenes:
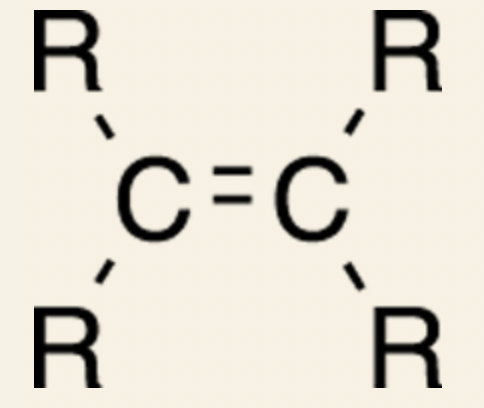
The R is an abbreviation for the rest of the molecule.
- Most often implies a hydrogen atom or a hydrocarbon group
Alkenes: found in natural products (compounds isolated from living organisms).
Alkenes are also used commercially.
- Ethylene polymerizes to make polyetylene (type of plastic used in grocery bags, milk bottles, etc.)
- __Polymerize:__ combines many small units to make large molecules.
Alkenes can also be arrnaged into rings like in cyclopropene (but this is generally not a favorable shape for them to stay in).
They are important to organic chemistry because they are the “go-betweens” in the synthesis of complex molecules.
The names of alkenes generally end with the suffix –ene.
@@Alkynes of fun@@
General form of alkynes:
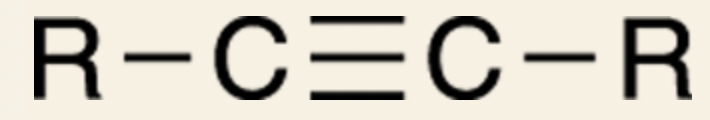
- The simplest alkyne is, acetylene, which has the 2 R groups substituted with hydrogen.
- It is a gas that is used as a welding fuel.
- Alkynes are less common than alkenes in nature, but there are some out there.
- Alkynes prefer to have bond angles of 180 degrees, where the triple bond and the two R groups on either side lie in a straight line.
- Triple bonds do not stretch much, so molecules with triple bonds are generally unstable in rings of fewer than eight carbons.
- Alkyne names end with the suffix –yne.
@@Smelly compounds: The aromatics@@
- They are also called ‘arenes’.
- __Aromatics:__ consist of rings containing alternating double bonds.
- Example: Benzene (a six-carbon ring with three alternating double bonds).
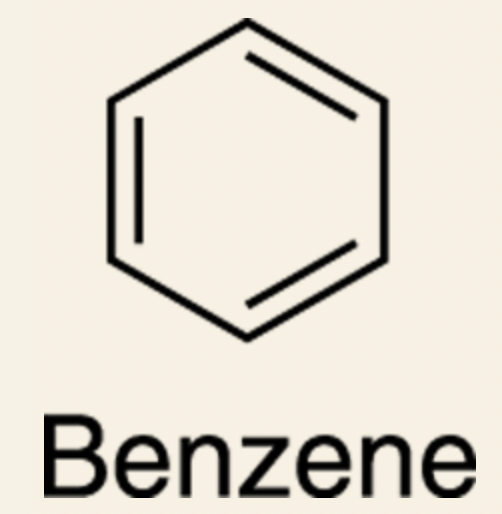
- Aromatics have a special property that makes them significantly more stable and less reactive than alkenes.
- They are called aromatics because the first of these compounds to be discovered (before their structures were determined) had funky smells.
- Many compounds made by living organisms contain aromatic rings.
==Singly Bonded Heteroatoms==
- Heteroatoms: atoms other than carbon or hydrogen.
- Include such important atoms as the halogens, oxygen, and sulfur.
- These are the components of the halide, alcohol, ether, and thiol functional groups.
@@Happy halides@@
- Halides are organic compounds that contain one or more halogens.
- 4 halogens that you frequently see in organic compounds: fluorine, chlorine, bromine, and iodine.
- General form of halides:
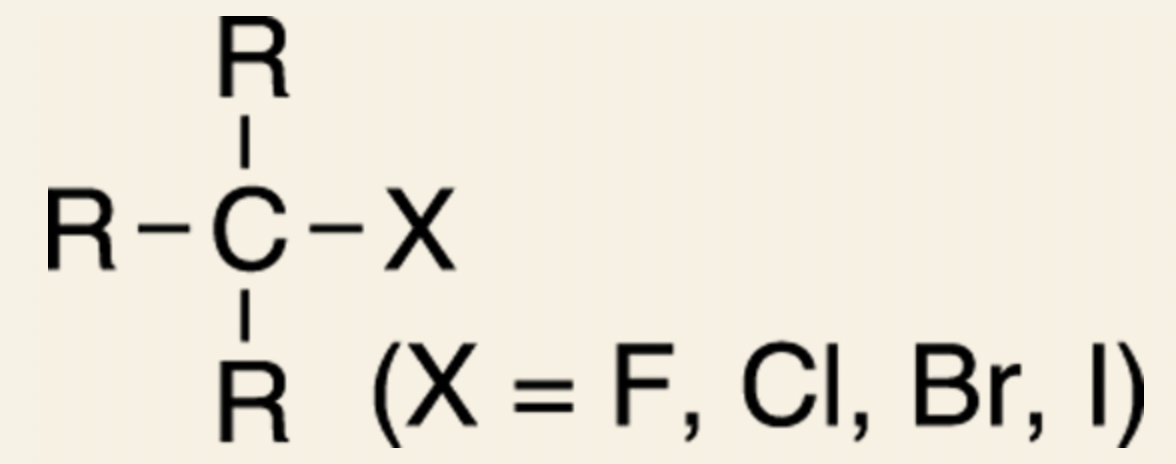
- Halides are not common in the natural world., and when they are found, they’re often in compounds that are toxins.
- Commercially, halides are used as propellants in aerosol cans such as hairspray and spray paint, solvents such as chloroform, and as refrigerants.
- Also as insecticides such as DDT. This is banned in the United States because it has caused unwanted harmful side effects in wildlife and in humans.
@@For rubbing and drinking: Alcohols@@
- Alcohols consist of the general formula R-OH.
- The names end with the suffix -ol.
- Some examples include: ethanol (alcohol found in beer, wine, and liquor), isopropanol (rubbing alcohol), and ethlene glycol (used in antifreeze).
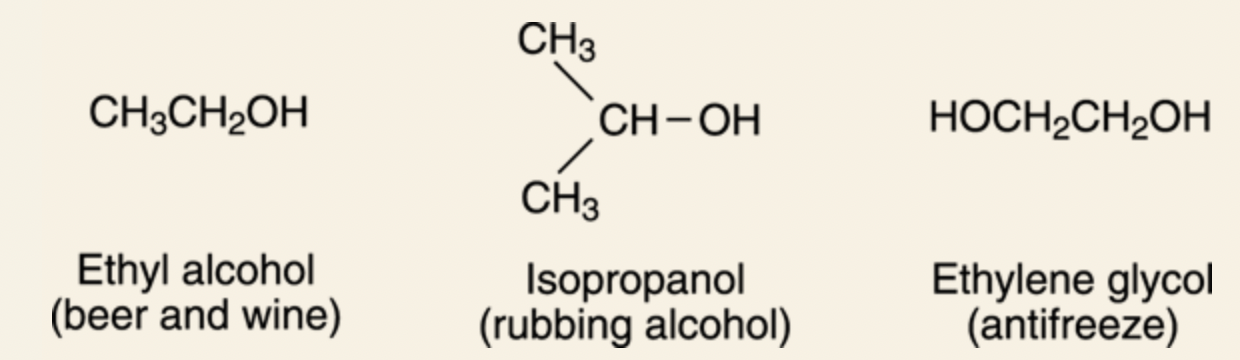
- Alcohols are also found in natural products.
- Like, sugar contains many -OH groups.
@@What stinks? Thiols@@
- They are foul-smelling compounds and contain the formula R-SH.
- Thiols: sulfur analog of alcohols/in other hideously unpleasant compounds.
- Like: flatulence, garlic, sewage, skunk spray, etc.
- \