Chem Organic
Organic chemistry includes compounds of carbon (hydrocarbons, or contain carbon carbon bonds)
EXCLUDES: oxides, CO2, ionic compounds of carbon based ions such as carbonate, cyanide, and carbide ions, such as H2CO3, HCN, and NaC.
9.1 Fossil fuels
Hydrocarbons: only contain carbon and hydrogen, covalent bonds.
Primary sources for hydrocarbons are coal, oil and natural gases.
Diplomas: know what certain organic compounds are used for. Ex. methanol for windshield fluid, anti freeze. Methane for warming houses. Etc.
9.2 Alkanes from Natural Gas
Natural gas has varying composition, mostly methane (CH4)
raw natural gas: seen as sour due to impurities.
sweet natural gas has NO hydrogen sulfide. (as carbon increases = boiling points increase = more LD forces)
How to name Alkanes: (# of carbons) + ane (REMEMBER THE ALKANE FAMILY OF ORGANIC COMPOUNDS) ((THE METH, ETH, PROP PARTS ARE THE ALK PARTS) 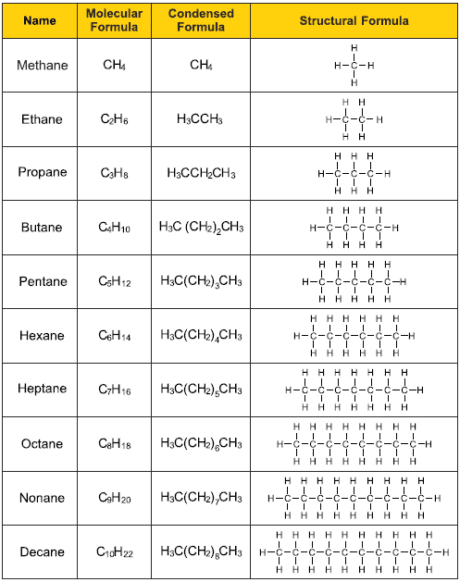
are a hydrocarbon with SINGLE bonds between carbons.
Are a homologous series, differ by repeating units FORMULA: CnH2n+2
Alkanes with 5+ carbons = LIQUID STATE
known as SATURATED hydrocarbons, maximum number of hydrogen atoms bound to each carbon.
Molecular Formula: type and number of Atoms. Ex. Methane = CH4 (g)
Structural Formula: Connectivity of atoms in a compounds (shown above)
Condensed Structural Formula: connecting groups of atoms in a compound. Ex. Propane: CH3 —CH2 — CH3
line structural Formula: Position of carbon atoms as intersections, ends of bonding lines. Ex. Hexane : /\/\/,, for dialkyl names on the same branch make an X Ex. for 2,2-dimethylpentane,
 BUT FOR stuff like ethylpentane
BUT FOR stuff like ethylpentane
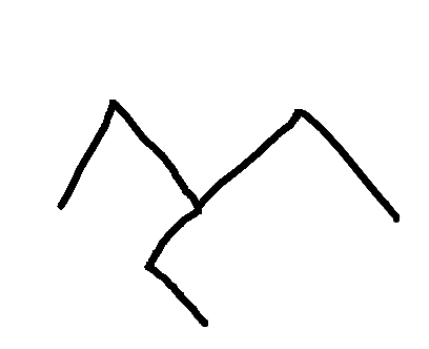
Structural Isomers: SAME molecular formula, DIFF structures Ex. butane and methylpropane.
Alkyl Branches: branch is any group of atoms NOT apart of parent / main structure of the molecule. Consists of only Carbon and Hydrogen Atoms.
Steps for Naming Branched Alkanes
find the longest chain of carbon atoms, TWO ENDS (a branch cannot be seen on 1 or 4)
counting from the branch closest to the end of the parent, number their locations going from left to right or right to left depending on said branch.
if identical branches indicate # of branches (di, tri, tetra, penta), IF NOT IDENTICAL, writing from left to right by ALPHABETICAL ORDER.
write the following: number of location (if its’ the only branch / its the only possibility its not necessary, otherwise carry on) DASH - alk name-yl (ex. meth-yl) ,, number of location DASH ALK name-yl (each branch name ALPHABETICAL) parent chain alk name-ane.
Cycloalkanes:
Structure = a closed ring
IF carbon-carbon bonds are single = cycloalkane.
Named via the cyclo in front of alkane name.
Higher boiling points than its respective alkane
smaller compounds MORE REACTIVE then respective alkane due to strained structure
numbering starts AT one of the branches, smallest number reigns supreme, going clockwise/counterclockwise accordingly ( if only one branch don’t need to add number)
9.3 Alkenes and Alkynes Cracking Natural gases
Alkenes (double bonded carbons)
Formula: CnH2n (as well as cycloalkanes)
Isomers: Cycloalkanes (larger than propene)
homogolous
UNSATURATED (fever hydrogen atoms attached to carbon chain) (if you add a hydrogen = saturated hydrocarbon, HYDROGENATION)
similar to alkanes’ properties
more reactive due to double bond
Alkynes (triple bonded carbons)
Formula: CnH2n-2 (as well as Cycloalkenes)
Isomers: cycloalkenes (larger than propyne)
homologous
UNSATURATED (fewer hydrogen atoms attached to carbon chain)
similar to Alkanes properties
small chain Alkynes have HIGHER boiling points than alkanes (due to better stacking/can be closer together)
highly reactive due to triple bond
LEAST TO MOST REACTIVE
alkanes, alkenes, alkynes
Naming Alkenes and Alkynes
parent chain longest + needs the multi bond (REMEMBER: 2 carbon-carbon = alkene, 3 carbon-carbon = alkyne)
number the branches by the end closest to the carbon-carbon BOND, to determine branch numbers.
Instead of ALK name-ane, you will write the lowest number of the carbon-carbon bond closest to the end of the parent chain in the middle followed by ene/yne. (but-1-yne)
You would write as the following: # of location-alkyl branch/s (alphabetical, if only one and the only possibility don’t write location #) DASH - , ALK name-yl, alk-# nearest to end -ene/yne (can have 1 bond and 2 bond Ex. 3-methylbut-1-ene)
Cycloalkenes and Cycloalkynes:
counting starts with the lowest number at the multi-carbon-carbon bond, so 1 and 2.
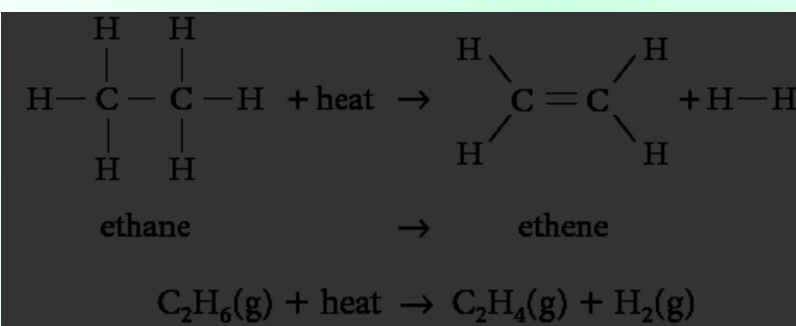
Ethane Cracking / DEhydrogenation
An industrial process in which large hydrocarbons are broken down into smaller hydrocarbons.
9.4 Aromatics
Benzene and structures based on benzenes are called Aromatics due to characteristic aroma.
Hydrocarbon ring with 6 untouched valence electrons shared equally to 6 carbon atoms.
This Phenomena is known as RESONANCE
Aliphatics:
Includes alkanes, alkenes, Alkynes + cyclo counterparts
colourless, odourless
IF added with Benzene, it becomes an aromatic
Naming Aromatics
As a parent chain: just write alk-name-yl(if it has a branch)benzene (use the number thing like alkanes and such IF more than one branch / more possibilities) ,, NUMBERING goes by alphabetical order of the branches and check all possibilities and go with the LOWEST number
As a branch: considered a phenyl group. TO KNOW ITS A PHENYL: if it ISN’T connected to an end, it usually is a branch.
9.5 Crude Oil Refining
Physical Processes of Oil Refining | Chemical Processes of Oil Refining | |
|---|---|---|
|
|
9.6 Complete and Incomplete Combustion Reactions
Complete Combustion: a hydrocarbon reacts with (excess) oxygen to produce CO2 and water vapour as the ONLY CHEMICAL products.
Incomplete Combustion: a hydrocarbon reacts with (in-sufficient) oxygen to produce products OTHER THAN CO2, water vapour Ex. carbon monoxide, carbon (soot)