Chapter 14:Salt and identification of Ions
Salt
A salt is a compound that is formed when the hydrogen of an atom in an acid is replaced by a metal.
It is formed by the neutralisation reaction between an acid and a base
[ ] Precipitation
[ ] Titration
Reactions
- Metal and acid➡️ salt and water
- [ ] zinc + sulphuric acid➡️ zinc sulphate+ Hydrogen
2. Metal oxide + acid ➡️ salt and water
- [ ] Copper(II) oxide + sulphuric acid➡️ copper sulphate+ water
3• Metal hydroxide + acid ➡️salt and water
- [ ] Potassium hydroxide+ hydrochloric acid ➡️ potassium chloride + water
4• Metal carbonate + acid ➡️salt+water+ carbon dioxide
- [ ] Zinc carbonate+ nitric acid ➡️ zinc nitrate +water+ carbon dioxide
Solubility
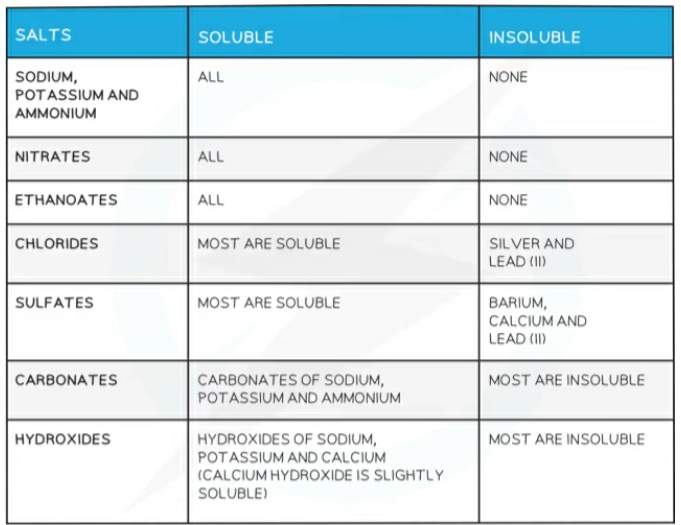
Preparation of soluble salts
- Metal and acid
- Metal oxide and acid
- Metal hydroxide and acid(titration)
- Metal carbonate and acid
Preparation of insoluble salts
Precipitation
[ ] Soluble salt+ soluble salt ➡️ insoluble salt
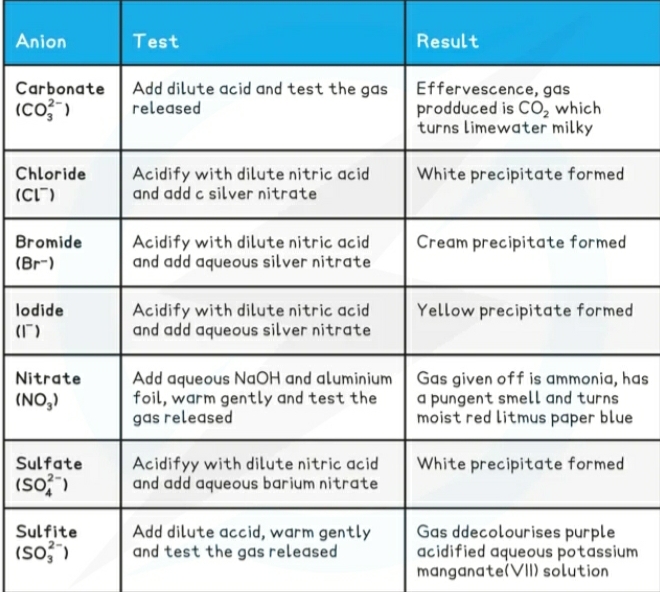
Identification of ions
- Cations (postive ions) they travel to the negative terminal Cathode during electrolysis.
- Anions (negative ions) they travel to the positive terminal Anode during electrolysis
Identification of gases
| Hydrogen | Pops with a lighted splinter |
|---|---|
| Oxygen | Relights a glowing splinter |
| Carbon dioxide | Turns limewater milky |
| Chlorine | Turns moist blue litmus red and then bleaches it |
| Ammonia | Turns moist red litmus paper blue |
| Sulphur Dioxide | Turns acidified potassium dichromate from yellow to green |