Chemistry IGCSE
Exam
General Notes
Spelling does not matter as long as meaning is not changed
If done know what observation says bubbles
What should the person do to make the experiment better: Repeat
Paper 1
2 Hours
Paper 2
1 hour 15 min
Ammonia in NH3 and Ammonium is NH4+
Inorganic Chemistry
(2a)
(2g) Acids, bases, and salt preparations
2.34 know the general rules for predicting the solubility of ionic compounds in water: • common sodium, potassium and ammonium compounds are soluble • all nitrates are soluble • common chlorides are soluble, except those of silver and lead(II) • common sulfates are soluble, except for those of barium, calcium and lead(II) • common carbonates are insoluble, except for those of sodium, potassium and ammonium • common hydroxides are insoluble except for those of sodium, potassium and calcium (calcium hydroxide is slightly soluble).
2.35 understand acids and bases in terms of proton transfer
Acids: proton donors because they have H+ ions
Bases: proton acceptors as they ionize
2.36 understand that an acid is a proton donor and a base is a proton acceptor
2.37 describe the reactions of hydrochloric acid, sulfuric acid and nitric acid with metals, bases and metal carbonates (excluding the reactions between nitric acid and metals) to form salts
2.38 know that metal oxides, metal hydroxides and ammonia can act as bases, and that alkalis are bases that are soluble in water
2.39 describe an experiment to prepare a pure, dry sample of a soluble salt, starting from an insoluble reactant
Preparation of soluble salt from insoluble base:
Mix the insoluble base and the acid
Add excess solid (to ensure all the acid has reacted)
Filter to remove the excess solid (you are not left with a solution of your product dissolved in water)
Evaporate most of the water until crystalls begin to appear
Leave to allow to crystallize slowly
Dry the crystals in warm oven (dry oven to dry faster)
It is important to know that excess solid is added to make sure ALL acid has reacted
2.40C describe an experiment to prepare a pure, dry sample of a soluble salt, starting from an acid and alkali
Preparation of a soluble salt from an acid and alkali:
Use a pipette to transfer the acid to a conical flask
Add a few drops of indicator
Place the alkali solution in the burette
Add the alkali from the burette into the acid, until the solution is neutral. Record the volume.
Repeat but without indicator using volume from step 4 (indicator will be impurity)
Evaporate most of the water until crystals begin to appear
Leave to allow to crystallise slowly
Dry the crystals in a warm oven
Step 1-4: to find the exact volume needed to netralise which is shown through the indicator
2.41C describe an experiment to prepare a pure, dry sample of an insoluble salt, starting from two soluble reactants
Preparation of an insoluble salt from two soluble (solution):
Mix the two solutions, a precipitate will form (the precipitate is the insoluble product)
Filter the mixture to separate the solid
Wash the solid with distilled water to remove impurities
Leave the solid on filter paper to dry
Dry the crystals in a warm oven
Allow to dry to constant mass
2.42 practical: prepare a sample of pure, dry hydrated copper(II) sulfate crystals starting from copper(II) oxide
Method:
2.43C practical: prepare a sample of pure, dry lead(II) sulfate
Organic Chemistry
(4a) Introduction
4.1. Know that a hydrocarbon is a compound of hydrogen and carbon only
Matching its name a hydrocarbon is a compound that is made of of however many hydrogen and carbon ONLY
4.2. Understand how to represent organic molecules using empirical formulae, molecular formulae, general formulae, structural formulae, and displayed formulae
Empirical Formula: The simplest possible ratio of the atoms/elements in the molecule
e.g. Ethane is C2H6 but its empirical formula is CH3
Molecular Formula: The actual “formula“ of the molecule, shows the actual number of atoms
e.g. Butene is C4H8
General Formula: The ratio of the atoms in a family of compounds in terms of “n“
Alkandes general formula is CnH2n+2 while Alkenes general formula is CnH2n
Structural Formula: Displays enough information to make the structure clear but does not show the bonds

Displayed Formula: shows the spatial arrangement of all atoms, molecule, and bonds

4.3. Know what is meant by the terms homologous series, functional group and isomerism
Homologous Series: A collection/series/family of organic compounds that have the same chemical properties due to having the same functional group
All compounds within the same homologous series have:
The same general formula
The same functional group
Similar chemical properties
Physical properties that change gradient
The difference between each consecutive member is CH2
Functional Group: A group of atoms bonded together in a very specific arrangement that influences what homologous series they are a a part of (the part of the compoud that makes it that specific homologous series/ the characteristic that makes you part of the homologous series)

Isomer: Compounds that have the same molecular formula but different displayed formula
4.4. Understand how to name compounds relevant to this specification using the rules of International Union of Pure and Applied Chemistry (IUPAC) nomenclature students will be expected to name compounds containing up to six carbon atoms
Naming an organic compound has two parts
The number of carbon atoms present in the compound
Name | Number of Carbons |
|---|---|
Meth… | 1 |
Eth… | 2 |
Prop… | 3 |
But… | 4 |
Pent… | 5 |
Hex… | 6 |
The homologous series it is part of
Name | Homologous Series | Functional Group |
|---|---|---|
…ane | Alkane | - |
…ene | Alkene | C = C Bond |
…anol | Alcohol | - OH |
…anoic acid | Carboxylic Acid | - C = O - OH |
…amine | Amine | -NH2 |
…yl …anoate | Ester | - C = O - O - |
4.5. understand how to write the possible structural and displayed formulae of an organic molecule given its molecular formula
If the molecular formula of a compound is C2H6, structural formula is CH3CH3
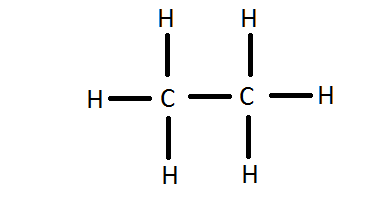
4.6. understand how to classify reactions of organic compounds as substitution, addition, and combustion knowledge of reaction mechanisms is not required
Substitution: a reaction that takes place when one functional group is replaced by another
Methane reacts with bromine under ultraviolet light
CH4 + Br2 → CH3Br + HB
Methane + Bromine → Bromomethane + Hydrogen Bromide
Addition: a reaction that takes place when two or more molecules combine to form a larger molecule with no other product
Bromine will react with ethene and the bromine molecule will react and add across the double bond of the ethene
C2H4 + Br2 → C2H4Br2
Ethene + Bromine → Dibromoethane
Combustion: a reaction where an organic substance reacts with oxygen to form carbon dioxide and water
Complete Combustion: If there is an unlimited supply of oxygen, the products are carbon dioxide and water:
CH4 + 2O2 → CO2 + 2H2O
C3H8 + 5O2 → 3CO2 + 4H2O
Incomplete combustion: If there is a limited supply of oxygen, the products are carbon monoxide and water:
CH4 + O2 → CO + 2H2O
(4b) Crude Oil
4.7. know that crude oil is a mixture of hydrocarbons
Crude Oil: a mixture of different hydrocarbons called fractions (together the substance is not very useful)
A finite resource
4.8. describe how the industrial process of fractional distillation separates crude oil into fractions
The fractions in crude oil are separated through the process of fractional distillation.
The size and length of each hydrocarbon molecule determine which fraction it is separated into
Inside the fractional column, the crude oil is heated
The oil evaporates up the column
The oil condenses as it goes up the column
The hydrocarbons with the highest boiling point condense first, at the bottom of the column (bigger)
Hydrocarbons with the lowest boiling point condense last, at the top of the column (small)
4.9. know the names and uses of the main fractions obtained from crude oil: refinery gases, gasoline, kerosene, diesel, fuel oil and bitumen
From Top to Bottom (low BP to high BP)
Refinery Gases
Domestic Heating
Cooking
Gasoline/Petrol:
Fuel for cars
Kerosene:
Aircratf fuel
Diesel:
Fuel for cars and buses
Fuel Oil:
Fuel for large ships and power stations
Bitumen (Bottom): Bitume for roads and roofs

4.10 know the trend in colour, boiling point and viscosity of the main fractions
Viscosity: how easily can the liquid flow. The number of hydrocarbons increases → attraction between hydrocarbons increases → liquid becomes more vicious
Colour: As carbon chain length increases → colour get darker (thicker)
MP and BP: As molecules get larger → intermolecular attraction becomes greater → more heat is needed to seperate the molecules → increased BP and MP
Volatility: tendency of a substance to vaporise. Increased molecular size → hydrocarbon liquid becomes less volatile (because the intermolecular attraction increases with increased size of molecules)
Sum: As you go up the column BP and viscosity decreases
4.11 know that a fuel is a substance that, when burned, releases heat energy
Fuel: substance that releases heat energy when burned