Nucleotides
Nucleotides are the monomers, which are joined in condensation reactions to form a polynucleotide strand in DNA or RNA. Nucleotides are also important components of other important biological molecules such as ATP and some coenzymes e.g. NAD.
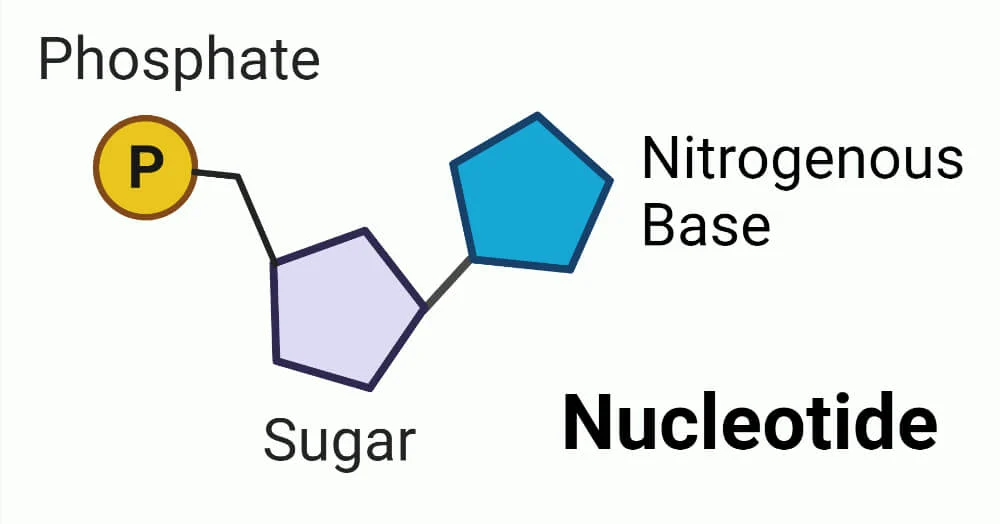
A phosphate, pentose sugar, and nitrogenous base join together via condensation reaction to form nucleotides. There are 2 types of nitrogenous base. Purines (adenine and guanine) are larger and have a double ring structure. Pyrimidines (cytosine, thymine and uracil) are smaller and have a single ring structure.
Polynucleotides
DNA and RNA are polymers of nucleotide monomers. Two nucleotides join together in a condensation reaction between the phosphate of one nucleotide and the pentose sugar of the next, to form a phosphodiester bond. The process is repeated many times to form a polynucleotide strand. The chain of nucleotides linked by phosphodiester bonds forms the sugar-phosphate backbone that the nitrogenous bases project out from.
ATP
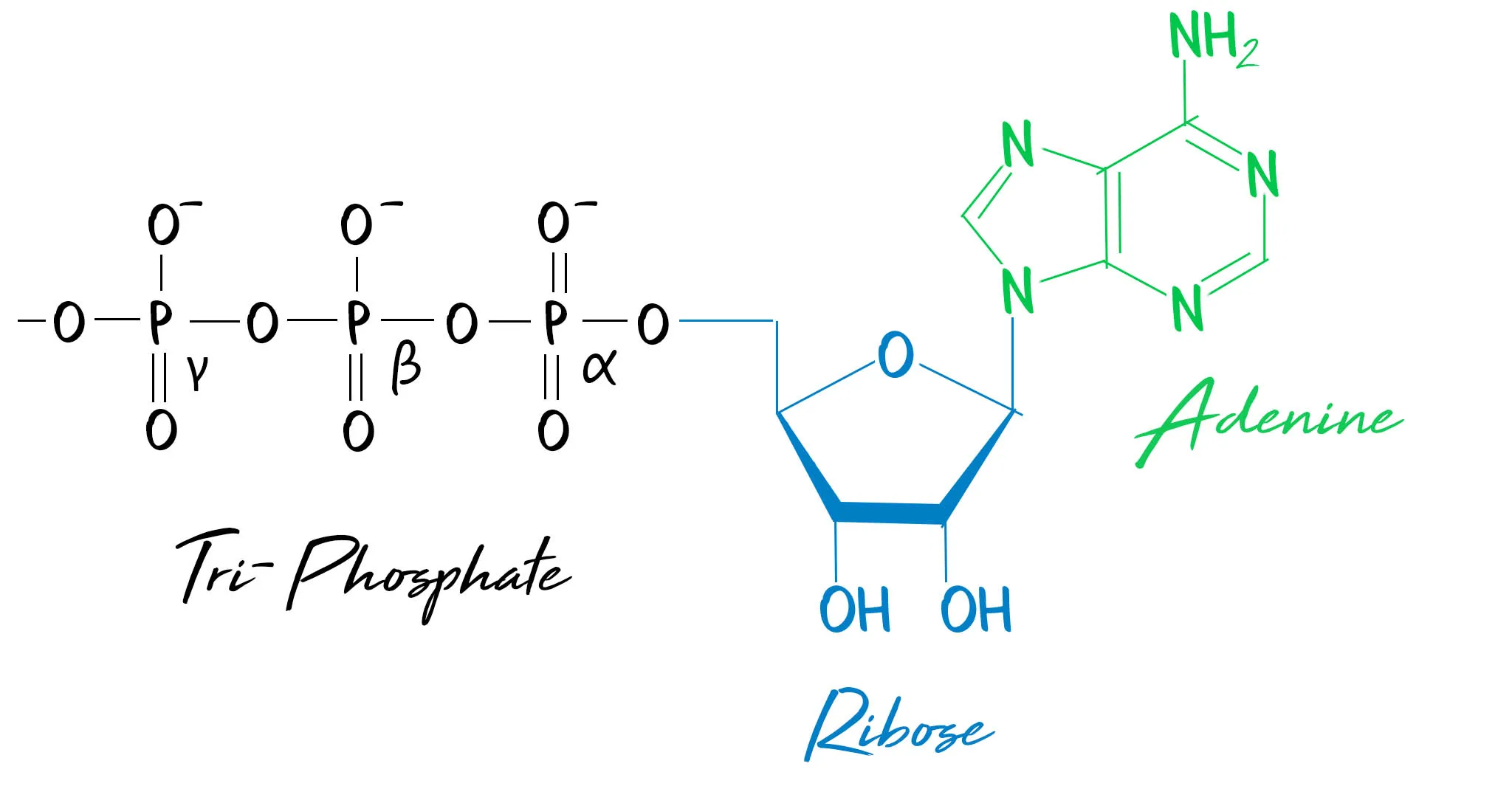
Adenosine triphosphate (ATP) is a phosphorylated nucleotide. It is composed of ribose and adenine, which form adenosine, with 3 phosphate groups attached.
ATP can be hydrolysed to release 30.6 KJmol-1 and form adenosine diphosphate (ADP). It takes the same amount of energy to re-synthesise ATP by the addition of a phosphate to ADP by phosphorylation. ATP is referred to as the universal energy carrier:
Universal: ATP is found in all cells/organisms.
Energy: Acts as an immediate source of energy for biological processes.
Carrier: ATP is small and water-soluble so is easily transported around the cell. Also links energy releasing reactions (respiration) to energy requiring processes e.g. active transport.
Other important nucleotides:
Coenzyme A (CoA): carries acetate groups in respiration.
Flavine Adenine dinucleotide (FAD): Hydrogen carrier in respiration.
Nicotinamide adenine dinucleotide (NAD): Hydrogen carrier in respiration.