Addition Reactions of Alkenes and Markovnikov's Rule
Addition Reactions of Alkenes
General form:
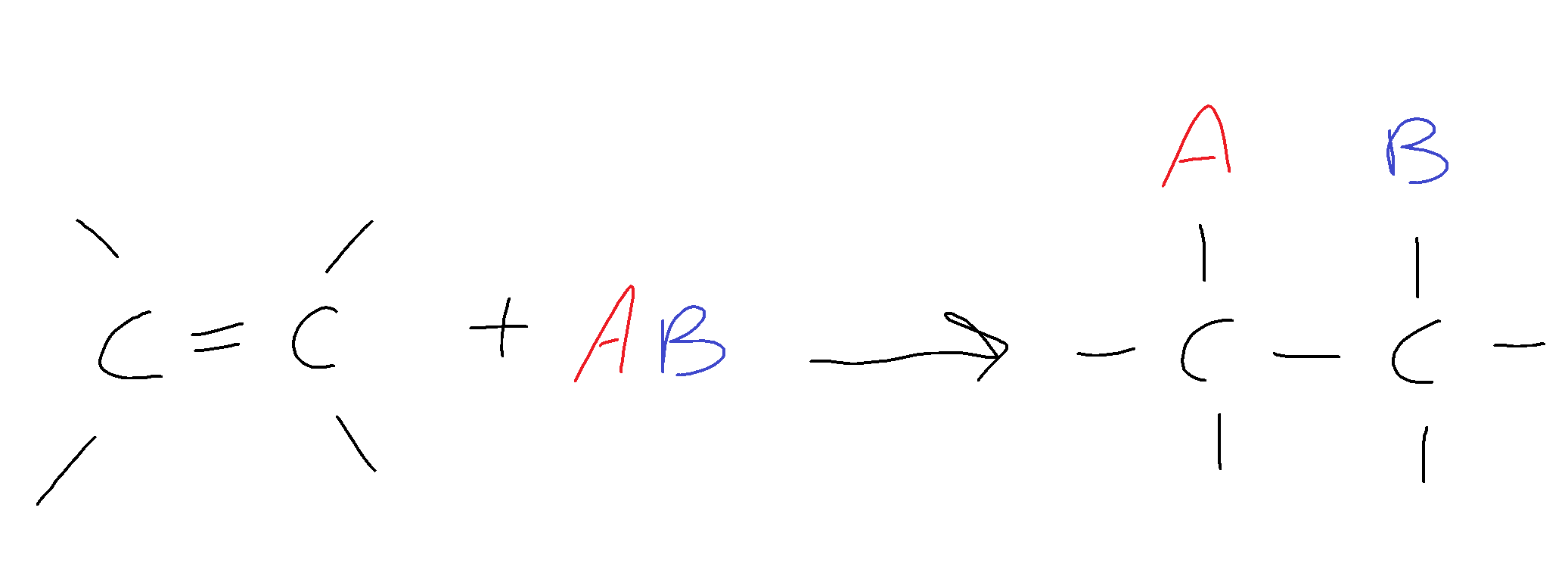
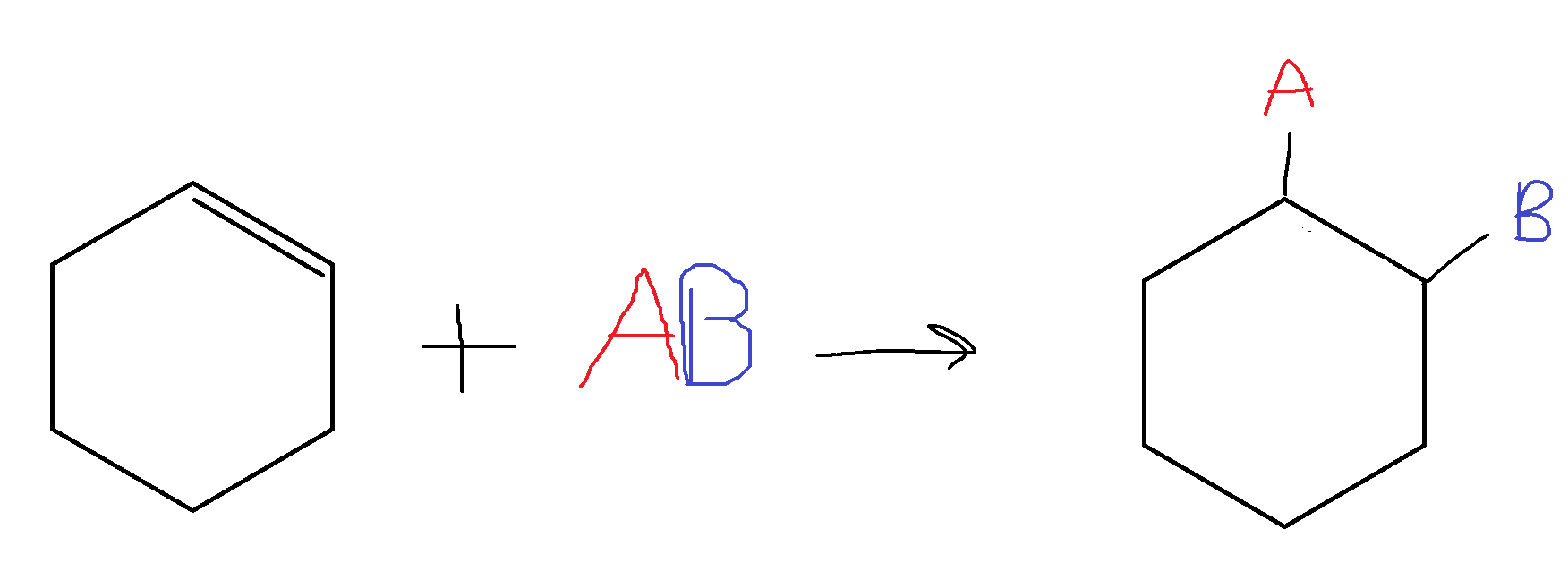
For a cyclic compound:
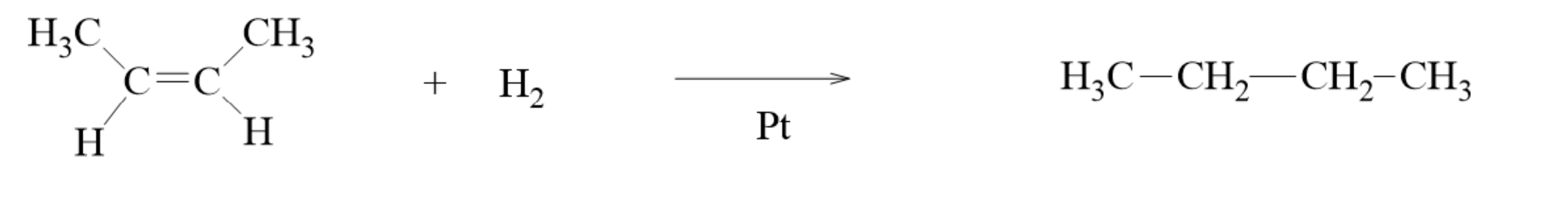
Catalytic Hydrogenation
- - the process of adding hydrogen to carbon-carbon double bonds to make the molecule more saturated in hydrogens
- This can be done by passing hydrogen gas through a solution of an alkene in the presence of a metal catalyst such as Pd, Pt, or Ni.
- The product is an alkane.
- having either the cis- or trans- isomer of the molecule gives you the same product
- The carbons in the double bond go from sp2 hybridized to sp3 hybridized
- This reaction occurs because sigma bonds tend to be stronger than pi bonds, so the pi bond in the double bond is broken easily.
- This process is enthalpy favored because the alkane’s single bonds are at a lower energy level than the alkene’s double bond(s).
- This process is not entropy favored because you are going from 2 reactants to 1 product.
- This reaction is enthalpy driven
Addition of HCl or HBr to form a Haloalkane
- The carbons in the double bond go from sp2 hybridized to sp3 hybridized
- This reaction occurs because sigma bonds tend to be stronger than pi bonds, so the pi bond in the double bond is broken easily.
- This process is enthalpy favored because the alkane’s single bonds are at a lower energy level than the alkene’s double bond(s).
- This process is not entropy favored because you are going from 2 reactants to 1 product.
- This reaction is enthalpy driven

Addition of Water to an Acid Catalyst to form an Alcohol
- the catalyst is the sulfuric acid
- H corresponds to A and OH corresponds to B in the general form
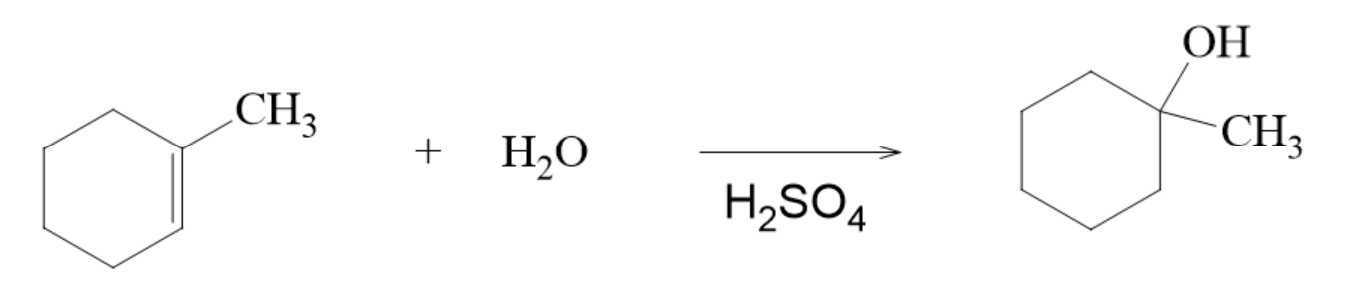
Markovnikov’s Rule
- The formation of haloalkane and alcohol are reactions- meaning the reaction favors the formation/breaking of a bond at a specific location over another
- In other words- “Hydrogen goes where hydrogen is”
- For example, the below reaction has two possible products:
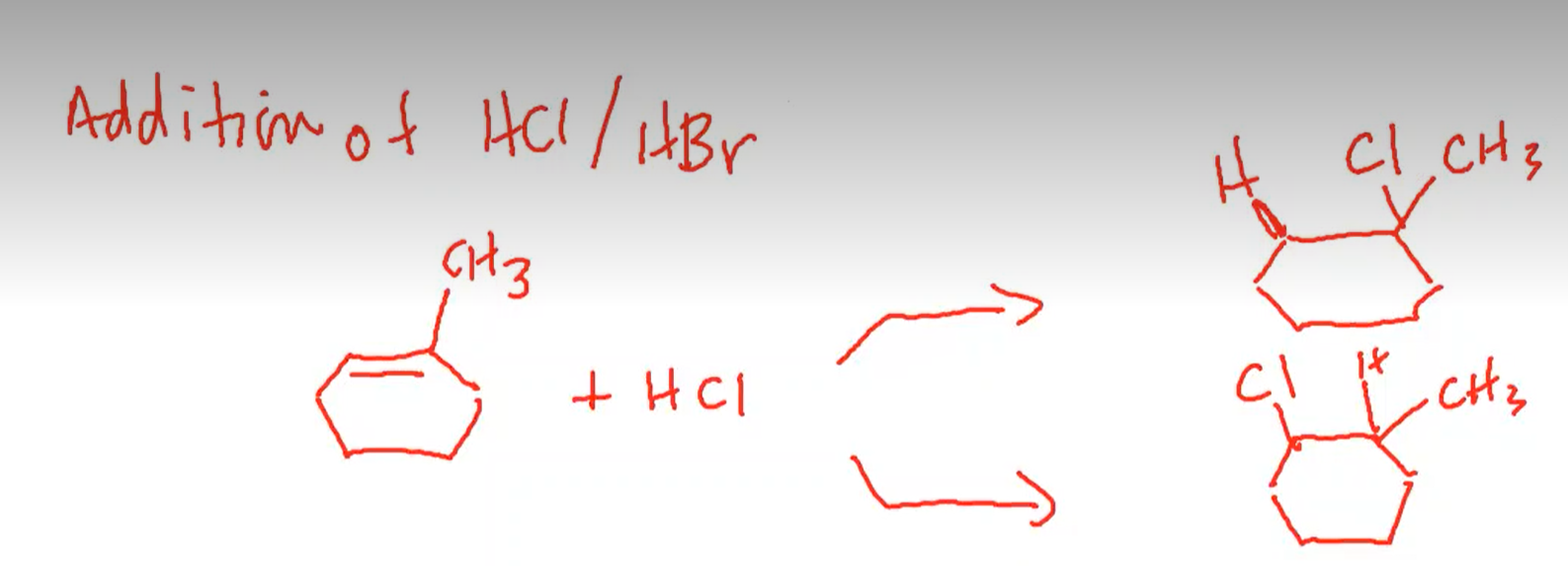
- The product at the top is favored because it is more substituted
- why is the more substituted product favored?- there’s a carbocation intermediate in the reaction mechanism
## Carbocation Stability
Order of carbocation stability:
- tertiary carbocations- carbocations that are attached to three carbon atoms/groups
- secondary carbocations- carbocations that are attached to two carbon atoms/groups
- primary carbocations- carbocations that are attached to one carbon atom/group
- methyl carbocations- a free-standing methyl group where the carbon has a positive charge
- Knowing this, we can see that the top product is favored because it allows for the formation of an intermediate tertiary carbocation that is more stable.
- because it’s more stable, this intermediate tertiary carbocation forms more rapidly
Mechanism of addition of HCl to an Alkene
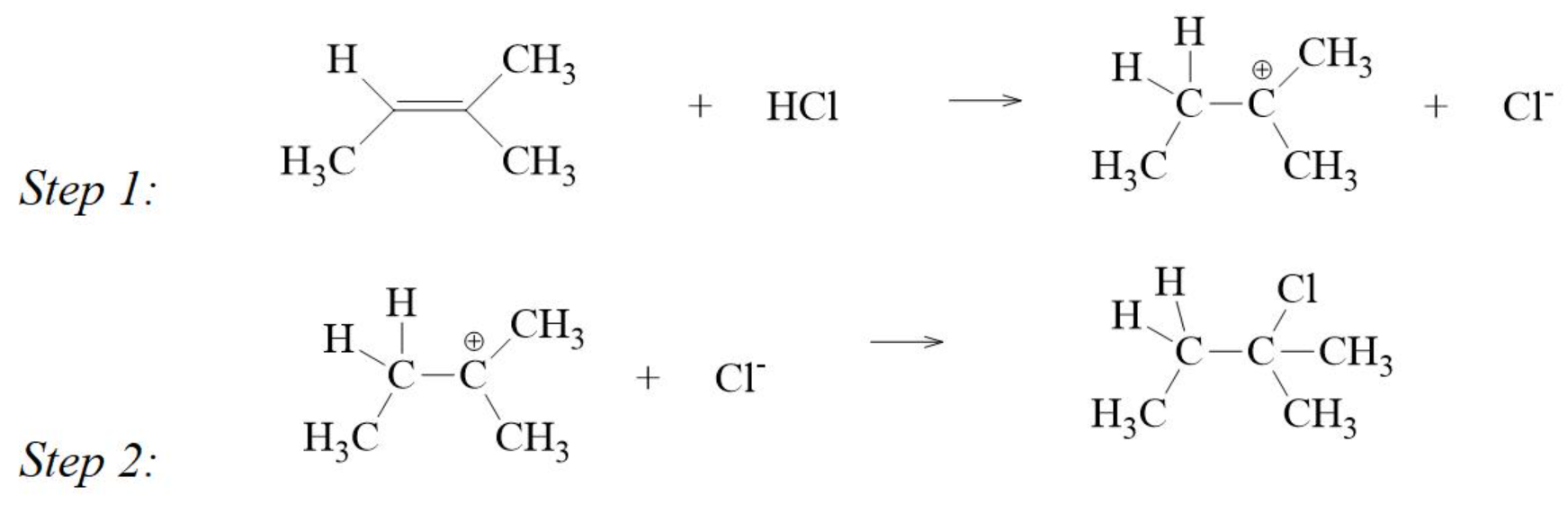
- step 1 is the slower step and determines the rate of reaction
- the addition of water to form an alcohol has a similar mechanism, and the more substituted alcohol is the major product