States of matter
In a physical change, no new substance has formed.
Examples include when the substance:
changes shape
breaks into smaller pieces
is dissolved
is mixed with another substance
changes state
In a chemical change, a new substance has formed.
Chemical changes include:
cooking
burning things
fruit ripening
dead animals or fruit rotting
explosion
Signs of chemical change
permanent colour change
gas production
precipitate
metal displacement
temperature change
All substances are made of matter. The matter is made out of tiny building blocks called atoms. The atoms are arranged in three different ways called the states of matter. These states are solid, liquid and gas. The states of matter are explained in terms of how the particles are arranged and how they move.
Solids
Solids have a definite shape.
They do not change shape on their own.
A force must be applied to them to change their shape.
The particles are strongly bound to each other.
The particles vibrate in one place but do not move around.
The particles vibrate faster when heated.
The particles are neatly arranged and packed closely together.
Solids cannot usually be compressed. (incompressible)
Liquids
Liquids have no definite shape.
They do change shape on their own.
The particles are weakly bound to each other.
The particles move around freely but gravity holds them down.
Liquids can spread, or diffuse, through each other.
The particles move faster when heated.
The particles are not neatly arranged.
The particles are fairly close together.
Liquids are incompressible.
Gases
Gases have no fixed shape.
They do change shape on their own.
Gases spread out, or diffuse to fill the available space.
The particles are not bound to each other.
The particles move around freely.
The particles move faster when heated.
The particles are not neatly arranged.
The particles are very spread out.
Gases are compressible.
Particles move more quickly when heated because they have more energy. When particles are cooled they lose energy and move more slowly. Changes in the energy, and hence speed of movement, of particles leads to changes of state.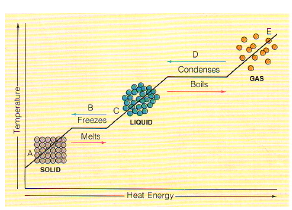 Mixtures
Mixtures
A mixture is formed when two or more substances or things are placed in the same container together. The two substances do not interact with each other. The parts of a mixture can be easily separated from each other by simple physical processes like filtering and sieving.
Dissolving
A solution is a type of mixture. The particles in this type of mixture are very small so you can't see them but like all mixtures they do not interact with each other. Often the particles of a solution can be separated by simply allowing the liquid part to evaporate away. Sugar mixed with water forms a solution. In a solution the liquid part is called the solvent and the dissolved substance is called the solute. The solute can be a solid, liquid or a gas. Lemonade is a solution formed when sugar (a solid) and carbon di-oxide (a gas) are dissolved in water (the solvent).
Diffusion
Both liquids and gases move to spread out evenly through the available space. The process of moving is a random one. The process of spreading out is called diffusion. Substances move from an area where they are highly concentrated to an area of low concentration.
Diffusion does not happen in solids because the particles are fairly fixed in position. The speed at which diffusion takes place depends on the concentration and the temperature.
If there is a big temperature difference then diffusion happens faster because the particles move quicker because they have more energy. Diffusion rate also increases as the concentration difference increases. The bigger the concentration difference the faster diffusion occurs.
Density
Density is a measure of how closely packed the particles are in a substance. The more particles that are packed into a particular volume the greater the density.
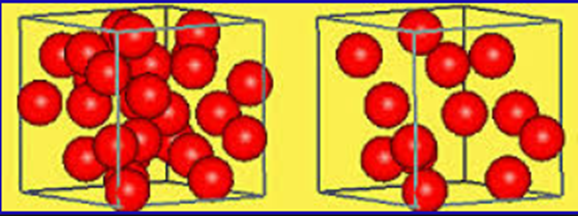 More dense vs. less dense
More dense vs. less dense
Density = mass/volume