Chapter 18- Speed of Reaction
- Different reactions take place at different rates. For e.g. fermentation is very slow while precipitation reactions are very fast.
- The speed of reactions can be measured by measuring any of the following per unit time: amount of reactant used up, amount of product obtained, volume of gas produced.
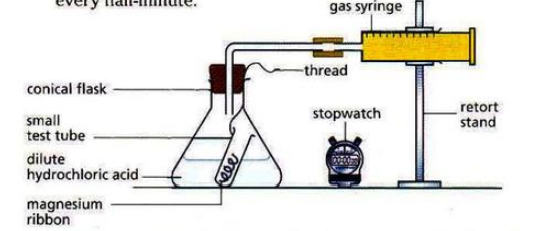
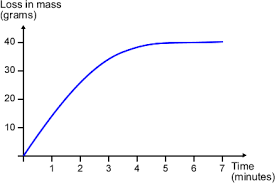
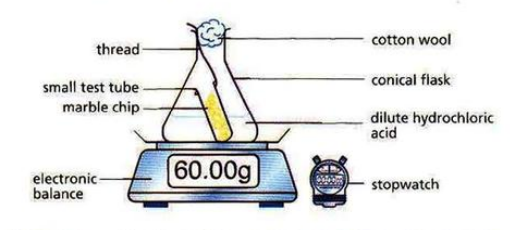
\
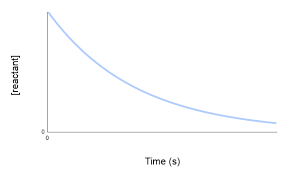
- For any reaction to occur, the particles must collide with each other with sufficient activation energy (minimum energy to start a reaction). The collisions that result in formation of product particles are called effective or successful collisions.
FACTORS AFFECTING SPEED/RATE OF REACTION
- Concentration of the reactants (directly proportional to the speed of reaction): When the concentration of the reactants is more, there are more particles per unit volume which increases the frequency of collisions.
- Pressure of gaseous reactants (directly proportional to the speed of reaction): When the pressure of the reactants is more, there are more particles per unit volume which increases the frequency of collisions.
- Particle size (directly proportional to the speed of reaction): When the particle size is smaller, more surfaces are exposed, increasing the surface area to volume ratio which in turn increases the frequency of collisions.
- Temperature of the reaction (directly proportional to the speed of reaction): When the temperature of the reactants is more, the particles gain kinetic energy and collide more frequently.
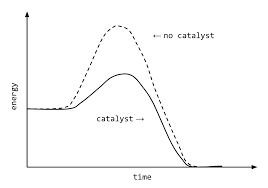
CATALYSTS
- Catalysts is a substance which increases the speed of the reaction while remaining chemically unchanged itself.
- A catalyst is needed in only a small account, increases the speed but not the yield, is selective in its action, is not used up during the reaction, and may be effected by impurities, heat and pH.
- Enzymes are biological catalysts which are sensitive to pH and temperature.
- Catalysts speed up the chemical reaction by lowering the activation energy for a reaction or providing an alternate pathway.
\
\ \