Chemistry Unit 3: Bonding <3
Same group = similar valence electrons = same properties = form similar ions and have similar roles in ionic compounds
Octet rule: 8 electrons = full
Halogens have 7 valence electrons, so rlly want one more -> most of the time -1 charge (anion)
E.x. fluoride anion, chloride anion
6 valence electrons, similar, want to get 2 more electrons -> most of the time -2 charge (anion)
E.x. oxide anion, sulfide anion
Alkali metals have only 1 valence electron, rlly want to get rid of -> +1 charge (cation)
E.x. sodium, potassium with positive one charge
Alkali earth metals -> +2 charge, magnesium, calcium, etc
Hydrogen is an exception! Can be +1 or -1 charge bc only one electron to lose/gain for a “full shell”
Ionic Bonds
Ionic bond when Opposite charges attract!, between metal and nonmetal forms ionic compound
Example: Halogen + Alkali metal ( -1 and +1 ions)
Ex. lithium (+1) + chloride (-1) -> lithium chloride
Sodium (+1) + chloride (-1) -> sodium chloride (table salt)
Can also have different amts of ions for each element. Just have to have neutral charge
Ex. Calcium iodide (two iodine ( each -1 = -2) and one calcium (+2))
Naming
Sodium chloride
^ ^
Cation Anion
Polyvalent ions
D-block elements how to name? Look at the anion.
Ex. cobalt sulfide. Sulfide is -3 so cobalt must be +3. Write as cobalt (III) sulfide
Polyatomic ions
These are covalently bonded groups of atoms that act as an ion with a net charge. Still form ionic bonds with other ions. Bond in a ratio. Use parentheses around if multiple
Example: ammonium sulfate
Covalent bonds: SHARE electrons, between two nonmetals
E.x. O2 (Diatomic element: element that doesn’t exist alone in nature as an individual
Water (H2O)
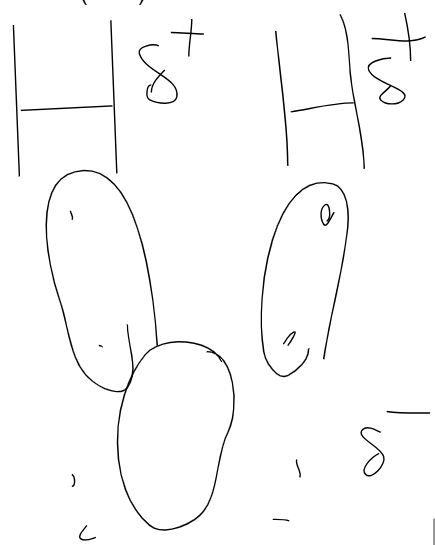
Oxygen pulls the pair closer to it tho, makes it’s side more negative. Polar molecule
Lewis structures
- means an electron pair, a bond
Lone pair: two dots, no covalent bond
Molecules, salts, compounds, and chemicals?
Molecule: group of two or more atoms covalently bonded together
Covalent network solids: covalently bonded substance that doesn’t consist of molecules, instead a network
E.x. silica (SiO2)
Connected in a network, rather than a molecule
One grain of sand is a giant molecule
Salt: cations and anions ionically bonded to each other
E.x. NaCl, table salt
Ionic solids are arranged in a crystal lattice structure, so that the charges work out
Compound: a chemical substance composed of two or more different elements. Either covalently or ionically bonded
Covalent compounds and ionic compounds
Chemical: any substance with a definite composition
Metallic bond:
Sea of valence electrons. Occurs between two metals. Allows electricity/conductivity and property of malleability