12-01: Functional Groups and Linkages
- Most of a living organism is water
- The remaining amount of “stuff” in an organism falls into 4 main categories:
1. Carbohydrates
2. Lipids
3. Proteins
4. Nucleic Acids
Biochemistry
- Many cellular molecules are complex and can be made up of thousands of atoms
- These molecules are made by stringing together many smaller units → Single units are called monomers, multiple units are called polymers
- Anabolic reactions: build up substances
- Catabolic reactions: break down substance
- Both catabolic and anabolic reactions occurring in an organism: metabolism
Intramolecular bonds: bonds within a molecule
3 types of intramolecular bonds
- Covalent bonds
* 2 atoms share electrons (e-) → 2 non metals - Ionic bonds
* 1 atom loses an e- and 1 gains an e- - Polar covalent bonds
* Sharing the e- less fairly
Electronegativity (EN): the strength in which an atom attracts e- → how aggressive it is
The type of intramolecular bond is distinguished by a difference in electronegativity (∆EN)
- Covalent: ∆EN < 0.4 → share equally
- Polar covalent: ∆EN = 0.5-1.7
- Ionic: ∆EN > 1.7 → atom with greater EN takes e- from atom with lesser EN and both become charged
Polar Covalent Bonds
- One atom has a stronger hold on e- they share
- One end of the molecule gets slightly positively charged and one end gets slightly negatively charged
δ+ → one end gets + charge (weaker EN)
δ- → one end gets - charge (stronger EN)
This influences what will be attracted and how things will biologically interact
Water is polar → has polar covalent intramolecular bonds
- Because of Oxygen (O)’s EN, e- tend to spend more time near the O atom than near the Hydrogen (H) atoms
- The unequal sharing e- creates a slight difference in the charge between the ends/poles of the molecule
- the O end is δ- and the H ends are δ+
* Due to its polarity, water forms H bonds with itself
* Very weak bonds, but many of which come together and become very strong together
Water also forms bonds through….
- Cohesion: water molecules are attracted to other water molecules
- Adhesion: water is a polar molecule and thus attracts other polar molecules
Intermolectular forces: bonds between molecules
- ==London dispersion forces==: very weak attraction between molecules, even non polar ones. Increases with molecule size
- @@Dipole dipole attraction@@: attractive force between 2 polar molecules
- @@H bonding:@@ special kind of dipole dipole attraction; between 2 polar molecules with Hydrogen bonded to N, O, or F
Carbon - the element of life
- Backbone of nearly every biological molecule (except for water)
- Organic compound: compound that contains carbon-hydrogen bonds (may also contain other elements like O, N, etc) and is often found in organisms
- ^^C and H form a non polar bond so hydrocarbons are non polar^^
* Polarity can be achieved by adding other atoms called (FGs)
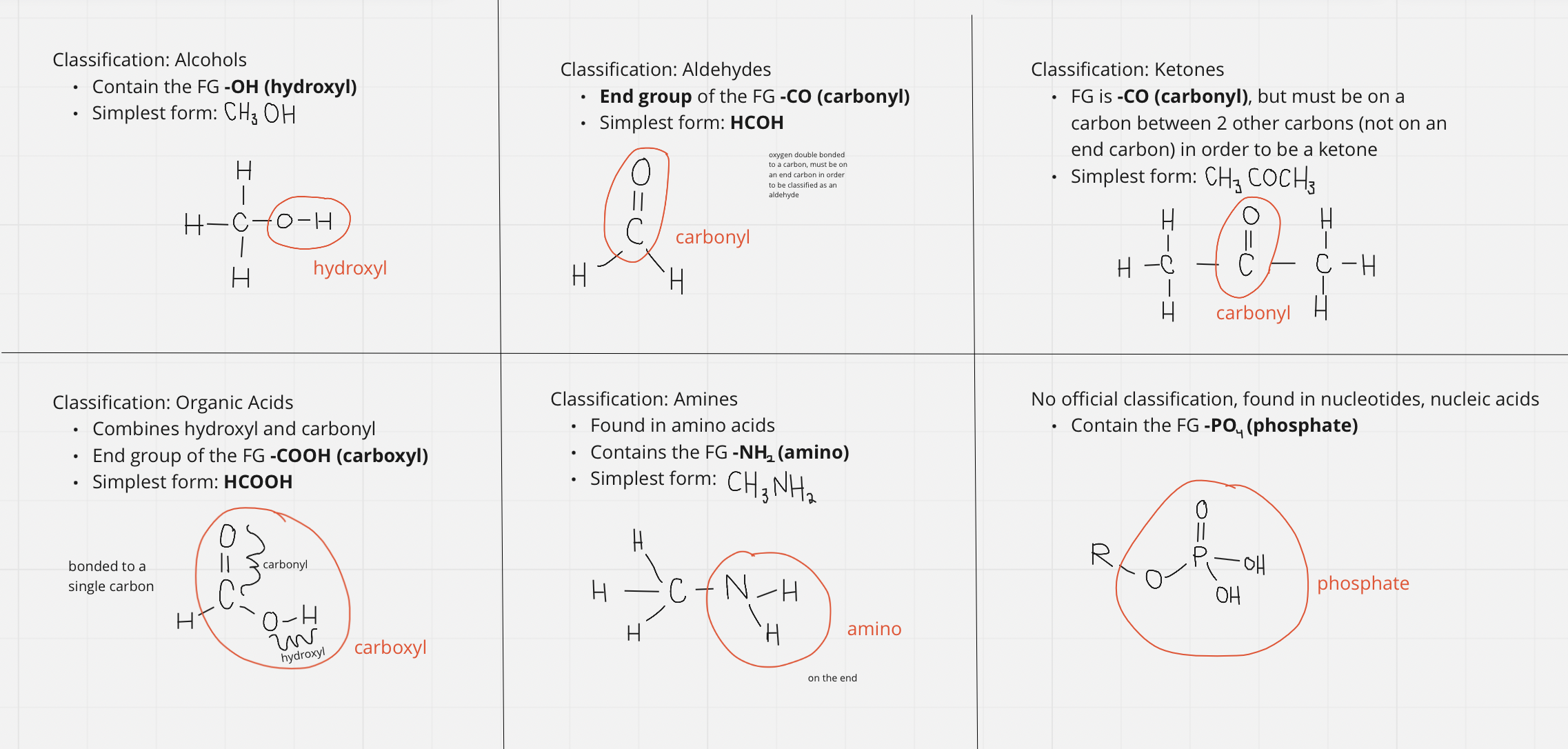
- Functional Groups -
- Molecules interact with each other at specific regions of their molecules (^^changes the dynamic of the molecule that allow it it interact differently^^)
- Used to classify molecule types
- After a reaction between 2 molecules’ functional groups, a linkage will be formed
* Monomers link to form polymers. This happens because of FGs reacting
Classifying FGs: if it has the FG, then it is classified that certain way
Types of Reactions
2 types:
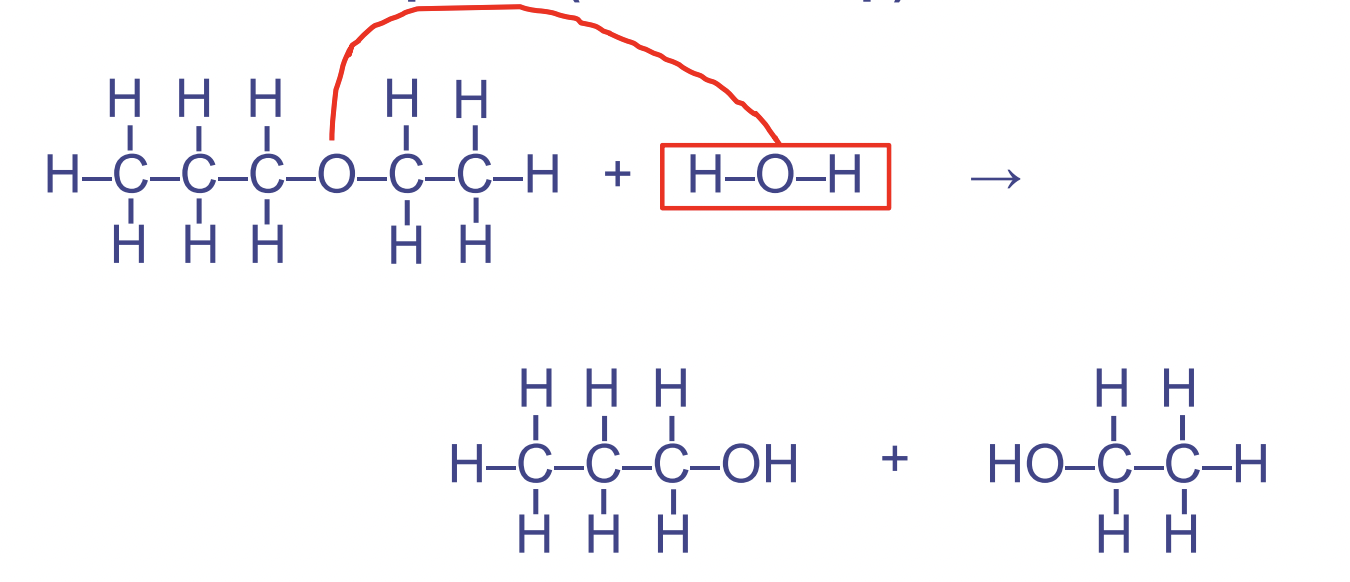
1. Hydrolysis
- Rupture → use of water to rupture, break down
- Catabolic
* Used to split larger molecules apart - Water is required and it is used up → it is a reactant
2. Dehydration synthesis (condensation)
- Condenses smaller particles into larger ones
- Anabolic
* Used to build up molecules - Water is released → it is a product
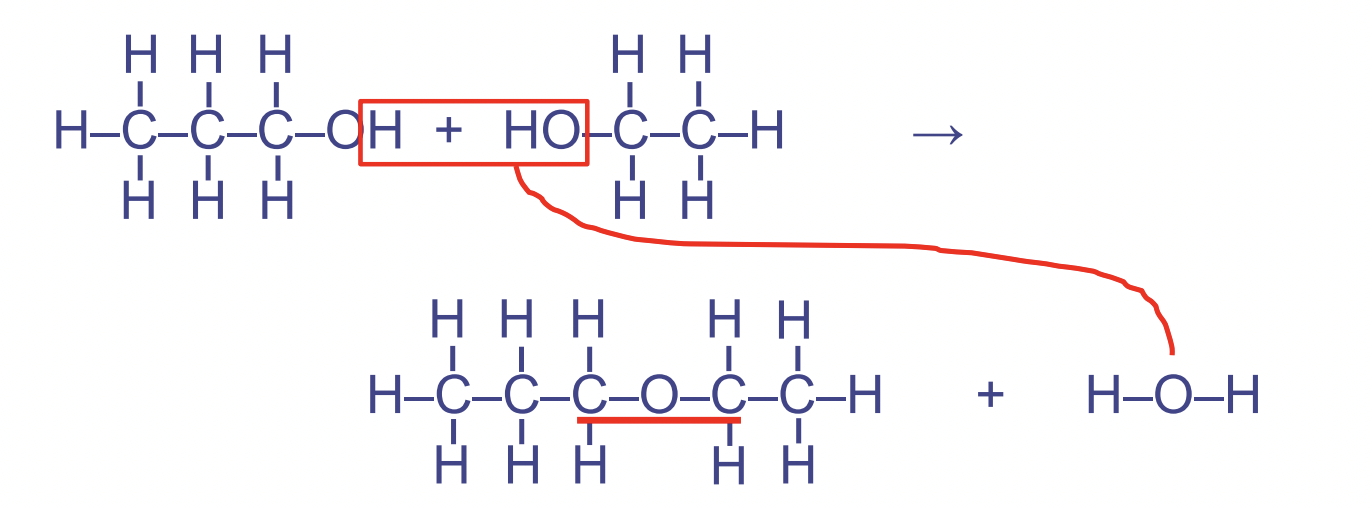
Linkages that form in dehydration synthesis reactions
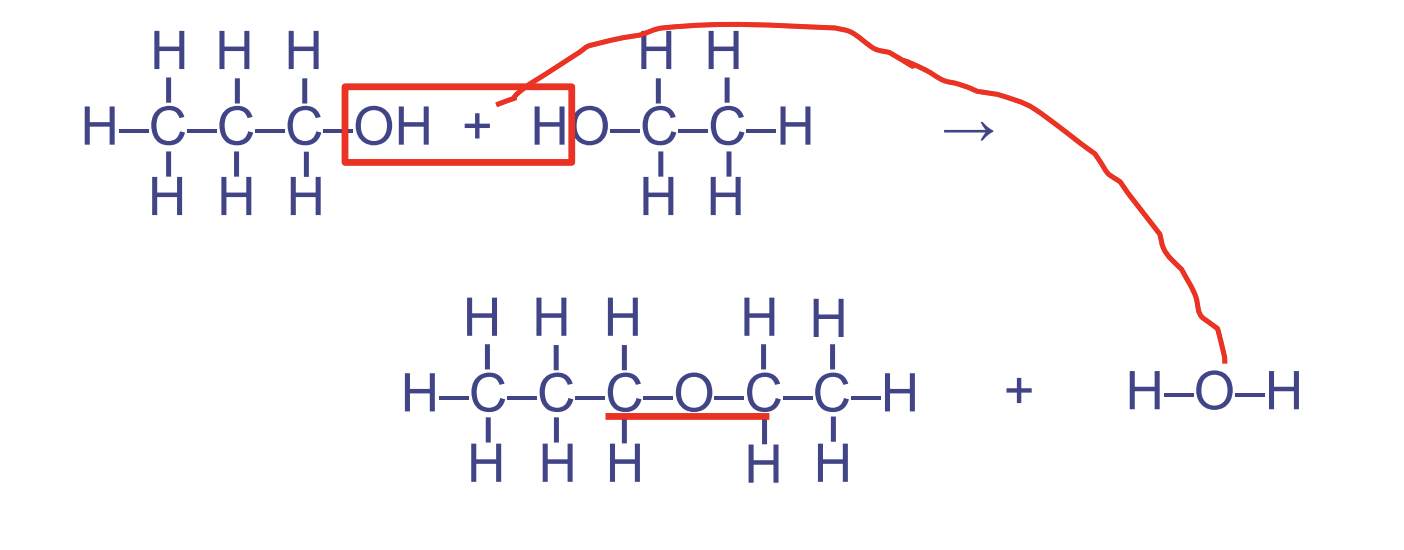
Ether Linkages
(glycosidic link between sugars, when it occurs between sugar molecules)
- Between %%2 hydroxyl groups%% (alcohols)
- Used in carbohydrates
- Forms the pattern COC
Ester Linkages
- Between a %%hydroxyl and a carboxyl group%% (when they react)
- Used in triglycerides
- Forms the pattern OCO
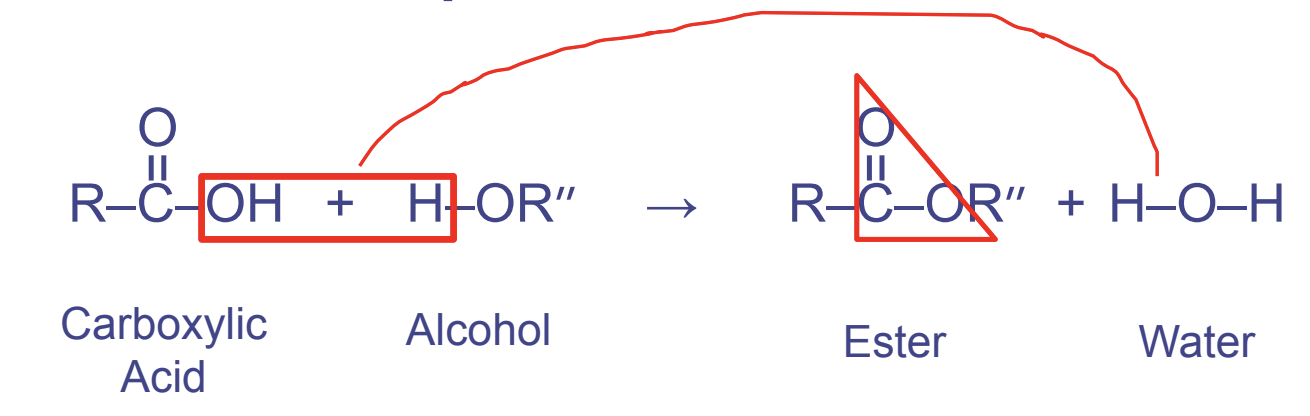
The double bond indicates that it is an ester linkage
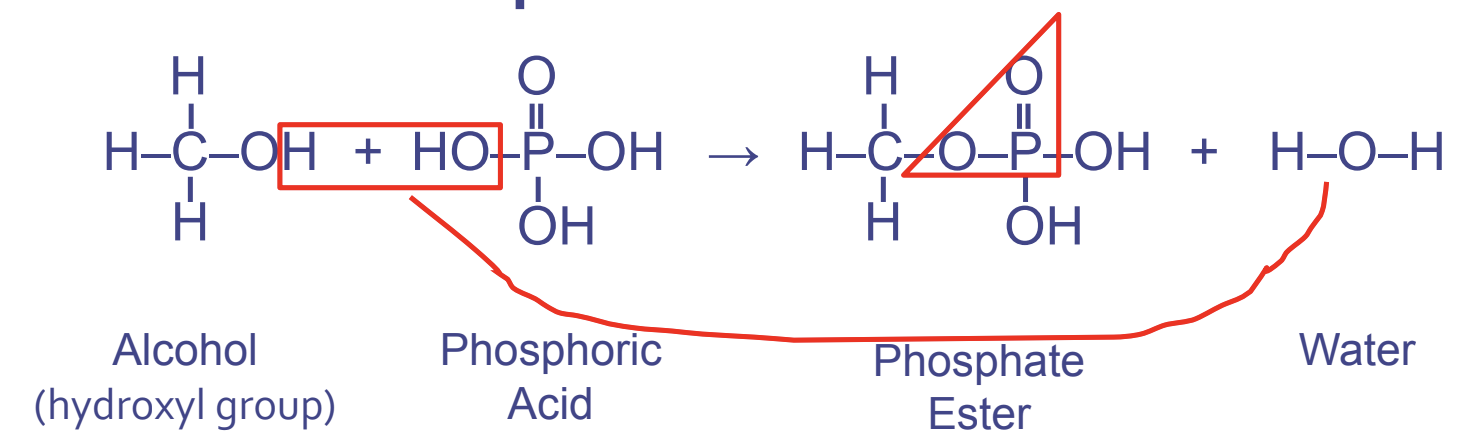
Phosphate Ester Linkages
- Between hydroxyl FGs (first) and phosphate FGs (second)
- Used in phospholipids and nucleic acid, which instructs the cells, DNA and RNA
- Forms the pattern OPO
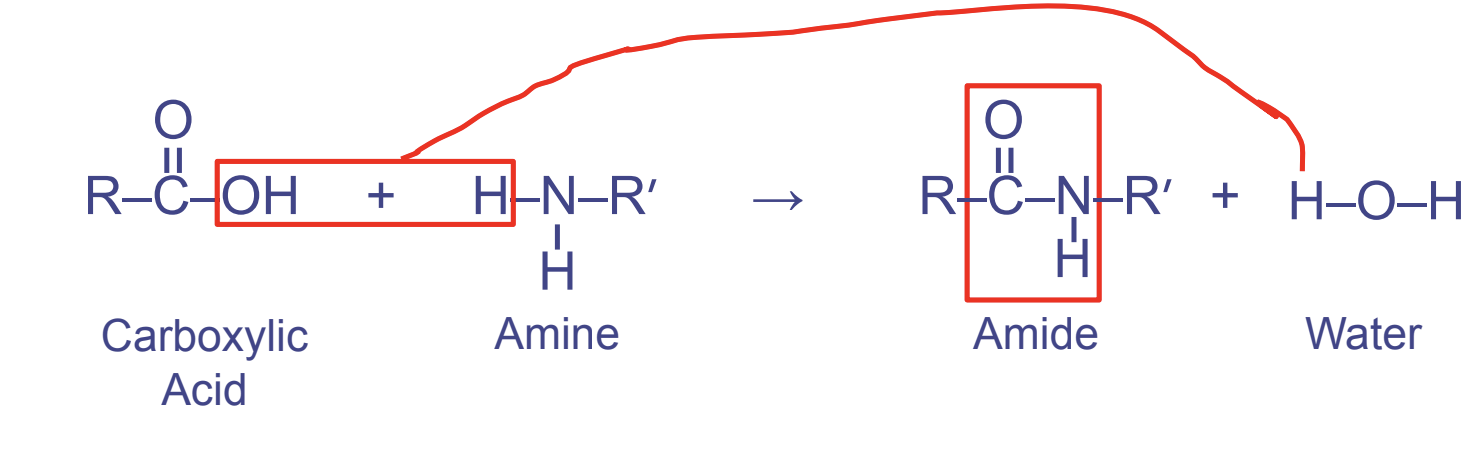
Peptide Linkages
- Between carboxyl FGs (first) and amino FGs (second)
- Links amino acids together
- Forms the pattern OCNH