PY 131 Chapter 16: Heat Transfer
Heat Transfer
- Heat moves due to temperature differences.
- Heat energy always moves from hot to cold.
- Heat is transferred from one object to another via three mechanisms: * Conduction * Convection * Radiation
- During conduction the atoms/molecules of the object transferring the heat do not move. * Conduction is the only method of moving heat through a solid.
- During convection the atoms/molecules do move. * Convection occurs in liquids and gases.
- During radiation heat (energy) is transferred through space by electromagnetic waves (light) or gravitational waves.
Conduction
- Conduction is the flow of energy between objects or from one object to another due to collisions between the atoms/molecules and without net motion of the material. * No net motion means the body does not move as a whole i.e. there is no bulk flow.
- Conduction can occur in solids, liquid and gases.
- In a solid the atoms are largely confined and collisions are the main mechanism by which the heat flows.
- In a liquid or gas the heat flows due to collisions of the atoms/molecules but also atoms/molecules can diffuse.
- Materials through which heat travels easily are called ==(thermal) conductors.== * Examples include metals, diamond (diamond is 250% better thermal conductor than copper).
- Materials through which heat travels with difficulty are called ==(thermal) insulators.== * Examples include glass, wood, plastic, air.
EXAMPLE 1
If you hold one end of a metal bar against a piece of ice, the end in your hand will become cold. In which direction is energy moving?
- ==From your hand to the ice==
Convection
- Convection is heat transfer through an object due to the bulk motion of the material.
- Convection requires there be a net force on a fluid element.
- Convection can occur in different ways depending upon the force e.g. buoyant convection or forced convection. * In buoyant convection, the force is buoyancy so buoyant convection requires there be gravity – without it buoyant convection cannot occur. * Forced convection is when you blow or pump the fluid.
- Consider a ‘fluid element’ with density ρb * Here b stands for ‘bubble’.
- The surrounding fluid has the same density ρ.
- What happens if this element moves upward by an amount Δy?
- The density will change to ρb +Δρb .
- At the new location, the surrounding fluid has a density ρ +Δρ.
- If ρb +Δρb < ρ+Δρ then the element will continue to rise due to the buoyancy force and the fluid is unstable to convection.
- Since ρb = ρ the criterion for instability is that Δρb < Δρ or Δρb Δ y ≤ Δρ Δ y A buoyantly convecting fluid forms Bénard (convection) cells.
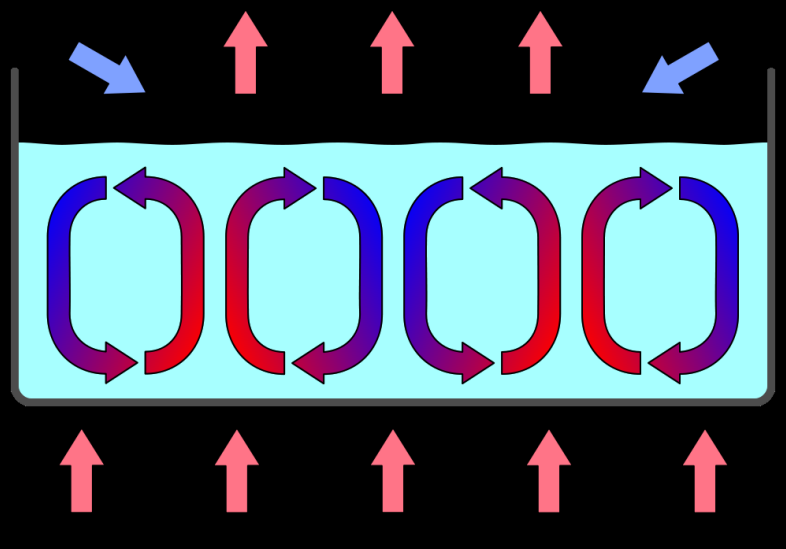
- High-resolution images of the Sun show its surface is broken up into granules: http://www.youtube.com/watch?v=W_Scoj4HqCQ
- The Bénard cells on the Sun are huge: each is ~1000 km across and lives for ~ 10 minutes.
- Careful observations indicate there are also supergranuales which are of order 30,000 km in size. * It is thought these may be the imprint of deeper lying convective cells.
- Convection is responsible for sea breezes. * The land heats the air above it better than the water heats the air above it. * The density of the air over the land decreases causing it to float (rise). * The air over the sea is pushed in by pressure forces in to replace it.
Newton’s Law of Cooling
- Consider two points in space separated by a distance Δx and with a temperature difference ΔT.
- The ratio of ΔT to Δx is called the temperature gradient. * ΔT/Δx
- The temperature gradient (which is a vector) will cause an amount of heat ΔQ to flow between the points in a time Δt.
- The thermal current I is * I= ΔQ / Δt
- It is found from the experiment that the thermal current is proportional to the temperature gradient. * I ∝ - (ΔT/Δx) * The minus sign is because heat flows from hot to cold.
- This relationship is called ==Newton’s Law of Cooling.==
- The larger the temperature difference, the faster a hot object loses energy or a cool object gains it.
- As the object cools, the heat flow slows down.
- In addition to the temperature gradient, the thermal current depends on the contact area and what the material is. * The smaller the contact area the smaller the heat flow. * Heat finds it harder to pass through some materials than others.
- Newton’s Law of Cooling works well for conduction. For convective cooling it works if the cooling liquid/gas is pumped/forced/blown: if the convective cooling is buoyancy driven then it doesn’t work as well. * It doesn’t take into account the fluid speed.
EXAMPLE 1
Why does it take an ice cube longer to melt on a winter day than on a summer day?
- ==The atmosphere is cooler==
Radiation
- Heat Transfer by Radiation is the transfer of heat via the emission and absorption of light.
- All objects with a temperature emit light. * This is not the light reflected from a light source e.g. the Sun.
- Usually, we don’t notice this radiation because, at typical everyday temperatures, the emitted light is mostly infrared. * It takes temperatures of ~ 500 Celsius before the emitted light starts to become visible light. * At very high temperatures the emitted light can be UV or X-rays.
- The emission of radiation causes an object to cool. * it’s the emission of radiation into space that cools the Earth at night.
- The temperature of an object is related to the frequency or wavelength of the light it emits which has the maximum intensity
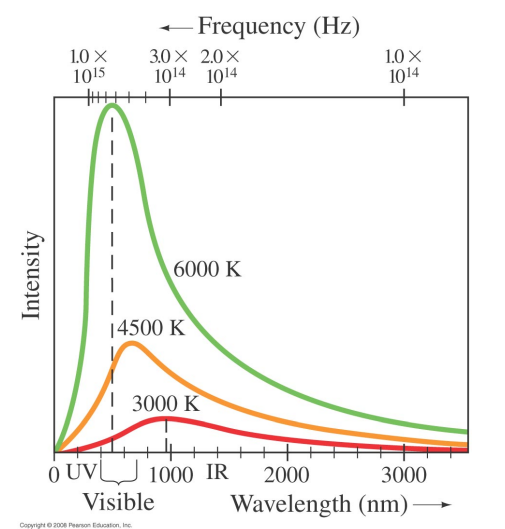
- This relationship is known as ==Wien's Displacement Law==.
- When radiation is incident upon an object, some of the light is absorbed, some is reflected, and some passes through. * The absorbed radiation will cause an increase in the object’s temperature. * The absorption of radiation emitted by the Sun heats the Earth during the day.
- An object which absorbs all the radiation incident upon it is called a blackbody.
- Objects which are good emitters of radiation are also good absorbers of radiation (and vice versa).
- To make life complicated, the amount of radiation absorbed and emitted depends on the type of light. * Some objects reflect visible light well but absorb all the IR, for example.
- Assuming a black object is black for all types of light, and a white object is white for all types of light, a black object radiates energy faster than a white object.
- An object’s opacity is a measure of the amount of light it absorbs as light travels through the object.
- The opacity can change with the ‘type’ of light. * an object (e.g. glass) can be very transparent to visible light but not transparent (opaque) to infrared or UV.
- The opacity of the atmosphere is very important to astronomers. * If the atmosphere is opaque, a telescope can’t see through it.

The Greenhouse Effect
- It is the difference of the opacity of the atmosphere to visible and infrared light which keeps Earth warmer than it should be given our distance from the Sun.
- If Earth were a blackbody with no atmosphere it would have an average temperature of ~ +5 °C.
- Given that Earth reflects about 30% of the incident sunlight, an Earth without an atmosphere would have an average temperature of ~ -20 °C.
- Earth’s actual average temperature is ~ +15 °C.
- The Sun emits a lot of its light as visible light. Visible light can largely pass through the atmosphere and be absorbed by the Earth causing Earth to be heated.
- Earth emits most of its radiation as infrared light.
- This should cool the Earth but the atmosphere is opaque to infrared light so energy gets absorbed.
- The heated atmosphere radiates some of its energy back toward the Earth.
- The net effect is that radiation finds it difficult to escape and the Earth is warmer than it would be if it could easily escape.
- This is called the ==Greenhouse Effect.== * Even though actual greenhouses work slightly differently.
- The same effects occur on other planets or moons with atmospheres. * On Venus the Greenhouse Effect has runaway and the temperature is 450 °C.
- The gases in Earth’s atmosphere which absorb most of the infrared radiation are water vapor and carbon dioxide.
- The amount of these gases in the atmosphere changes over time due to natural processes such as volcanoes, ice ages, sun cycles etc. * It even changes over the cycle of the year.
- Over the last 200 years the amount of carbon dioxide in the atmosphere has roughly doubled.
- Since 1960 the average temperature of the Earth has increased by ~1 °C.
\