HL BIO Unit 1
Molecules and Water
origins of life were in water and water is a medium for life
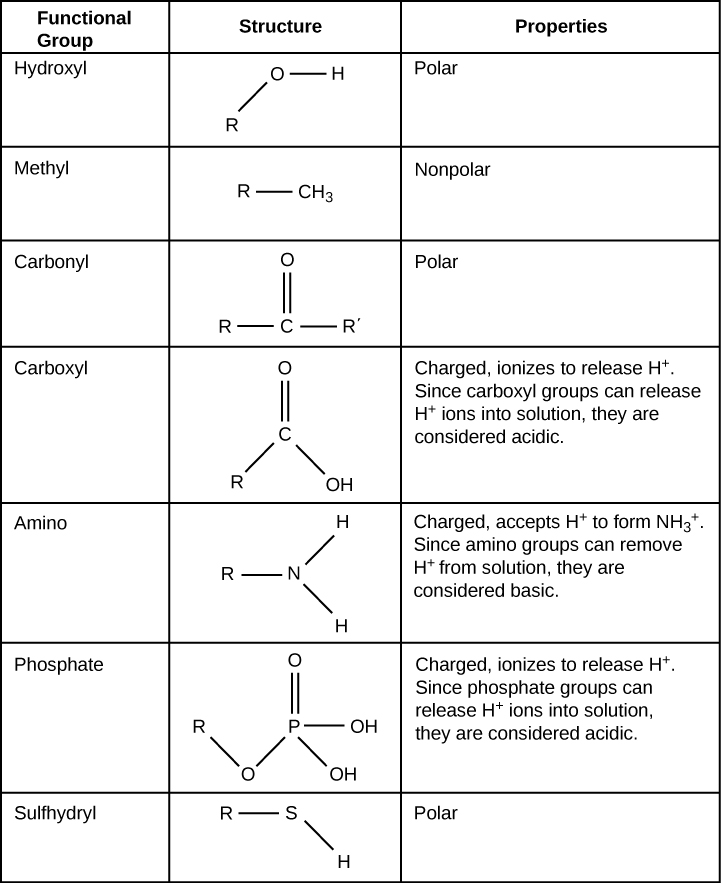
electron configuration determines the chemical behaviour of an atom, which depends on the # of electrons in the outermost shell
all atoms with incomplete valence shells are chemically reactive
London Forces - exists in all molecules
Dipole-dipole forces - exist between polar moles. Attraction between a partial positive and partial negative
Hydrogen bonds - forms between molecules containing H-F, H-O, H-N bonds. Much stronger
these intermolecular forces give water many unique properties
Ion-dipole interactions - form between ion and polar mole. (full charge with smt like water)
Cohesion and Surface
it sticks to each other and forms droplets, it sticks to something else (polar)
It allows things like insects to sit on top.
Adhesion (ex capillary action)
ex plants, water is pulled up roots/stem
hydrophilic - water loving
polar or charged molecules
hydrophobic - water fearing
non polar
Blood Plasma:
polar dissolves, like the polar amino acid molecule is small and will dissolve in blood
but the non-polar does not dissolve, these are instead trsnported by hemoglobin which is O2
HEMOGLOBIN: oxygen comes from lungs, then oxygen bonds to hemoglobin and oxygen is released thru tissue
Lipoproteins - have phospholipids which can be polar and non-polar
Physical properties:
Buoyancy is the living things that have overall density similar to water
Viscosity - hydrogen bonds in water increase the viscosity to many organic solvent, more dissolved solutes
Specific Heat Capacity → amount of energy required to increase the temp by 1 degree Celsius.
water has a high thermal conductivity - conducts heat quickly. Opposite, fats and oils conduct heat much slower and better insulators
The goldilocks zone → there is a perfect state of theu universe for all the living things on earth
Carbohydrates:
Carbs → simple sugars → carbon, hydrogen and oxygen → energy storage for short term
2
Chemical Reactions in Living Organisms:
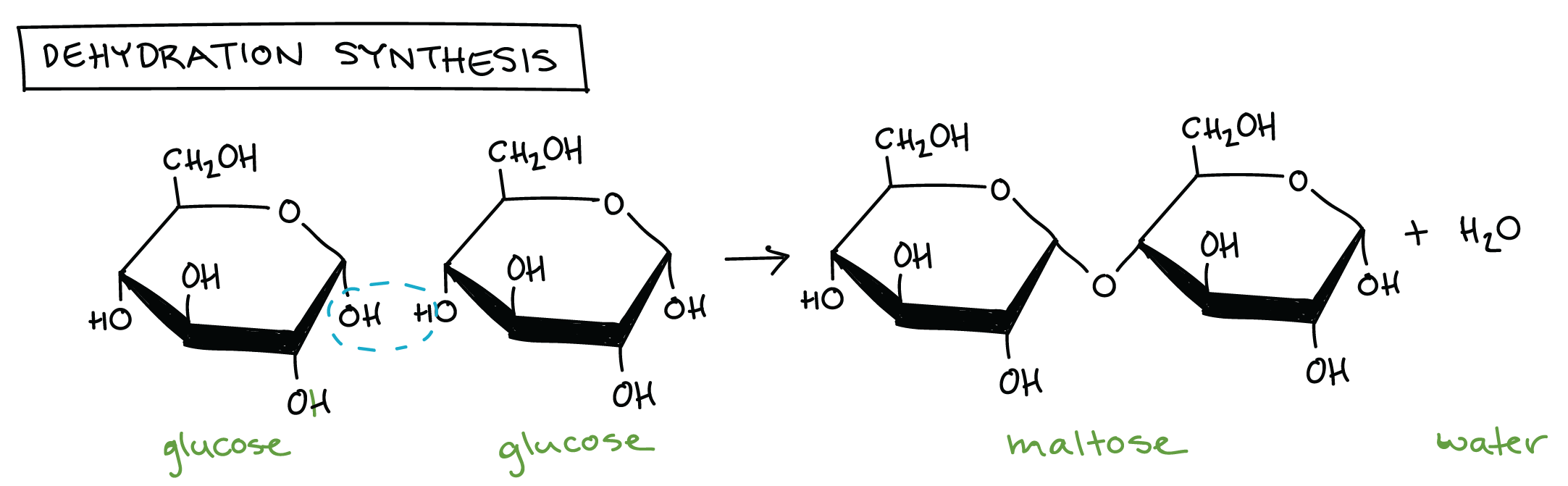
Dehydration → removes a water
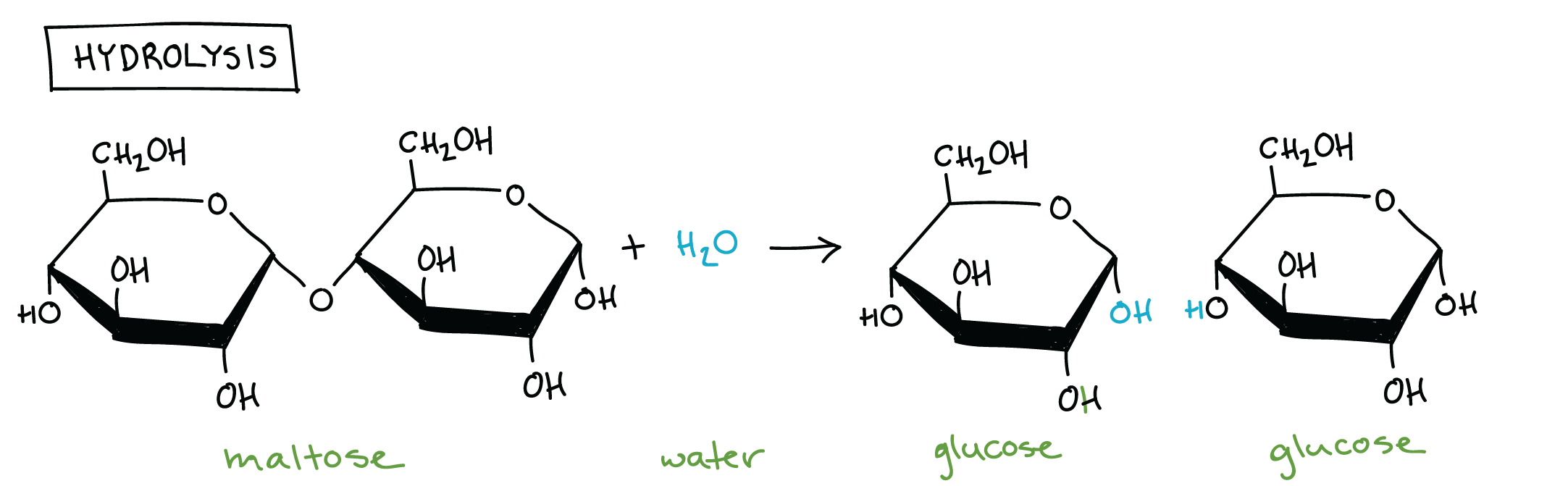
Hydrolysis → add a water
Neutralization →
Redox Reactions → electrons lost from one atom and gained by another. Loses electron is oxidized and gains electrons is reduced
Carbs:
chemically stable, good for quick and short term energy
important in structural molecules in cells
simple (sugars C(H2O) ) and complex (starches C6H12O6)
MONOSACCHARDIES (CH2O)n GLUCOSE
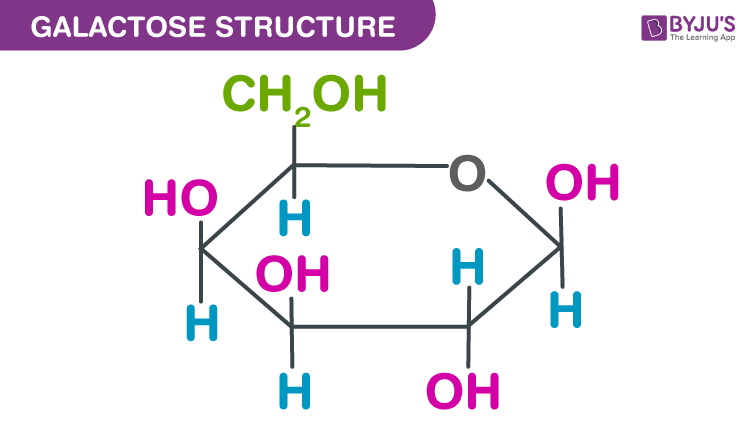
glucose, fructose, galactose
found in cyclical structures
hexose - 6 carbons
pentose - 5 carbons
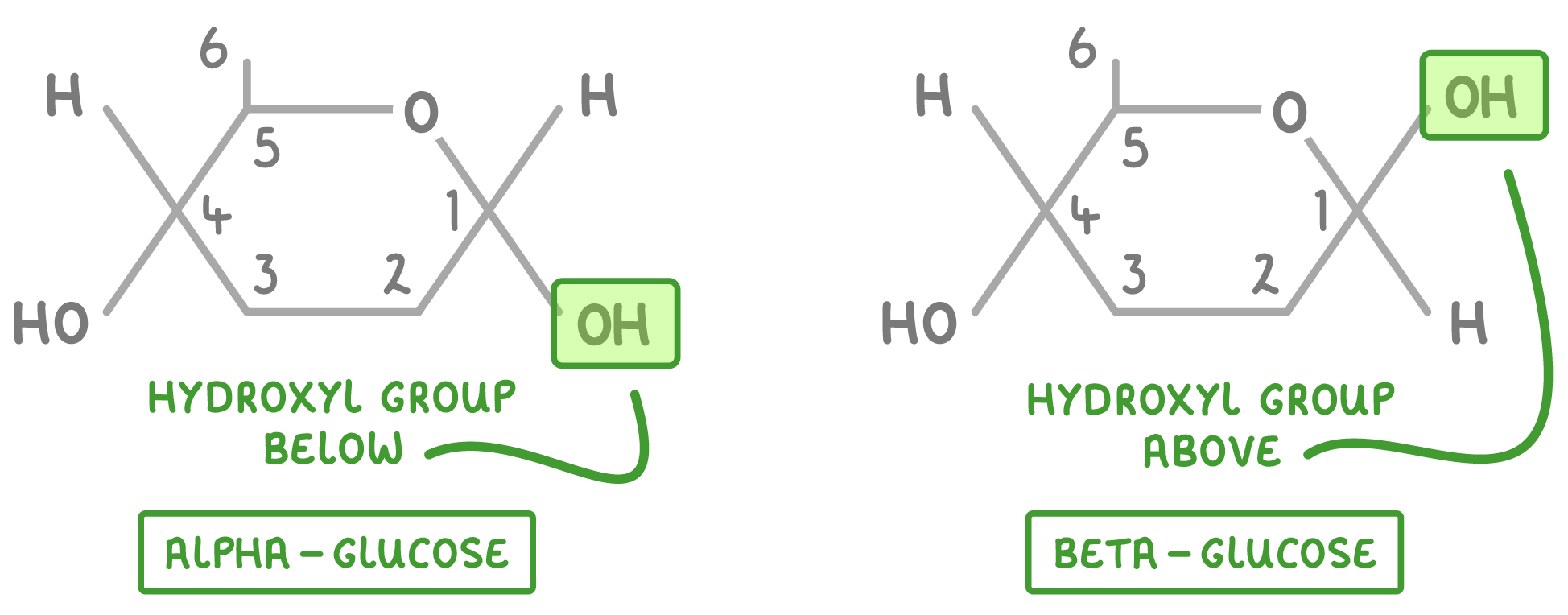
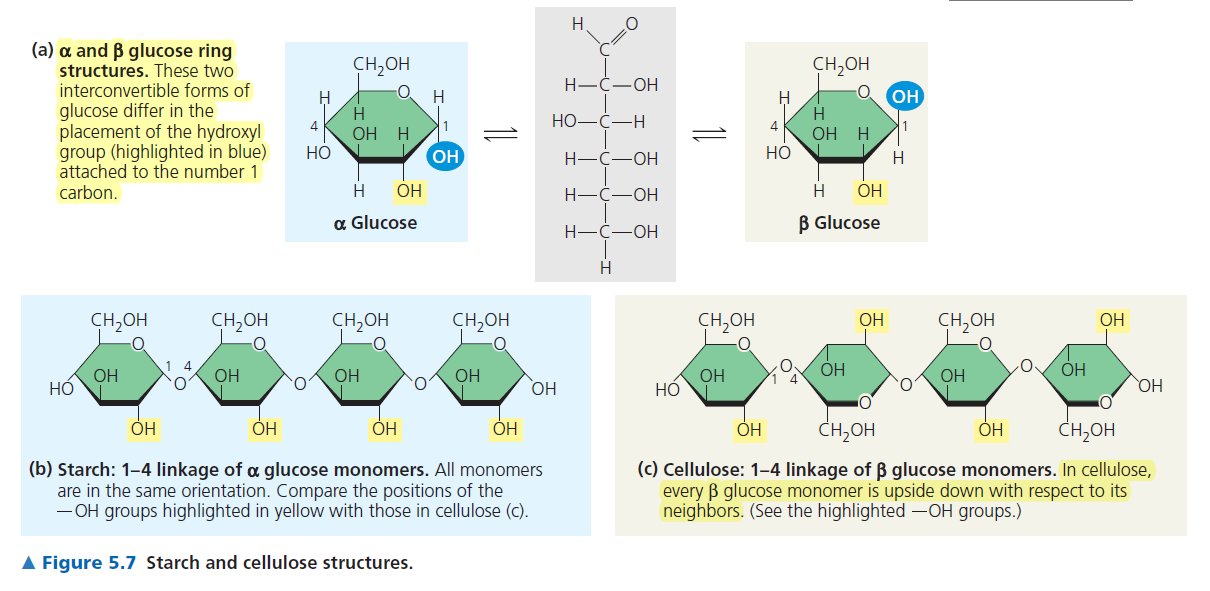
Alpha (dominating so thats why 1st is down) is OH down, down, up, down and Beta is OH up, down, up, down
glucose relases energy when its oxidized, so it loses electrons (respiration)
Galactose OH is up on C4
isomers - same chemical formula diff properties
Glycosidic bonds → anabolic process that needs energy, its the building of larger molecules its the between C1, oxygen, C4 in maltose after dehydration
Disaccharides -
maltose (alpha glucose- alpha glucose)
sucrose (glucose and fructose)
lactose (galactose-glucose)
Polysacchardies - complex carbs
polymers are chains of the identical monomers bonded together
examples starch (amylose and amylopectin), glycogen, cellulose
Amylose → alpha 1,4-glycosidic bonds
Amlyopectin → alpha 1-4 but sometimes 1-6 glycosidic bonds. It is more branched
starch is storage molecule found in plants
glycogen is storage molecule found in animals (muscles and liver)
more ideal energy stroage than monosaccharies because large size decrease solubility
Cellulose - structural polysaccharide found in plants (cell walls)
the hydrogen bond hold celluose microfibrils together to give them a HIGH TENSILE STRENGTH
straight chain with hydrogen bonds holding it together
Starch is all alpha glucose when cellulose is beta-glucose, B1-4 glycosidic linkage
Glycoproteins - short chain carbs (oligosaccharides) attached to proteins
component of cell membrances and important in cell recongition (example blood types)
Lipids :
non-polar molecules made of carbon, hydrogen, and oxygen
long term energy storage
important for cell memberances and signalling molecules
animals store energy
its more stable
non-polar so no water needed
high amount of energy per gram
liquid at body temp
oils - melts below 20º
fats - melts between 20º and 37º
waxes - melts above 37º
steroids
Fatty Acids:
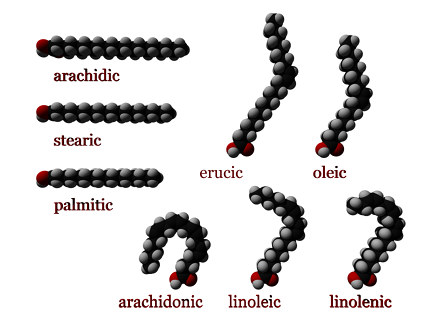
carbon chain (14-20 carbons) with a carboxyl (double O and OH)
Saturated are straight chained
pack together
solid animals fats
less flexible
Unsaturated - mono has kink in bend in chain, poly double bonds so more kinks
less than max amount of hydrogen
double or trips bonds between C
more space between molecules
liquids and oils
more flexibility in memberane
CIS vs TRANS: carbons on same side vs carbons on opposide sides of double bond
trans unsaturated is elaidic acid
cis unsaturated is oleic acid
OMEGA
omega-3 double bond on the C3
omega-6 double bond on the C6
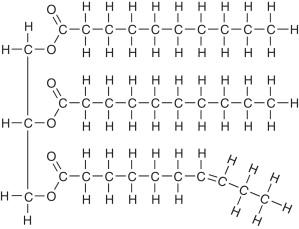
Triglycerides → glycerol + 3 fatty acid chains
bonded by ester bonds
may be the same of different, so saturated or unsaturated
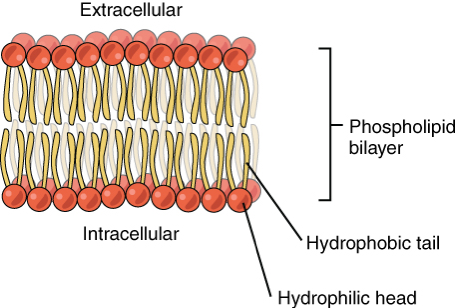
Phospholipids:
glycerol bonded to two fatty acids and phosphate group
primary component of cell membrances
amphipathic (polar (phosphate) and non polar regions (fatty acid) )
Waxes:
hydrocarbon chains linked to alcohols or carbon rings
very hydrophobic, provide waterpoof proteciton for plants and animals
Steriods:
four fused carbon rings with side groups giving molecules specific properties
many hormones are steroids
non polar so can pass directly through cell membrane
Proteins
essential for parts of living, virtually participating in every procress
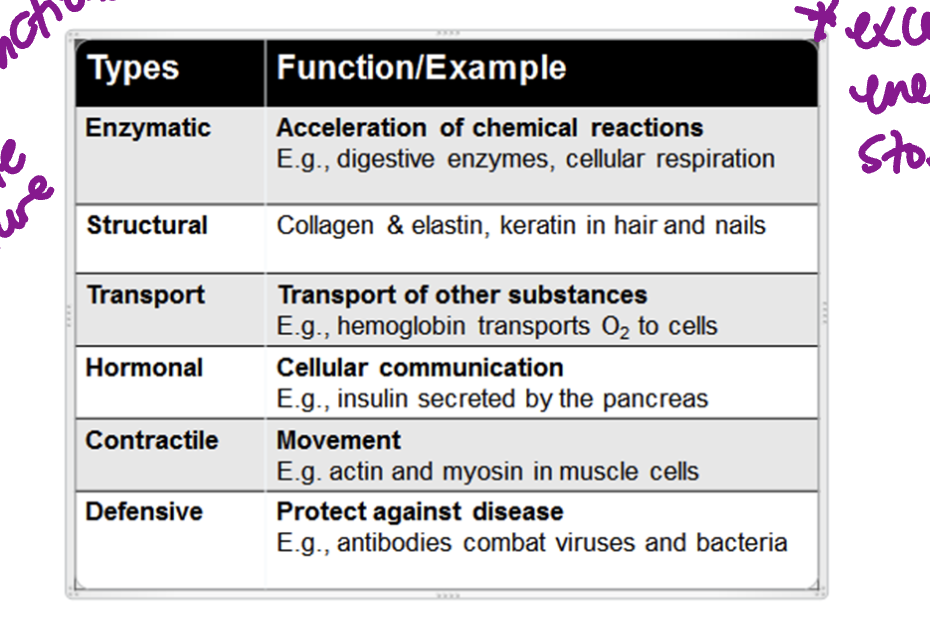
polymers composed of amino acid monomers
Amino Acids have 20 different side groups (‘R’ Group)
nonpolar - C and H chains
polar - OH, NH2, no charge
charged acidic - negative charge
charged basic - positive charge
Last universal common ancestor (LUCA)
Primary Structure - specific sequence of amino acids joind by PEPTIDE bonds called a polypeptide
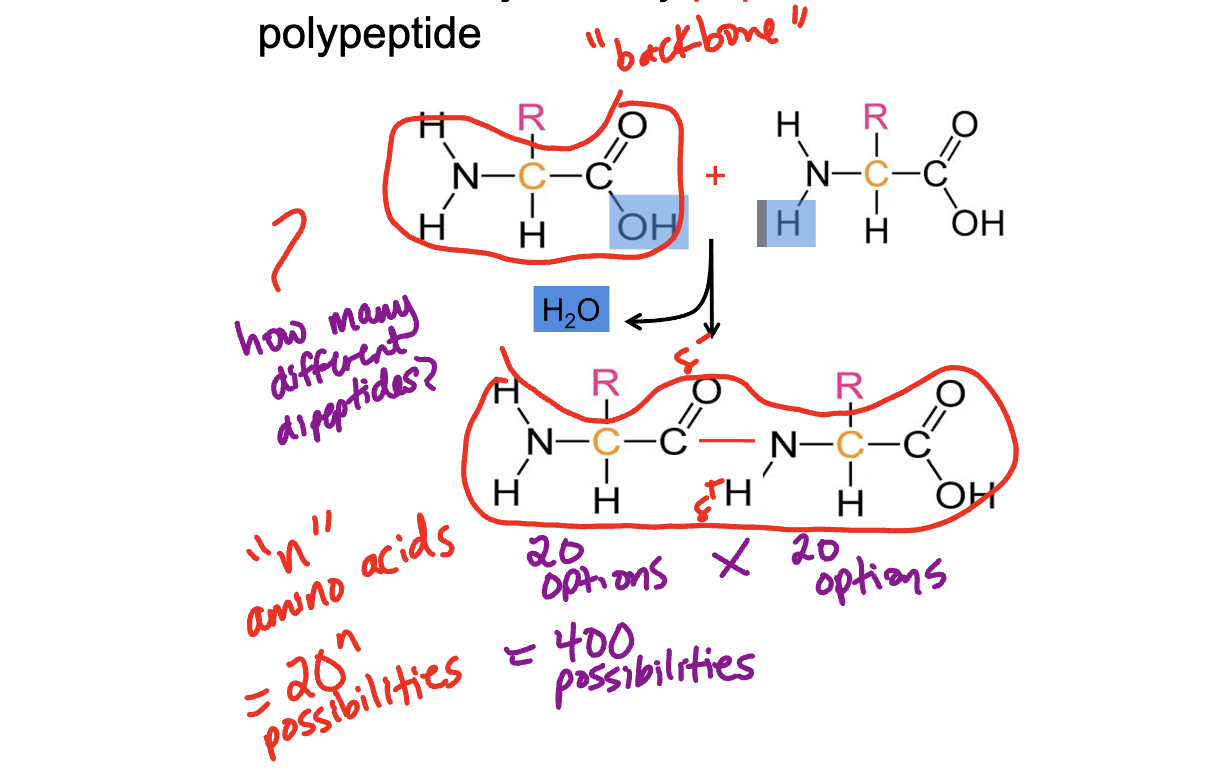

Secondary Structure - hydrogen bonding between the amino acid backbond
alpha helix (spiral) and beta pleated sheets (parallel lines)
Tertiary structure - interactions between chains (R groups)
ionic bonds (charged)
hydrogen bonds (polar)
hydrophobic (nonpolar)
disulfide bridge (cysteine amino acid S-S)
hydrophilic on outisde (exposed to cytoplasm/water) and hydrophobic amino acids fold no to middle
Quaternary structure - two or more folded polypeptides come together to make final functional protein (not for all proteins)
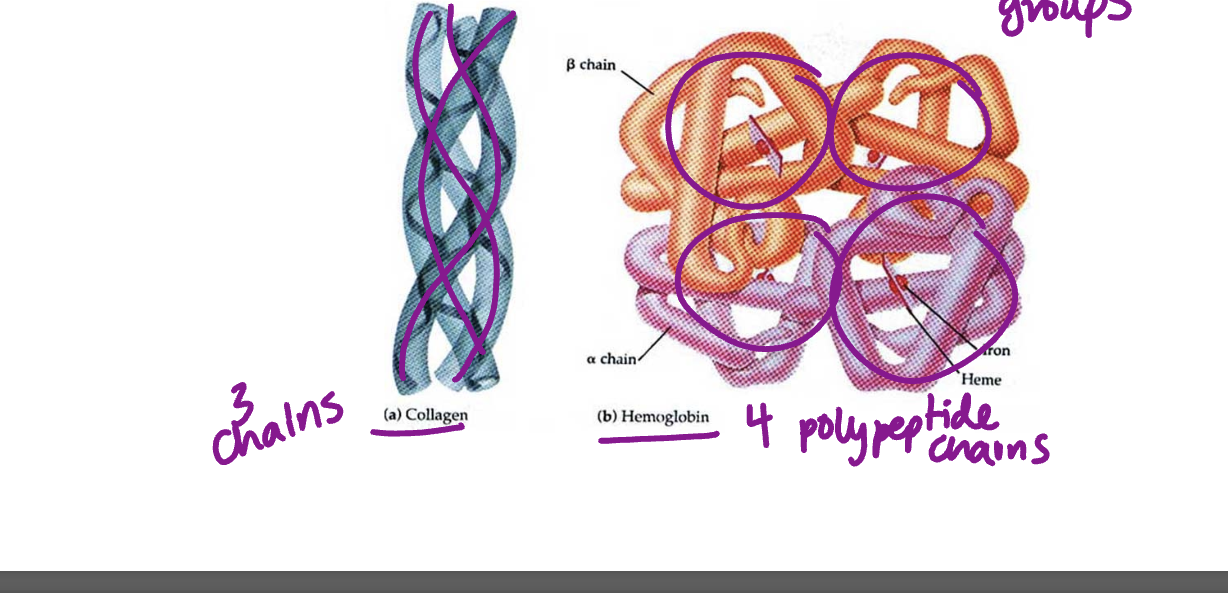
polypeptides held by ionic bonds, hydrogen bonds and etc depending on R groups
Non-conjugated protein - only poplypetide subunits like insulin and collagen
collagen - fibers strong, structured
insulin - hormone for communcation
conjugated protein - includesw one or more non polypetide units in addition to polypeptides like hemoglobin contains heme group
hemoglobin dissolves in blood
compact easy to move through blood/blood vessels
heme is iron to bind to oxygen
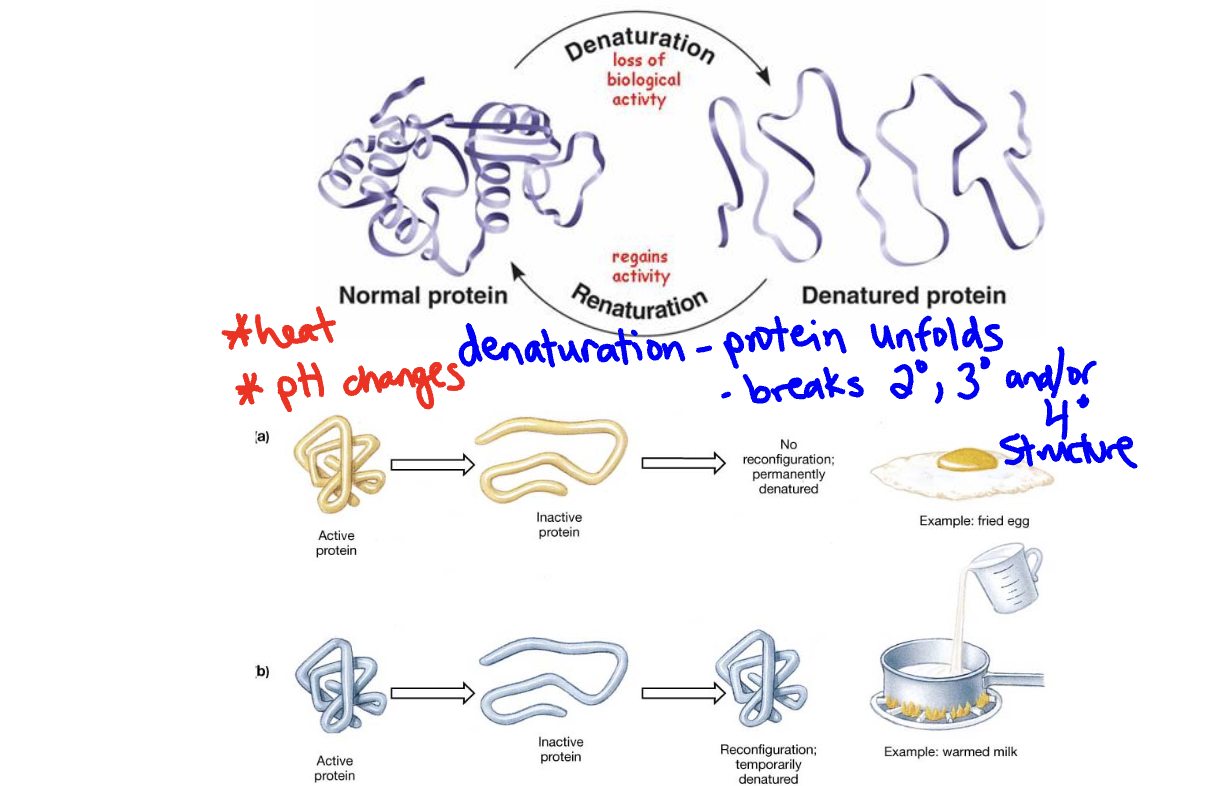
Proteome - all of the proteins produced by a cell, tissue or organism (each individual has their own)
Proteomics - study of which proteins are active in a certain cell at a certain time
Nucleic Acids