2.2G Hybridization
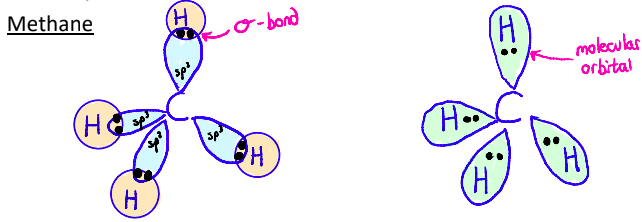
Theoretical and Experimental Structures of Methane
Experimental evidence shows that carbon is tetrahedral
All four C–H bonds are the same
To get methane, carbon would need to have four identical orbitals
Each orbital would hold 1 electron and form one σ-bond
However...
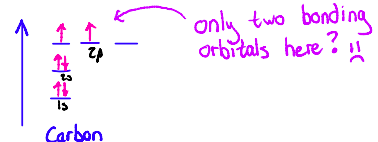 Hybridization
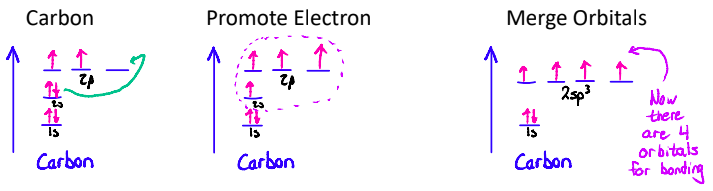
Hybridization
the mixing atomic orbitals to form new hybrid orbitals for bonding.
It involves promoting electrons and merging orbitals in order to get new orbitals, whose shapes match experimental evidence. Hybridization can occur in any covalently bonded molecule.
 Types of Hybridization
Types of Hybridization
sp3 orbitals
Hybridization gives a model that matches bond geometry
sp3 orbitals have 25% s-character and 75% p-character
109.5° apart from one another
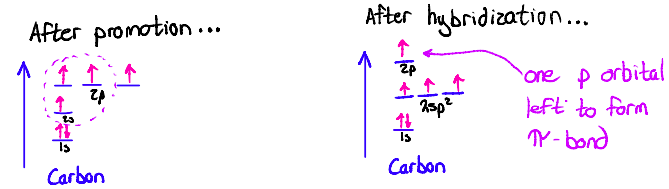
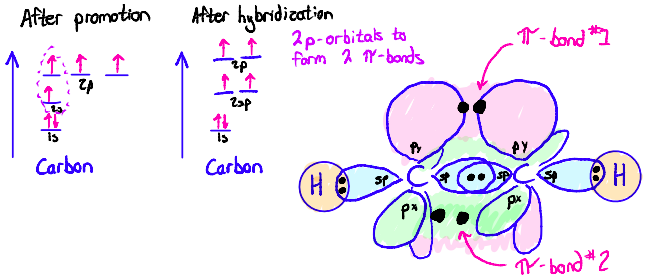
sp2 orbitals
Ex: Ethene
π-bonds form via side-to-side overlap of p-orbitals
If a double bond is present, a p-orbital must still remain after hybridization
 sp2 look like sp3 but are shorter and wider
sp2 look like sp3 but are shorter and wider
sp3 orbitals are oriented 120° apart from one another
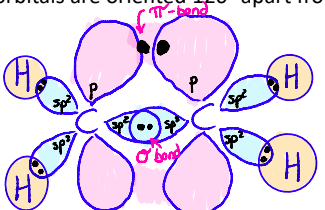
sp orbitals
Ex: Ethyne

 Shortcut for Determining Hybridization
Shortcut for Determining Hybridization
Steps
Look at atom in molecule
Count the number of electron domains
2 E.D. = sp
3 E.D. = sp2
4 E.D. = sp3
5 E.D. = sp3d
6 E.D. = sp3d2