Topic 1.2 - Elements of Life
^^Organic Chemistry^^: The study of compounds with covalently bonded carbon
^^Organic compounds^^: Compounds that contain carbon and hydrogen
- Carbon has , which allows it to form diverse molecules because it still has 4 electrons remaining in its outer shell.
Single, double, or triple covalent bonds can be formed with carbon
- Allows for long chains to be formed by linking carbon molecules together
Most commonly formed with , , and
- Type and number of bonds carbon forms affects the and the
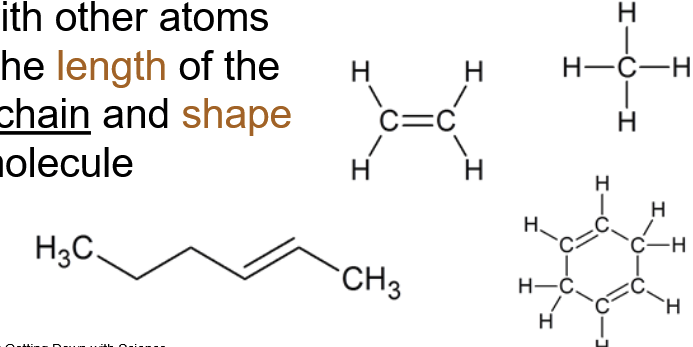
^^Hydrocarbons^^: Organic molecules consisting only of carbon and hydrogen
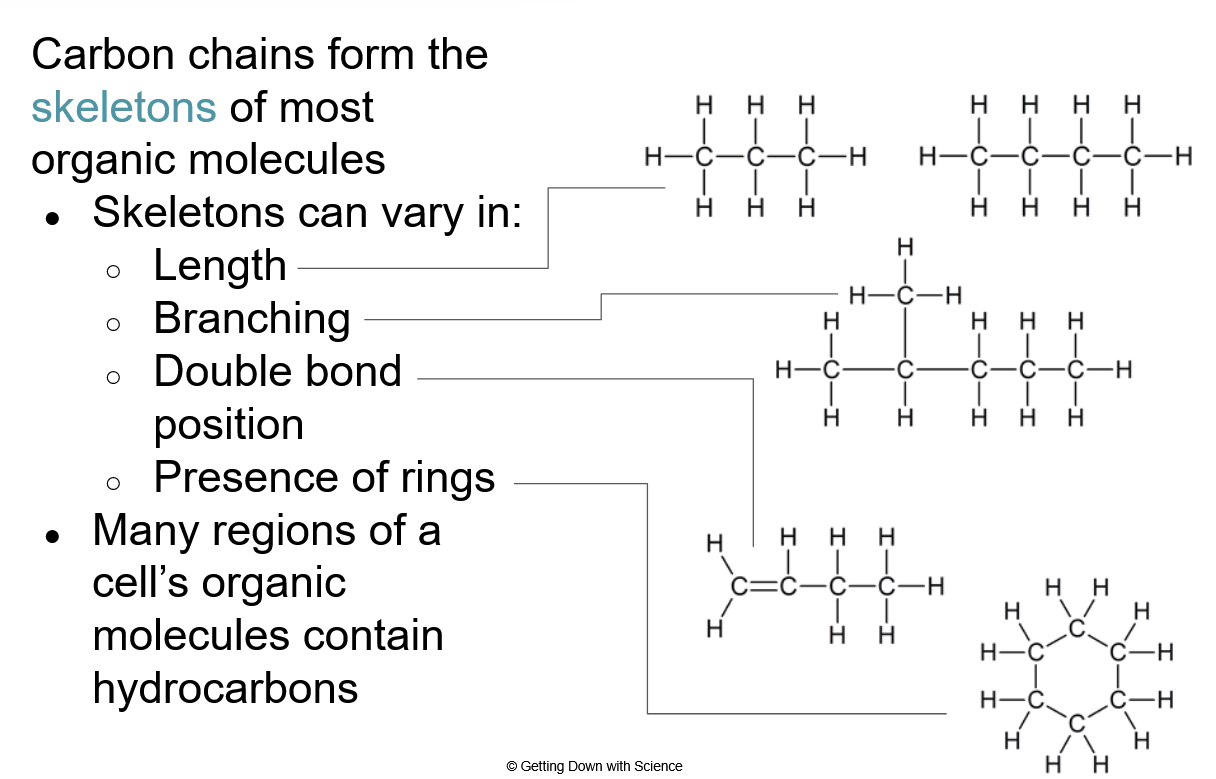
- Framework/skeletons for more complex molecules
They vary in length, branching, double bond positions, and presence of rings.
^^Functional groups^^: Chemical groups attached to the skeleton that participate in chemical reactions.