Unit 3: Chapter 2.1 - Dynamic Equilibrium
Notes
Open Systems: Can exchange energy and matter with surroundings
Closed Systems: Can only exchange energy with surroundings
Irreversible Reactions: a reaction in which the products cannot be converted back to the reactants; only occur in one direction
Reversible Reactions: a reaction in which the products, once formed, can react together to re-form the reactants (⇌ is used to indicate a reversible process)
Equilibrium: achieved in a closed system when the rates of the forward and reverse reactions are equal; it appears as if there is no further change (Cannot be achieved in an open system)
- e.g. N2 (g) + 3H2 (g) ⇌ 2NH3 (g)
- N and H collide and form NH3 (forward reaction). This causes the causes the concentrations of the reactants to decrease and the concentrations of the products to increase
- At the same time, NH3 collides and re-form N and H (reverse reaction)
- Eventually the forward and reverse reactions proceed at the same rate, NH3 is being formed at the same rate as it is decomposing; concentrations remain constant, appears to have stopped to observer.
Saturated Solution: contains the maximum amount of solute dissolved in the solution
Dynamic Equilibrium: when a reversible reaction reaches a point where the rate of the forward and reverse reactions are equal
Extent of a Reaction: indicates how much product is formed at equilibrium
Questions
What is the difference between open and closed systems? In an open system matter and energy can be exchanged with the surroundings, whereas in a closed system, only energy can be exchanged with the surroundings
True/False: The extent of reaction indicates the rate of the reaction, and indicates the time taken to reach equilibrium (F)
True/False: The extent of reaction is the point where there are equal amounts of reactants and products (F)
True/False: The extent of the reaction indicates how far the reaction has proceeded in the forward direction when equilibrium is achieved (T)
True/False: The extent of the reaction indicates the rate of reaction and is the point when the rate of the forward reaction is equal to the rate of the reverse reaction (F)
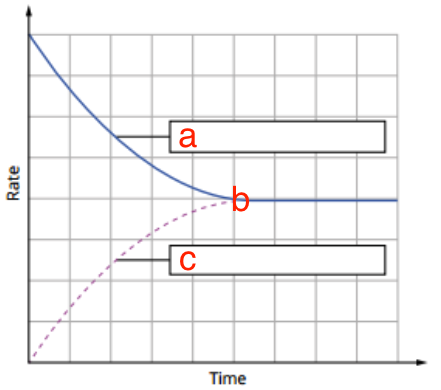
Describe a b and c in the rate-time graph above. a: forward reaction, b: point where equilibrium is first established, c: reverse reaction
Yes/No: Is a salt crystal slowly dissolving in a beaker of water at equilibrium? (No)
Yes/No: Is a saturated sugar solution with sugar remaining on the bottom of the cup at equilibrium? (Yes)
Yes/No: Is an open bottle of perfume at equilibrium? (No)
Yes/No: Is the combustion of a log of wood in a campfire at equilibrium? (No)
Yes/No: Is an open bottle of sparkling mineral water at equilibrium? (No)
Yes/No: Is an unopened bottle of sparkling mineral water at equilibrium? (Yes)