Chemistry as a Foundation for Biology
Big ideas - atoms and portons and electrons, organizing elements in the periodic table
How do we define elements? - specific number of protons and neutrons
How do you know the number of electrons? - same as protons
Why are electrons important? - means by which atoms interact and bond, creating molecules which biologists care a lot about, critical in thinking about energy
What do elements in a column (group) have in common? - same number of valence electrons
Valence electrons - electrons on the outermost orbital
What do elements in a row (period) have in common? - same number of electron orbitals
Elements of life - carbon, oxygen, hydrogen, nitrogen, phosphorus, sulfur, potassium, calcium, sodium, chlorine, magnesium
Basics of Bonding
Chemical bond - a ‘stable’ attraction between two atoms
How are the number of bonds an atom can form determined? - number of unpaired (valence) electrons in the outermost shell
HONC1234 - Hydrogen 1 bond, oxygen 2 bonds, nitrogen 3 bonds, carbon 4 bonds
How is the nature of a bond determined? - relative electronegativities of two atoms
electronegativity - a measure of an elements’ ability to attract electrons
How can you determine electronegativity from the periodic table - going up and to the right is more electronegative
What is the relationship between atomic radius and electronegativity? - the higher the atomic radius, the lower the electronegativity
Why does this relationship exist? - electron orbital is closer to the nucleus in smaller atoms
What type of bonds do atoms that have similar electronegativities form? - equal sharing of electrons
What kind of bonds do atoms with very different electronegativities form? - transfer of electrons
Nonpolar covalent bonds - similar electronegativities, atoms have no charge
Polar covalent bonds - slightly different electronegativities, atoms have a partial charge
Ionic bonds - very different electronegativities, atoms have a full charge because an electron was “stolen”
Why does the addition of a phosphate group have an effect on the structure and function of the molecule? - the oxygens make their end of the molecule more electronegative meaning that it interacts more with polar molecules
Application of Bond Principles: Water, Functional Groups, Organic Molecules
Explain the role relative electronegativity plays in bond formation. - relative electronegativity, or the electronegativity that bonded atoms have in relation to each other, plays a large role in bond formation because when two atoms have vastly different electronegativities, they will form an ionic bond, whereas atoms with some difference in electronegativity will form polar covalent bonds, and atoms with very little or no difference will form nonpolar covalent bonds.
What is the guiding “mantra” of biology? - structure = function
When talking about solutions, what is water referred to as? - universal solvent
Important functional groups - hydroxyl, methyl, carbonyl, carboxyl, amino, phosphate, sulfhydryl
Organic molecules - proteins, carbohydrates, nucleic acids, lipids
Water: The Solvent of Life
Solvent - what you dissolve something into
Why does water have partial charges? - polar covalent bonds within them due to differing electronegativity
Hydrogen bonds - partial charges cause attraction between oxygen of one molecule and hydrogen of another, relatively weak in terms of bonding which allows liquid water to change shape
What happens to hydrogen bonds when energy is added to the system? - hydrogen bonds are broken (boiling and turning to gas)
What happens to hydrogen bonds when energy is added to the system? - hydrogen bonds become stronger (ice formation)
Cohesion - water molecules stick together
Adhesion - water molecules sticking to other surfaces
What kinds of molecules does water “like” to stick to? - polar molecules
Surface tension - when cohesion is greater than adhesion
Specific heat - energy needed to raise temperature
Organic Molecules and Carbon: The Solutes of Life
What properties of carbon make it a logical backbone for organic molecules? - 4 valence electrons allows 4 bonds to occur, tetrahedral shape of bonding, optimal size for electronegativity
If so many molecules have carbon, why are they different? - different structure and other molecules involved
Organic Molecules and Functional Groups
R group - stands in for “something else”
Hydroxyl - polar
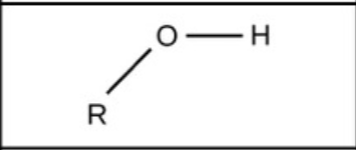
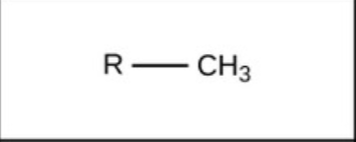
Methyl - nonpolar
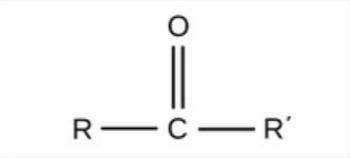
Carbonyl - polar
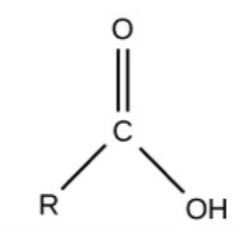
Carboxyl - charged, ionizes to release H+, acidic
Amino - charged, accepts H+ to form NH3+, basic
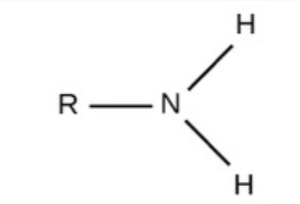
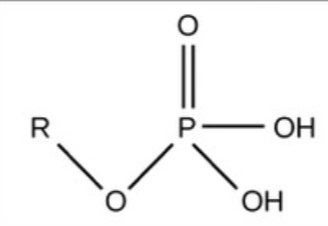
Phosphate - Charged, ionizes to release H+, acidic
Sulfhydryl - polar
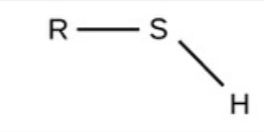
What are organic molecules made up of? - part hydrocarbons (hydrogens and carbons) and part functional groups which typically include oxygen, nitrogen, phosphorus, and sulfur
Hydrophilicity - polar molecules and ions dissolve readily in water
Hydrophobicity - nonpolar molecules do not dissolve readily in water
Amphipathic - a molecule that has both polar and nonpolar properties
What is the structure/function relationship of water that allows it to play such a central role in biology? - the structure with oxygen being more electronegative than hydrogen causes polarity allowing it to interact more specifically with charged and partially charged molecules
Why do hydrophilic and hydrophobic mean “loved by water” and “hated by water” rather than “loving water” and “hating water”? - water is the focus of the word and the concept
What do we mean by “dissolve” chemically? - atoms split from their molecular partners
Culminating in Organic Molecules
Function of proteins - “workers” of life, 55% of cell mass
Structure of proteins - made of amino acids, shapes driven by functional groups
Function of carbohydrates - energy, structural integrity, 10% of cell mass
Structure of carbohydrates - made of sugars, long chains, hydrophilic
Function of nucleic acids - cell energetics, information flow, 25% of cell mass
Structure of nucleic acids - made of nucleotides, mostly hydrophilic, helical in shape
Function of lipids - cell energetics, structure, signaling, 10% of cell mass
Structure of lipids - made of fatty acids, three types, mostly hydrophobic
3 types of lipids - fats, phospholipids, steroids
Why do oil and water not mix? - oil is nonpolar and water is polar which is why they cannot interact with each other
How does salt melt ice? - the salt interferes with the bonds between the water, causing it to change state from solid to liquid
How does guar gum reduce the abundance of ice crystals in ice cream? - guar gum is hydrophilic so it interrupts the bonds between the water
Structure and Function of Organic Molecules
Structure of amino acids: wide range
Function of amino acids: enzymes, signals, structural
Structure of carbohydrates: regular, repeating structures
Function of carbohydrates: cell walls, energy
Structure of nucleic acids: helical
Function of nucleic acids: information
Structure of lipids: hydrophobic or amphipathic
Function of lipids: membranes, energy, signals
Metabolism
Chemical reactions: changes to the sharing of electrons and the rearrangement of bonds
Catabolic reaction: breaking down molecules into subunits/monomers
Anabolic reaction: building molecules into macromolecules/polymers
How to remember CATabolic reaction: cats knock things off counters which makes them break
Hydrolysis reaction: breaking down molecules requires water addition
Dehydration reactions: building molecules removes water from the organic molecule
Formation of proteins: amino acid + amino acid = protein + water; anabolic and dehydration
ATP hydrolysis: ATP + H2O = ADP + Pi; catabolic, hydrolysis
Reaction Energetics
Gibbs Free Energy Equation:
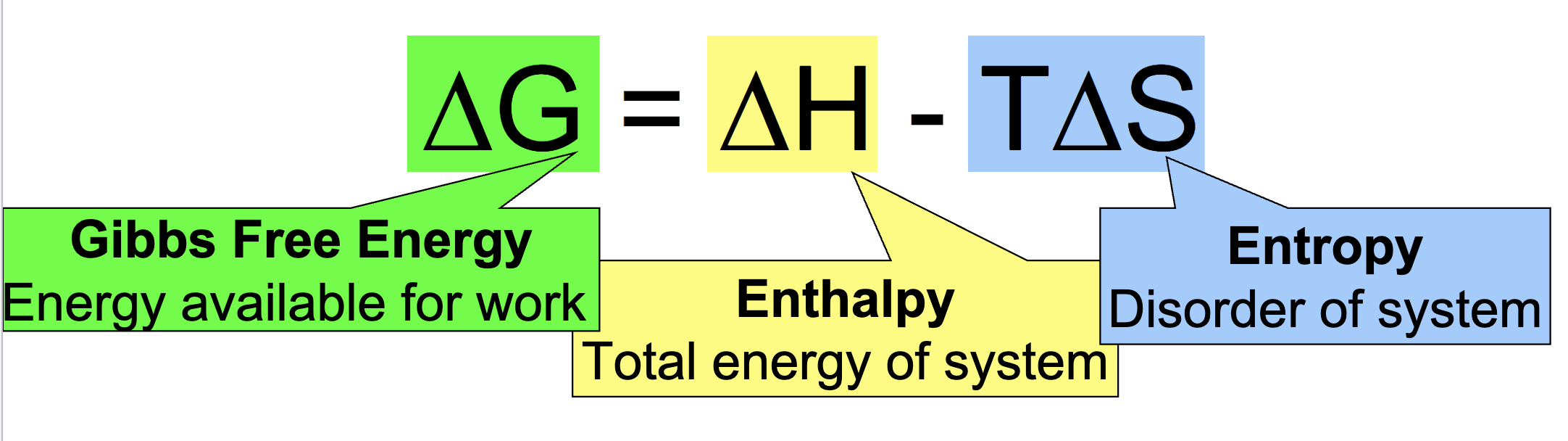
Gibbs free energy: energy available for work
Enthalpy: total energy of the system
1st law of thermodynamics: enthalpy
Why do the electrons in the outermost shells have the greatest energy?: requires potential energy to hold the negatively charged electrons away from the positively charged nucleus
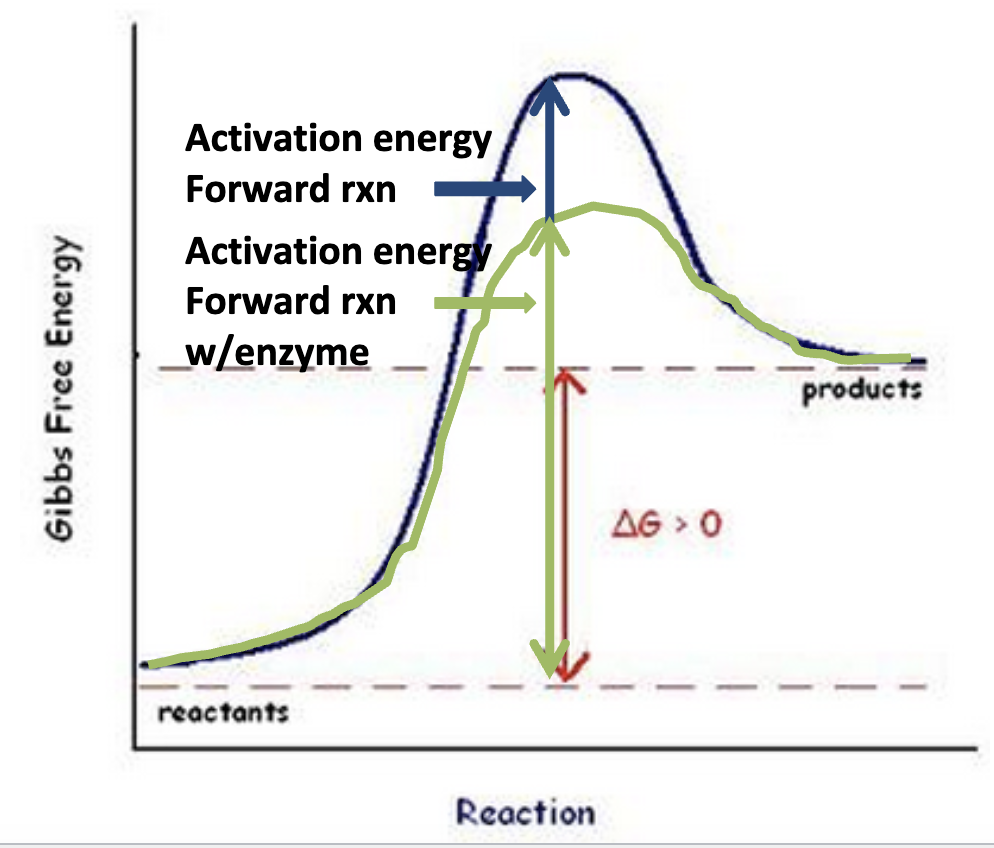
For anabolic reactions, is the change in enthalpy positive or negative?: positive
For catabolic reactions, is the change in enthalpy positive or negative?: negative
Entropy: disorder of system
2nd law of thermodynamics: entropy
For an anabolic reaction, is the change in entropy positive or negative?: negative
For a catabolic reaction, is the change in entropy positive or negative?: positive
Endergonic: anabolic
Exergonic: catabolic
Organic Molecules 1: Proteins
What determines protein shape?: amino acid sequences and the environment
What determines protein function?: protein shape
How do amino acids have diverse chemical identities?: different R groups
Types of proteins: antibodies and complement, contractile and motor, enzymes, hormones, receptors, structural, transport
Amylase: breaks down starch into sugars
Histone deacetylase: determines which genes to express
Sodium/potassium pump: complex protein that works with neurons
Actin: “road” in the cell
Kinesin: motor proteins that “carry” things
Insulin: signaling molecule
Insulin receptor: interacts with insulin in order for the signaling cascade to work
Antibody: y shape, helps fight off bacteria and viruses
Generic amino acid structure:
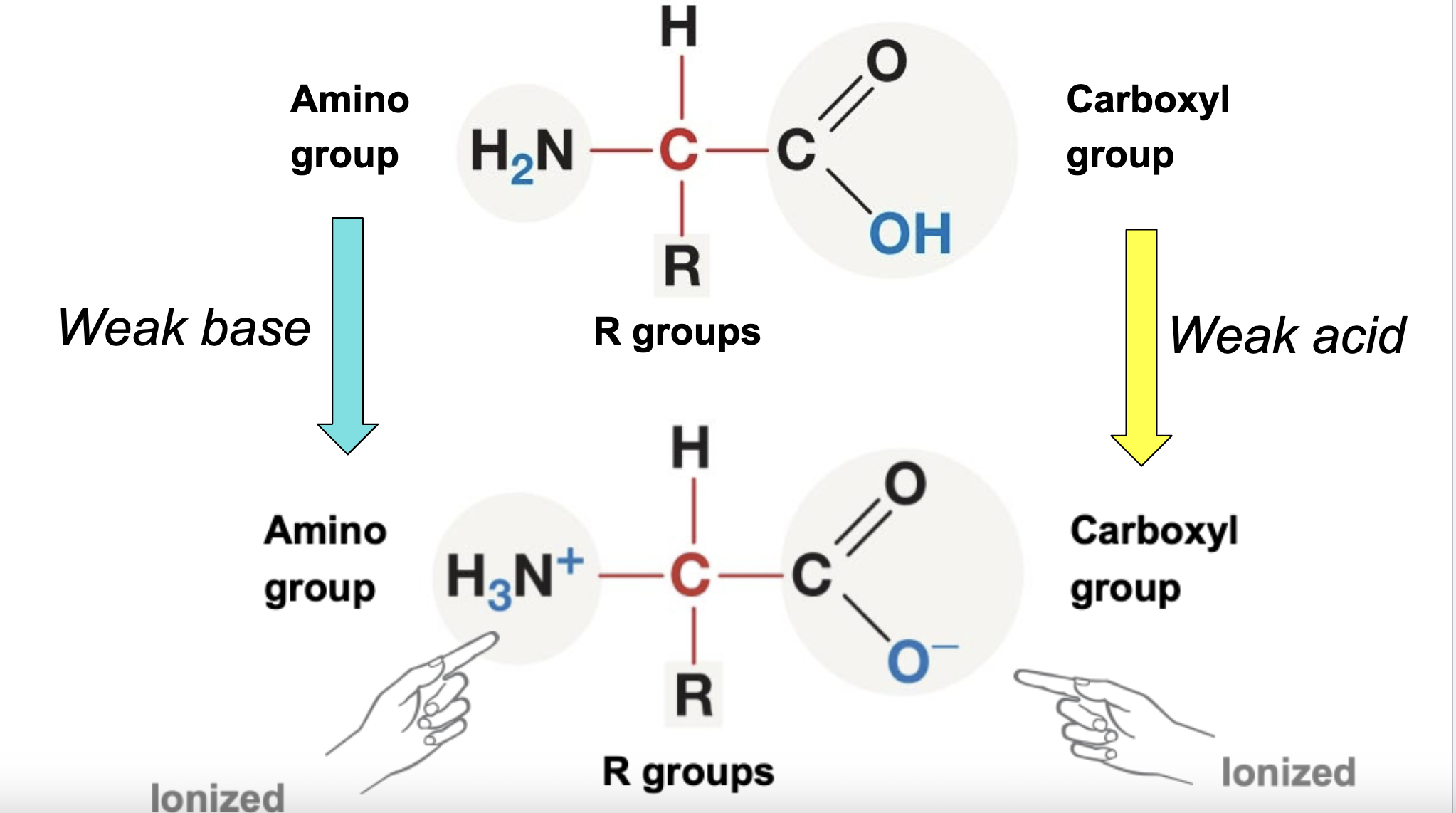
Nonpolar amino acids’ properties: mostly made up of hydrocarbons which have similar electronegativities, meaning that they are non-polar and are hydrophobic, but will interact with each other
Polar and charged amino acids’ properties: many amino and hydroxyl groups, hydrophillic, will dissolve in water and will interact favorably with it
Special amino acids: Glycine, proline, cysteine
What makes these amino acids “special”?: glycine is very flexible, proline is very inflexible
Primary structure: sequence of the amino acids
N-terminus: leftmost group, called this because of amino groups
C-terminus: rightmost group, called this because of the carboxyl group
What do proteins always start with?: methionine
Secondary structure: results from interactions between nearby amino acids
Tertiary structure: three-dimensional shape of a protein, far away amino acids interacting
Quaternary structure: proteins interacting with each other
Alpha helix: each carbonyl group in the backbone forms a hydrogen bond with an amide group
Beta sheets: adjacent strands can run in the same direction or in opposite directions, hydrogen bonds form between carbonyl groups in one polypeptide and amide groups in a different part of the polypeptide
Peptide bonds: bonds between amino acids (forming protiens)
What happens when two cystine molecules interact?: they lose a hydrogen and bond together
Putting Proteins to Work: Enzymes
Enzymes: reusable biological catalysts that perform chemical reactions
Catalyzing: changing the transition state chemistry (and therefore energy) to make the reaction more likely to happen
Why are enzymes necessary?: chemical reactions happen too slowly to be useful in real biological time, allow cells to regulate chemical reactions
Why are two different enzymes necessary for the forward and reverse direction of a reaction?: enzymes are specific to their substrate, different molecules means different substrates
The Protease Trypsin
What does a protease do?: break peptide bonds to catalyze the breakdown of proteins
Active site: the place on the enzyme where the reaction occurs
What amino acids does Trypsin interact with?: lysine and arganine
Reaction Energetics
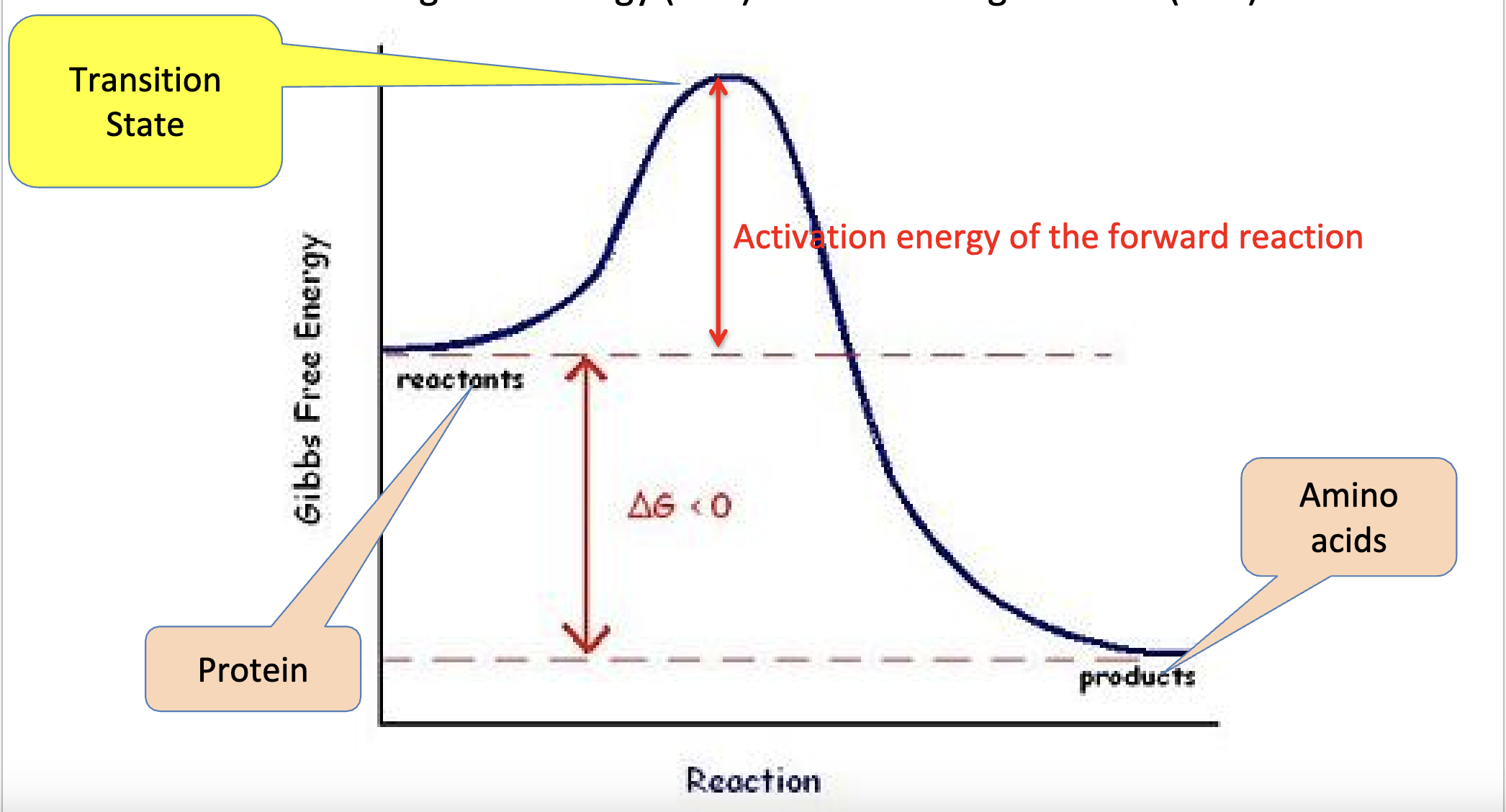
(catabolic) reaction without an enzyme:
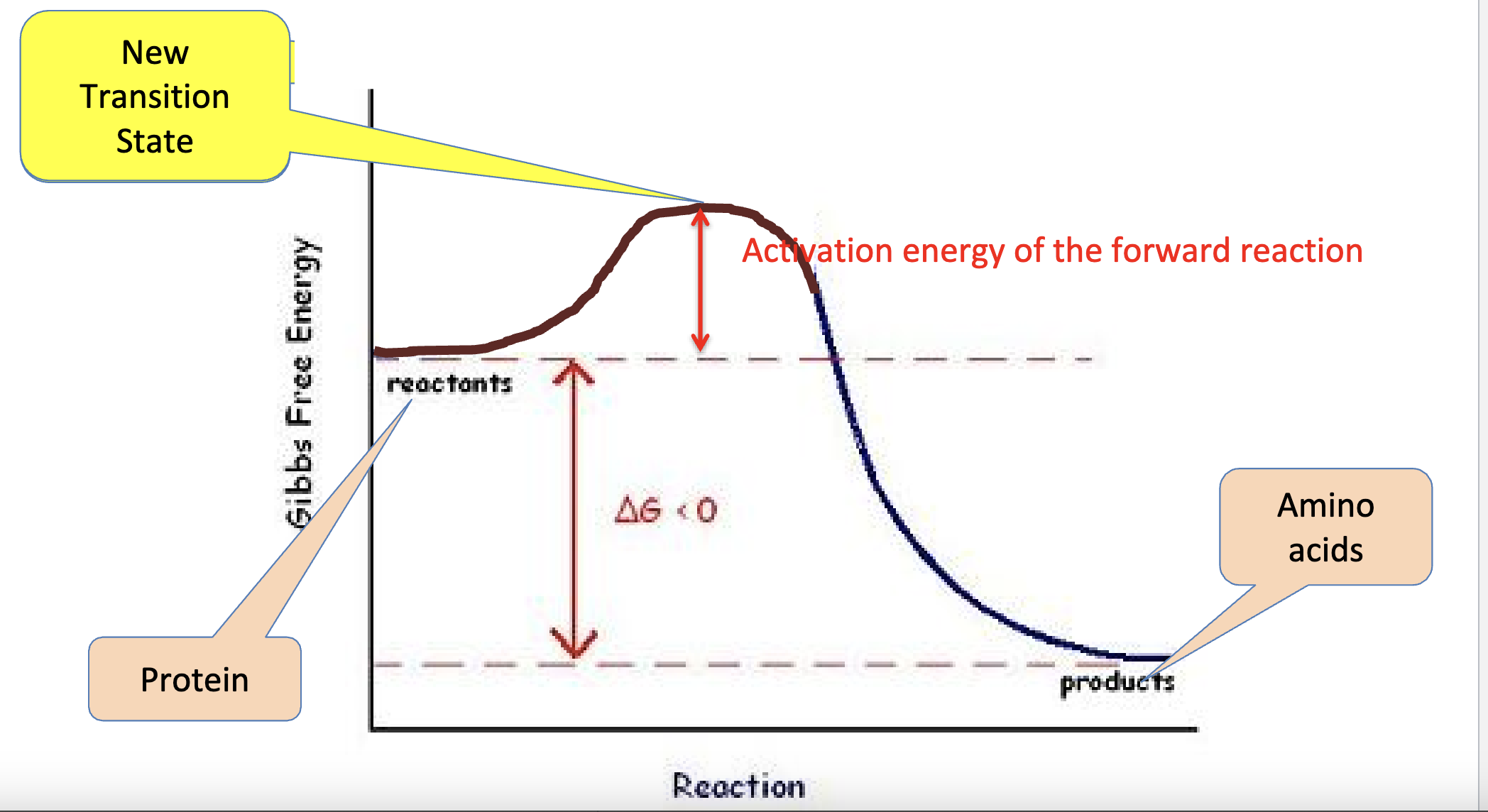
(catabolic) reaction with an enzyme:
Transition state: has an arrangement of atoms and electrons different from either reactants or products
When are transition states less stable?: when they have higher energy (no enzyme to decrease the energy)
Activation energy: the amount of energy it takes to get from the Gibbs’ free energy of the reactants to the Gibbs’ of the products
How do enzymes accelerate reactions?: they change the transition by bonding to the substrate, holding reactants in a proper formation, making the exchange of electrons change
Structure/Function Relationships in Enzymes
Why are enzyme-substrate relationships so specific?: other substrates literally would not fit in the active site
Cofactors
Cofactors: vitamins or a divalent cation, help the enzyme achieve its proper folding and to hold a substrate properly to catalyze a reaction
Do cofactors affect the activation energy?: indirectly by helping the enzyme
Organic Molecules II: Carbohydrates
Functions of Carbohydrates: energy, structure, identity
Structure of carbohydrates: sugars are the building blocks, total structure is determined by the identity and arrangement of the sugars
Monomers of carbohydrates: monosaccharide, sugar, often has the suffix “-ose”
Dimers of carbohydrates: disaccharide, sugar, often has the suffix “-ose”
Polymers of carbohydrates: polysaccharide, carbohydrate, often has the suffix “-ose” and the prefix “glyco-”
How are carbs used for energy?: storage of energy, immediate use of energy (glucose, fructose)
How much of our diet should be carbohydrates?: about 45-60%
How are carbohydrates used for structure: cell walls, exoskeletons, organic molecules like the backbone of nucleic acids
How are carbs used for biological specificity?: cells have sugars on the outside of them that they use to interact with the environment
Structure of carbohydrate monomers: carbon backbone with hydrogen and hydroxyl groups attached, and then functional groups, most sugars have 5 or 6 carbons in the backbone
Spontaneous reaction of sugars: linear form of the sugar forms covalent bonds between an oxygen and a carbon on opposite ends, forming a ring structure in the molecule that is characteristic of monosaccharides. it can also go the other way and changes between forms often
In a cell, would we find glucose in the linear, alpha ring, or beta ring structure?: all of them because of the spontaneity of the change
How does a cell control the specificity of polysaccharide assembly?: enzyme specaficity
How would we draw a Gibbs free energy graph for the reactions that shift the structure between linear, alpha ring, or beta ring structures?: straight line because no energy is entering or leaving the system
Structure of carbohydrate polymers: multiple sugars attached together through an anabolic reaction
Glycosidic bond: bond between two sugars forming a polysaccharide
Is polymerization of sugars anabolic or catabolic?: anabolic
Is polymerization of sugars exergonic or endergonic?: endergonic
Is polymerization of sugars hydrolysis or dehydration synthesis?: dehydration synthesis
Gibbs free energy of an anabolic reaction:
How are carbohydrates used on the outside of the cell?: extend opportunities for favorable interactions with the environment
Organic Molecules III: Nucleotides and Nucleic Acids
Function of nucleic acids (polymers): information storage and utilization
Function of nucleotides (monomers): roles in cell energetics and cell signaling
Function of nucleic acids in energy: ATP, GTP
Function of nucleic acids in signaling: cyclic AMP
Function of nucleic acids in information storage: DNA and RNA
Function of nucleic acids in information utilization: mRNA, tRNA, rRNA
Function of nucleic acids in metabolism: ribozymes
Structure of nucleotides:
Sugar in DNA: deoxyribose
Sugar in RNA: ribose
What is the difference between the sugars in DNA and the sugars in RNA?: there is a hydroxyl group on carbon 2 in ribose, where deoxyribose only has a hydrogen
How is the difference between sugars reflected in the full names of DNA and RNA?: Deoxyribonucleic acid means lack of an oxygen
What is the functional consequence of the difference between the sugars of DNA and RNA?: DNA is better at long-term information storage
What give nucleotides their identity?: nitrogenous bases
Pyrimidines: thymine, cytosine, uracil
Purines: adenine, guanine
What are the 4 DNA nucleotides?: dATP, dTTP, dGTP, dCTP
To which carbon is the nitrogenous base bonded to on the sugar in a nucleotide? What about the phosphates?: 1 and 5
Why is the term “nitrogenous base” both appropriate and redundant?: there is nitrogen in the molecules, but nitrogen makes the molecule have basic properties
What are the differences between UTP and dTTP?: UTP has a hydroxyl group where dTTP does not, different nitrogenous bases
What is the source of energy for the anabolic reactions that build nucleic acids?: triphosphate monomers provide their own energy
Phosphodiester bonds: bonds between phosphate group and sugars
DNA structure: double-stranded, antiparallel helices with sugar-phosphate backbone on the outside and nitrogenous bases hydrogen bonded to each other on the inside
mRNA structure: carries information to make proteins, mostly single stranded
tRNA structure: translator between the language of nucleic acids and that of proteins, usually double stranded
Organic Molecules IV: Lipids
Function of lipids: energy storage (triglycerides), chemical signals (steroids and fatty acids), cell membranes (phospholipids and steroids)
Structure of lipids: no true monomer/polymer, most are hydrophobic which is key in their function
Why are lipids mostly hydrophobic?: hydrocarbon chains (fatty acids)
Saturated fats: single bonds connecting the carbons
Unsaturated fats: some double bonds connecting the carbons
What types of lipids are hydrophilic and why?: some steroids due to hydroxyl groups and phospholipids due to phosphates, amines, and carboxyls
Amphipathic: both hydrophobic and hydrophilic
Structure of steroids: rings fused together
Are steroids hydrophobic or hydrophilic?: hydrophobic
Structure of triglycerides: three long fatty acid tails bonded to a head group made mostly of oxygen
Are triglycerides hydrophobic or hydrophilic?: overall hydrophobic, fatty acid tails are hydrophobic, backbone is hydrophilic
Structure of phospholipids: two fatty acid tails with a hydrophilic head group
Why don’t we need a reaction to form lipid polymers?: since these reactions happen in an aqueous environment, polar molecules need work to break their interactions with water, but fats cannot interact with water so they do not need work to interact with one another instead of water
How do we build a (simple) fat?: glycerol + 3 fatty acids = 1 triglyceride + 3 H2O
How many reactions are required to make one fat molecule?: 3
Are enzymes required for synthesizing fats?: yes for regulation and decreasing activation energy
In a fat synthesis reaction, would the reactants or products have a higher Gibbs’ free energy?: products
What gives fats their identity?: fatty acid chains that are different due to length and saturation
Why are unsaturated fats usually runnier than saturated fats?: saturated fats’ straight structure allows them to stack better meaning that they do not move around as much, making them solid
Why do lipids hold more energy than carbohydrates?: mostly nonpolar
Do polar or nonpolar covalent bonds have more energy?: nonpolar because the electrons are farther away from the nucleus
Would a teaspoon of sugar or a teaspoon of butter have more energy?: butter
Are phospholipids polar, nonpolar, or amphipathic?: amphipathic
What gives phospholipids their identities?: different head groups and fatty acids
How are phospholipid bilayers created?: phospholipids assemble spontaneously
Glycolipids: combination of carbohydrate and lipid
Why are vitamins A, D, E and K called fat-soluble?: nonpolar so they interact favorably with fats
How does soap help remove grease?: soap is amphipathic so its hydrophobic end bonds with the fat (the grease) and its hydrophilic end is attracted to water so the grease and soap can be rinsed away