
CHP207 Human Physiology.docx
CHP207 Human Physiology
Week 1:
Ventilation and FRC Compliance
Compliance
● Ventilation requires:
○ Change in thoracic volume (P1V1=P2V2)
■ Normal Breathing = active inspiration, passive expiration
Ptp, greater lung V).
cavity with thin layer of pleural fluid with a negative pressure of -4mmHg
● Ventilation is affected by: ○ Compliance
○ Airway resistance
Pulmonary Compliance
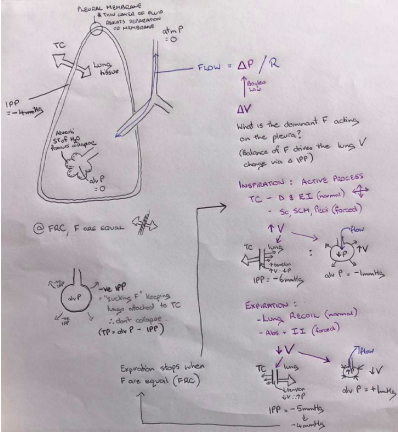

volume
○ Specific compliance = compliance/lung volume
(FRC)
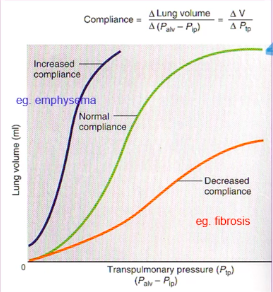
● Lung compliance is affected by:
expansion
ions.
surface tension between alveoli of different sizes
throughout the breathing cycle.
becomes more thinly spread, increasing surface
tension and decreasing rate of inflation
more surfactant therefore decrease surface tension
and slowing contraction of the alveoli
reduces surface tension and decreases the amount of
elastic recoil.
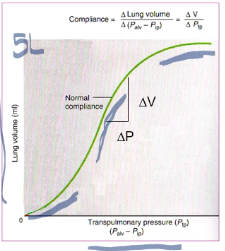
the lung at any pressure.
required to expand the lung.
Week 2
Airway Resistance
mouth and alveoli) and resistance to flow.
them open and allow easy passage of air to and from the alveoli ○ Trachea
■ Has cartilage rings, some smooth muscle ○ Bronchi
■ Irregular cartilage plates offer some rigidity and allow sufficient motion for expansion and contraction, smooth muscle.
○ Bronchioles
the alveoli
mainly pulmonary epithelium, some fibrous tissue and smooth
muscle fibres).
● Resistance to airflow in the airways
○ **exists only when air is moving
respiratory passage (resistance).
○ Poiseuille’s law: R=8ln/πr(^4)
○ Where l=length, n=gas viscosity, and r=the radius of the tube.
increases from the trachea (2-3cm2) to the alveoli (50-100m(^2))
the number of parallel terminal bronchioles (65 000) which air must pass through
the cross sectional area of the airways, therefore the velocity of the air moving through the airways decreases as it travels from the trachea to the periphery of the lung.
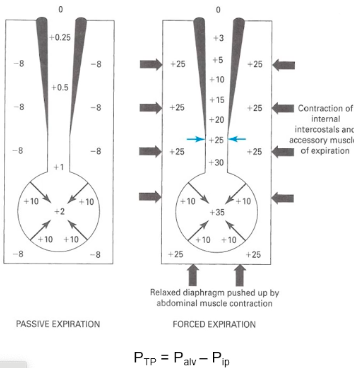
Elastic Recoil & Dynamic Airway Compression
● R is greatest upon expiration
○ Recoil of the lung during expiration is dependent on the condition of the lung parenchyma
○ Bernoulli principle - P will decrease as we move towards the mouth (a
constriction).
Dynamic Airway Compression ● During expiration:
● Forced breathing increases that pressure (from
muscles pushing)
pressure in the airways, compression of the airway may occur
If disease:
○ Can occur in diseased individual which is when gas is trapped in alveoli
because of the increase intrapleural pressures
Bronchial Smooth Muscle Tone
● Smooth muscle in the airways is under control of efferent fibres of the ANS
■ Released by mast cells, cause bronchoconstriction Work of Breathing
Week 3:
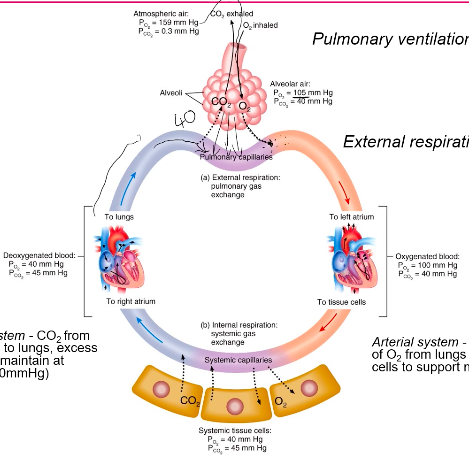
Pulmonary Diffusion & Gas Transport Atmospheric Air
Alveolar Air
The composition of alveolar air is not the same as that of the atmosphere because:
the air.
Therefore the composition of gas exchange as it is inhaled from the atmosphere, through the respiratory passage and to the alveoli.
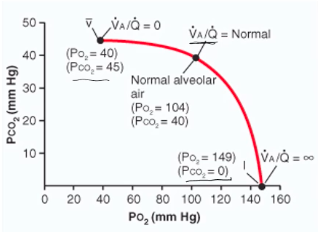
The alveolar Partial Pressure of Oxygen (ideally 104mmHg) and carbon dioxide (40mmHg) is determined by:
Ventilation (VA) and perfusion (Q) Regional differences in ventilation
Matching Ventilation and Perfusion (VA/Q)
gas exchange and influence alveolar partial pressures.
○ Alveolar ventilation of ~5L/min and pulmonary blood flow of ~5L/min ● A mismatched can result in:
without undergoing gas exchange remains only partially oxygenated. This in effect ‘dilutes’ the oxygenated blood entering the left atrium.
■ In addition, some venous blood from coronary and bronchial circulation is added to the arterial blood diluting it slightly with venous blood (ie. normal anatomical features and venous admixture - slightly similar to dead space).
venous admixture
respiratory dead space.
category.
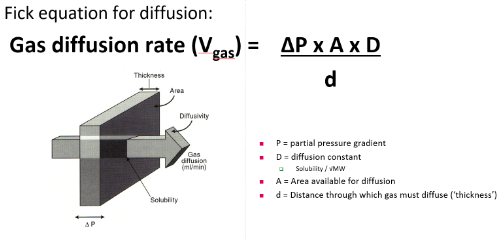
Diffusion across the Respiratory membrane:
following equation:
●
Anatomical Factors:
Factors relating to respiratory gases:
Partial pressure gradient:
What is the normal partial pressure gradient across the respiratory membrane for PO2 and PCO2?
PO2= 40 mm Hg and a PCO2= 45 mm Hg???
Diffusion constant: CO2 is 23 times more stable than O2, which molecule has a larger molecular weight? CO2
Diffusion Gradients
gradient.
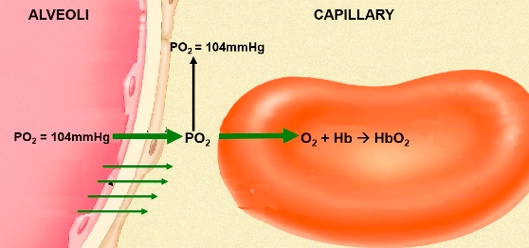
Gas Transport Oxygen
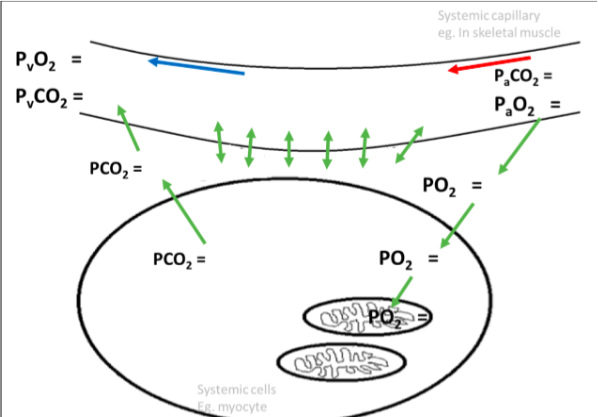
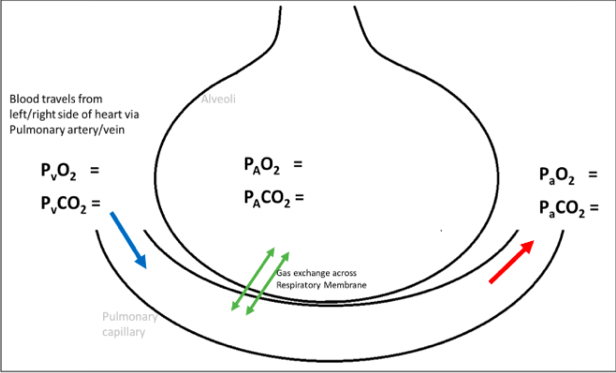
Alveolar air:
Po2 = 105mmHg Pco2 = 40mmHg
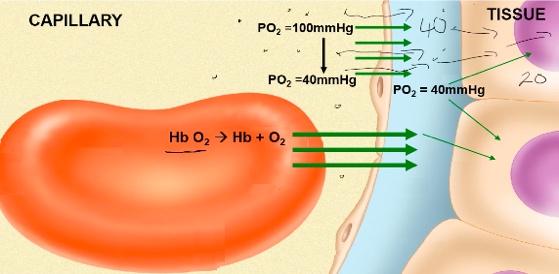
Oxygenated Blood: Po2 = 100mmHg Pco2 = 40mmHg
Systemic tissue cells: Po2 = 40mmHg Pco2 = 45mmHg
Deoxygenated blood: Po2 = 40mmHg Pco2 = 45mmHg
Transport of Oxygen
● Only 3% of oxygen travels dissolved in the blood
○ Henry’s law
Hb+O2 -> Hb-O2
(4 times resting O2 requirement).
Haemoglobin (Hb):
○ Two alpha and two beta chains
Hb ranges from 0-4
○ Pulse oximetry (absorbance of light to determine saturation)
molecule which increases its affinity for O2, and therefore enhances binding
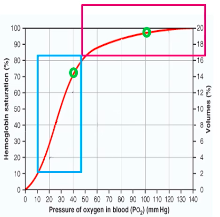
Oxygen-Hb Dissociation curve
saturation
saturated
(P50 for Hb) At lungs:
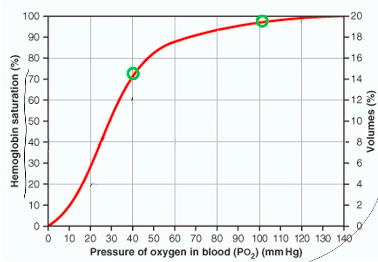
○ 50ml/L O2 delivered to the tissues for a ~60mmHg change in PO2
Factors which ‘shift’ the curve
● There are a number of factors which will change the
affinity between O2 and Hb, and therefore shift the curve left/right
○ Shift in the curve will change the P50 ● These include CO2, pH, temperature and
2,3-diphosphoglycerate (2,3 DPG or BPG). These factors facilitate
loading/off-loading of O2 at the lungs or tissue. Bohr Effect: pH and CO2
● At the tissues, the increase in blood CO2 and H+ enhances the release of O2 from the blood into the tissues
○ Decreased affinity -> right shift, increase P50 (increased oxygen off-loading) ● At the lungs, decrease in CO2 and H+ enhances oxygenation
○ Increased affinity -> left shift, decrease P50 (increased oxygen loading)
Hypoxia: low O2 at tissues
aerobic metabolism
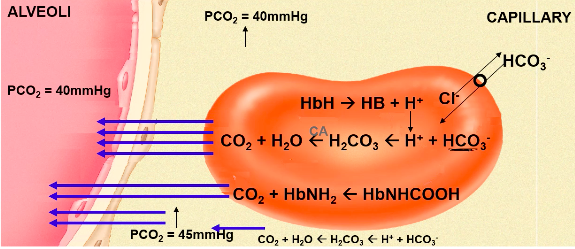
Carbon Dioxide
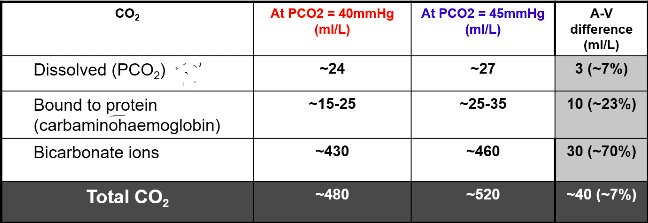
● We carry CO2 in 3 forms:
CO2 Transport:
● 7% dissolved in plasma
○ It is this form that contributes to PaCO2
●
carbonic anhydrase (CA)
○ Facilitates CO2 loading at tissues, offloading at lungs
Carbon Dioxide dissociation curve
More linear relationship between PCO2 and CO2 in blood
Much steeper than the oxygen dissociation curve
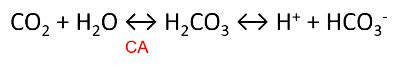
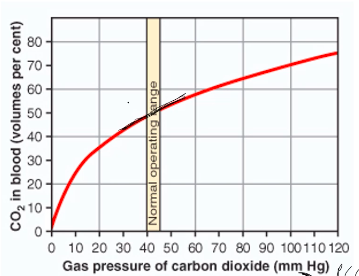
At tissue:
At Lungs:
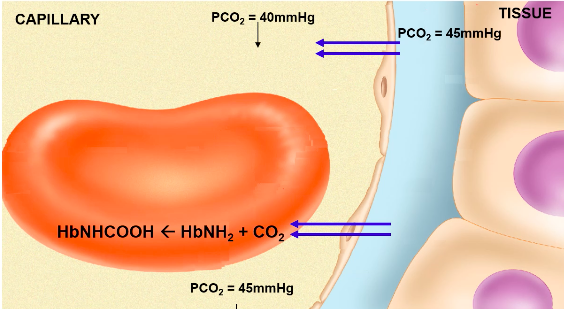
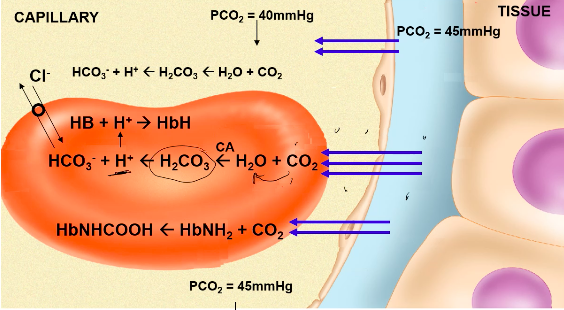
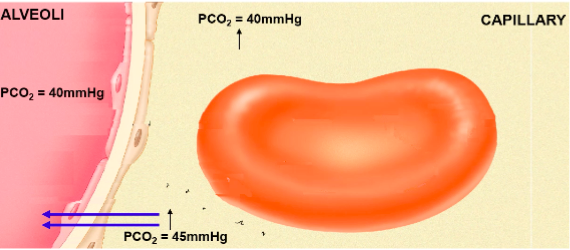
Bohr Effect: pH and CO2
● Bohr effect - how CO2 and H+ affect Hb’s affinity for O2?
○ At the tissues, the increase in blood CO2 and H+ enhances the release of oygen from the blood into the tissues.
■ Decreased affinity -> right shift, increase P50
○ At the lungs, decrease in CO2 and H+ enhances oxygenation
■ Increased affinity -> left shift, decrease P50 ● This occurs because:
affinity of Hb for O2. Haladane effect
carbonic acid are buffered by Hb
carry H+ ions
becomes a more effective buffer -> ‘haldane effect.’
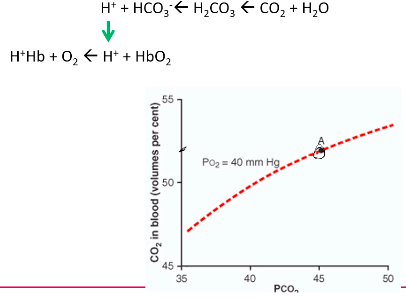
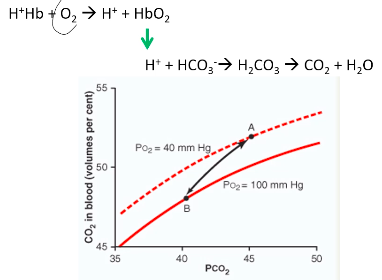
excess H+ ions and displaces carbon dioxide
Carbon Dioxide and regulation of acid-base balance
Excretion of CO2 by the lungs is an important mechanism in the regulation of acid-base balance
CO2 in the blood:
●
dioxide (VCO2) and inversely proportional to PCO2.
○ Eg 5L/min = 0.863 x 200ml/min / 40mmHg
Hypercapnia = elevated PaCO2 Hypercapnia may be caused by:
Week 4: Ventilation/ Perfusion Matching
● For efficient and adequate gas exchange to occur, ventilation must be matched to perfusion.
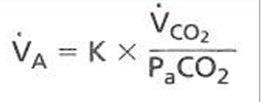
V/Q Matching
● In the lungs as a whole, ventilation and perfusion are well matched (ventilation is
~5L/min matched to cardiac output of ~5L/min). However there are regional differences in both ventilation and perfusion which lead to natural regions of mis-match.
Ventilation (V)
● There is a mismatch in ventilation due to gravity and the alveolar being smaller and
tightly packed in the bottom of the lung. This means that ventilation is greatest at the
bottom of the lung due to greater compliance of the alveoli and a larger SA. Perfusion (Q)
● In an upright subject, ventilation increases more slowly than blood flow in the apex of the lung to the base. Hence, the V/Q ratio at the apex of the lung is much greater than 1. Whereas the V/Q ratio at the base of the lung is much less than 1.
V/Q ratio and pulmonary gas exchange Ventilation
● Apex
● Base
Perfusion
● Apex
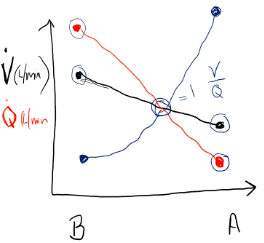
● Base
● VA/Q
○ No VA but normal Q (VA/Q=0), where units
ratios can range across extremes from:
contribute to the venous admixture
become part of the respiratory dead space.
within the ‘normal’ category.
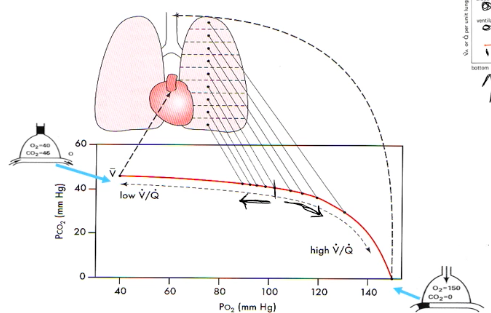
Regional Differences Alveolar PO2/PCO2
(under-perfused) has higher PO2 and lower PCO2 than at the base, which is relatively under-ventilated (over-perfused).
Effect of Exercise on V/Q ratio
● Exercise, by recruiting more alveoli and more pulmonary capillaries throughout the
whole of the lung, but more especially in the apical regions, produces a smaller range of V/Q ratios.
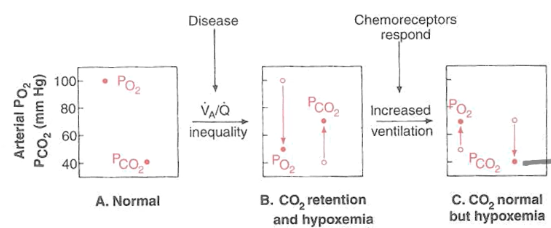
Disease and V/Q mismatch
production).
○ Embolism or thrombosis.
EG. V/Q mismatch
● V/Q mismatch and PCO2/PO2 and chemoreceptor response
○
Control of Breathing Medullary Respiratory Centre
○ The most important of these are:
■ Dorsal respiratory group (DRG) ● Inspiratory centre
■ Ventral respiratory group (VRG) ● Expiratory centre
● The DRG comprises entirely of inspiratory neurons, whereas the VRG comprises both inspiratory and expiratory neurons.
VRG and forced breathing
forced expiration.
VRG expiratory neurons.
Pontine Respiratory Centre
medullary respiratory centre and fine-tunes respiratory rhythm.
phase of breathing.
● The apneustic centre in the lower pons prolongs inspiratory activity
○ The pneumotaxic centre usually keeps the apneustic centre suppressed.
Factors which alter ventilation
● THere are a number of factors which can influence the respiratory centre and alter
breathing pattern:
Central and Peripheral Chemoreceptors
The chemical control of breathing is one of the main regulators for our ventilatory pattern helping us to achieve our goal of maintaining arterial partial pressures and pH.
● Central chemoreceptors respond to increase in CO2 and H+ (low pH).
○
○ Doesn’t respond to O2.
Aortic Body and Carotid Body:
glomus cells.
their neurotransmitters out.
potential down the neuron.
potentials being sent along the neuron.
and therefore CO2 builds up in the cell, setting off the vesicles. Additionals points:
● Chemoreceptors only detect/respond to the dissolved for of each gas - ie. PO2 and PCO2 - they do not detect changes in the oxygen and/or carbon dioxide that is bound to Hb.
response. Essentially the rate of firing from the chemoreceptors will decrease (“quieten”).
Chemical Control of Breathing
● Among the most important inputs to the brainstem control centres originate in the
chemoreceptors
○ Centrally located in the medulla
■ Changes in PCO2, H+
○ Peripherally located in the aortic and carotid bodies
■ Changes in PO2, PCO2, H+ Peripheral Chemoreceptors
forms a branch of the glossopharyngeal cranial nerve (IX) which projects to the
medulla.
Peripheral Chemoreceptors: PO2
ventilation?
PO2, we still have a high range of % saturation and can safely deliver enough
oxygen to cells.
drops drastically.
Peripheral Chemoreceptors: PCO2 and H+
● Peripheral chemoreceptors respond to PCO2 in arterial blood.
○ They respond more rapidly than central chemoreceptors, but overall provide
only 25-30% of the total ventilatory response to PCO2.
● Unlike the central chemoreceptors, peripheral chemoreceptors respond to changes in arterial [H+].
○ Eg. ketoacidosis.
● Unlike O2, HCO2 only needs to change by 1mmHg to get a response.
PACO2 and ventilation
linear.
there is a significant increase in the slope of ventilatory response to CO2. Chronic effects of Hypercapnia
~1/5th its initial effect due to renal compensation which normalises pH.
○ Eg. COPD - increased PCO2, with normal blood pH.
Week 5
Nervous System Membrane Potential
would take to move a charged particle (eg ion) from point A to point B.
particles in that area and there you are going up a concentration gradient.
charges to positive voltages.
force trying to drive them inside the cell.
Potential
Potential DIfference
Charged particles can move in a conductor
Important Points:
voltage.
The Equilibrium Potential
respectively).
respectively).
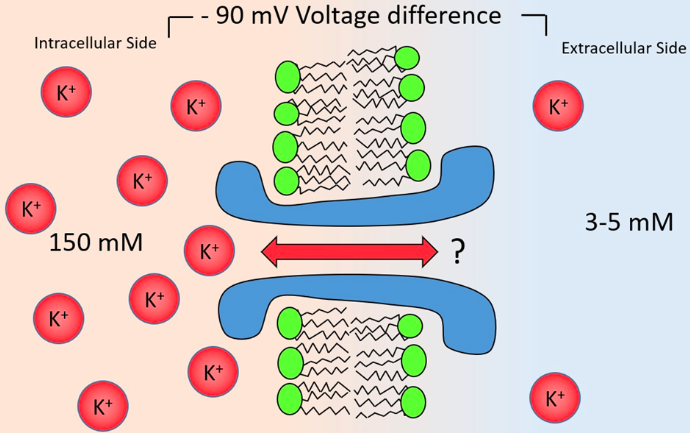
+40mV.
far fewer of them.
than Na+.
○
amount of substance and temperature.
amount of a substance and its electric charge.
were allowed to flow.
○ To calculate where the membrane potential actually gets to, you need to know
how easily the ion flows, as the bigger a proportion of the total ionic flux an ion makes up, the closer the resting potential will be to that ions equilibrium potential.
The resting membrane potential and Action potentials Electrochemical Gradient
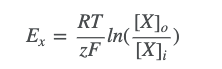
outside.
to draw it back in.
enough potassium going out to reach equilibrium potential.
Nernst Equation
● At the equilibrium potential for ion X (Ex), the two balance:
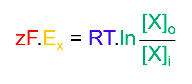
○ ○ ○ ○ ○ ○ ○
●
Z: valence for ion
F: faraday’s constant (96,500 coulombs per mole) R: universal gas constant (8.314 J deg-1 mole-1)
T: absolute temp (kelvins; 0oC = 273K, 37oC = 310K) [X]o = concentration of ions inside cell
[X]i = concentration of ions inside cell
- rearranged formula to find equilibrium potential
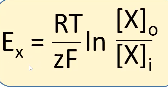
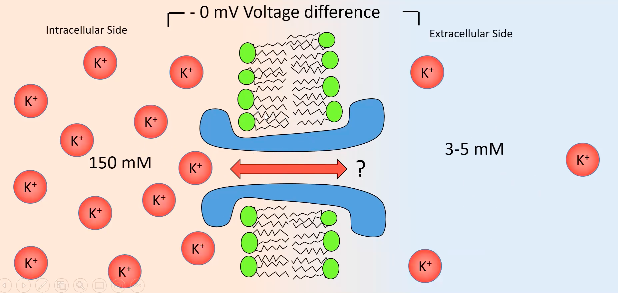
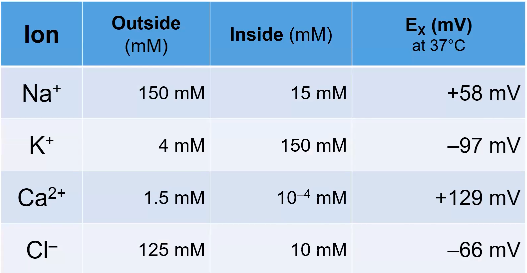
Equilibrium potential:
Eg:
RT/F = 27 “mV” Potassium: [K+] = 4mM
[K+] = 150mM Z = +1
Hint: think of cells inside sea water, salt is high in water, therefore NaCl is high outside cell.

Calculating Resting membrane Potential
as the reciprocal of resistance.
and low resistance.
●
Important Points:
to the flow due to concentration gradient.
membrane potential of a cell.
ion channels.
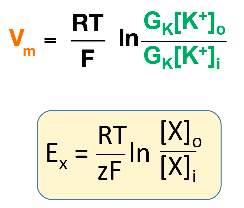
![]()
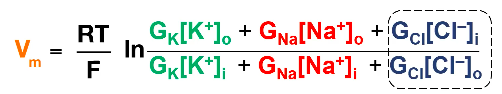
Action Potential
(H+H = Hodgkin + Huxley)
must be associated with the flow of an ion with an equilibrium potential of +40mV or
above - Na+ (+40mV) and Ca2+ (+120mV).
happens if you remove Ca2+ so therefore action potential must involve Na+.
increase in the conductance of an ion with an equilibrium of -70mV or lower - K+
(-90mV) and Cl- (-70mV).
K+ involved.
conductive to Na+ ions.
and become very permeable to K+
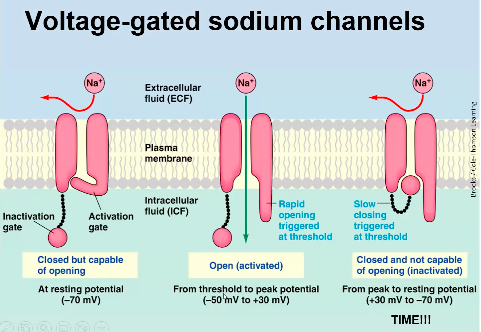
What currents cause the action potential?
Voltage Gated ion channels:
potential to depolarise a little bit, this then causes a few voltage gated sodium channels to open (activate).
○ After being open for a millisecond
or two, the inactivation gate slams shut, putting the gate into an inactivated state.
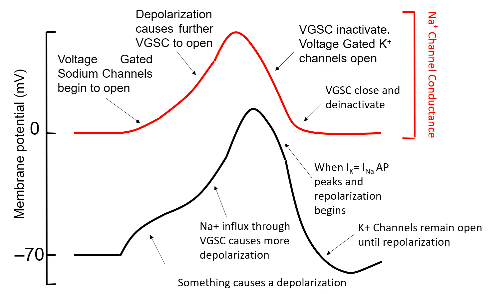
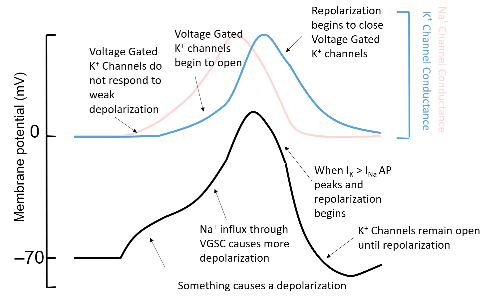
1. They conduct K+ not Na+
2. They do not inactivate: if the cell is more depolarised than about -40mV, these
channels open.
3. Importantly, these channels open more slowly than Na+ channels.
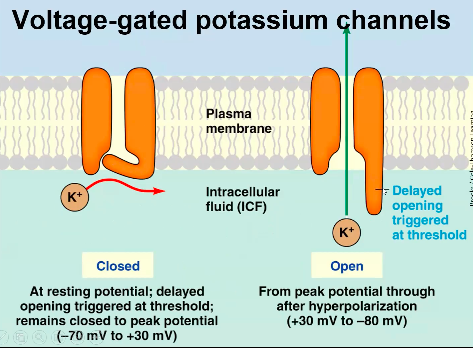
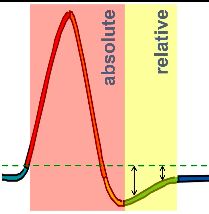
Relative Refractory Period
further than usual from threshold.
to excite to threshold.
larger than usual stimuli.
Important Points:
channels.
Week 6 - Nervous System II
Neurotransmission
other neurons.
to the synapse.
initial segment.
channels, this not only depolarises the axon initial segment, but it also beings to
weaky depolarise nearby parts of the axon.
sodium channels to cause the action potential to begin there too.
sodium channels to open, and so on, down the length of the dendrite.
What happens when the action potential gets to the end of the axon?
presynaptic terminal.
channels to open, allowing Ca2+ to enter the presynaptic terminal.
them to eject their contents into the synaptic cleft, the small (20nm wide) gap between the presynaptic terminal and the postsynaptic cell.
● The time delay between when Ca2+ influx occurs and synaptic release happens is as brief as 100μs.
● This is notable bc Ca2+ diffuses very slowly within the cell due to the presence of a huge variety of proteins which bind calcium.
● This means that in order for release to happen this quickly, the neurotransmitter containing vesicles must be physically very close to the voltage gated Ca2+ channels.
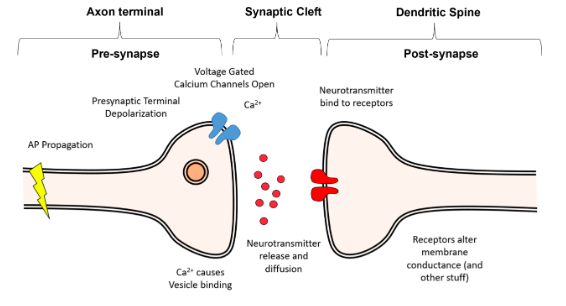
Synaptotagmin - the calcium sensor of the vesicle, once calcium activates synaptotagmin, the process of exocytosis begins.
The SNARE Complex - a complex of proteins which tether the vesicle to the membrane and force it into the membrane to cause release, once synaptotagmin is activated by Ca2+.
● Once released into the synapse, the neurotransmitters simply diffuse across the synapse.
● Diffusion can be very slow over long distances, however, given the minute distance the neurotransmitters have to diffuse over the synapse (~20nm), the diffusion is almost instant on a biological time scale, taking only a few nanoseconds.
A = Action potential
B = Ca2+ entering the cell just after the action potential starts (measured in Amps).
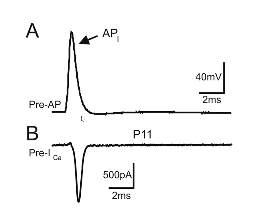
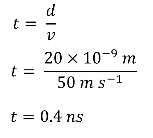
Overview
Glutamate Neurotransmission
(there are approx 100 in total).
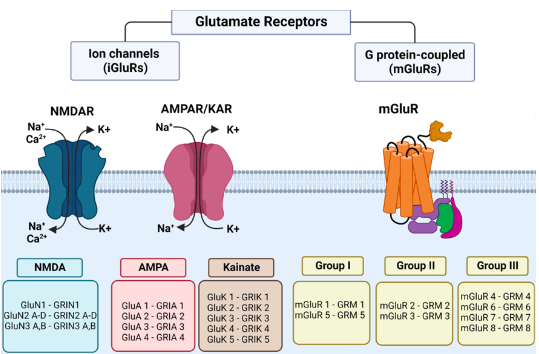
Glutamate Receptors
● Glutamate acts on glutamate receptors, which fall into 3 broad classes:
3. Metabotropic glutamate receptors - which commonly have the name mGluR with receptor number.
AMPA/KA Receptors
acid (KA) allowed the identification of two receptors found on almost all neurons.
close to 0mV.
and GluA4 subunits, and typically made up of 2x GluA1 + 2x GluA2 OR 2x GluA2 + 2x
GluA3.
NMDA receptors
GluN2C, GluN2D, GluN3A and GluN3B subunits.
and hence have an equilibrium potential near 0mV.
Ca2+ is about 10% that of Na+/K+, so it doesn’t really effect their equilibrium potential
but the permeability is enough to provide a massive source of calcium).
become significantly conductive when the cell is depolarised beyond -40mV.
receptor is impermeable to Mg2+ and hence it essentially plugs the NMDA receptor.
receptor, allowing Ca2+ (and Na+/K+) to enter/exit the cell.
allows strong excitatory input to leave a biochemical “footprint.”
Ca2+ will enter the cell, modulating Ca2+ sensitive enzymes/proteins.
Metabotropic Glutamate Receptors (mGluRs)
trimeric protein which splits in two when activated.
beta/gamma subunits module ion channels.
GABA Neurotransmission
amino acid glutamine.
GABAA Receptors
binding - in this case chemical GABA].
receptor different properties.
and are often expressed in different locations and drug designers had high hopes for
being able to treat specific diseases by targeting different types of GABAA receptors.
membrane becomes more permeable to Cl-.
○ That depends on the equilibrium potential for Cl- in that cell.
● Does this mean that GABA has no action in these neurons?
○ No, It means that if any other source of depolarisation is impacting on the cell,
then the action of the GABAA receptors will work to bring the membrane
potential back to the Cl- equilibrium potential/resting membrane potential.
cell to the AP threshold.
GABAB Receptors
their ligand, they activate the G-protein biochemical cascade.
differs on where they are located.
gated calcium channels, meaning that when the action potential invades the presyaptic terminal, less Ca2+ enters the terminal, and hence less neurotransmitter is released.
Neurotransmitter Clearance
from the synaptic cleft.
gradient, so the concentration of neurotransmitters outside the synapse must be kept
close to 0.
neurotransmitters, or by transporters, which pump the neurotransmitters into a cell.
transporter of glutamate is particularly noteworthy.
○ Once glutamate is released, it is taken up by glutamate transporters on the
processes of astrocytes (type of glial cell) that surrounds excitatory synapses.
Week 7 - Nervous System III
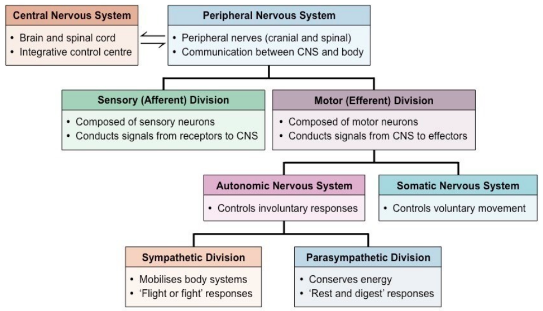
Coordinated Action of the Nervous System
movement and control of involuntary responses which can maintain organ function.
recived in the brain and activation of the somatic motor system to
initiate a motor action.
system which control involuntary responses.
■ Effector actions include reflexes, coordination and optimisation which
tunes the body to suit certain behaviours or actions.
○ Endocrine - these responses are unto the control of the hypothalamus that
can control endocrine output.
■ Effector actions include systemic control and whole organism action as
a result of secretion of hormones that influence function. Overview of Nervous System:
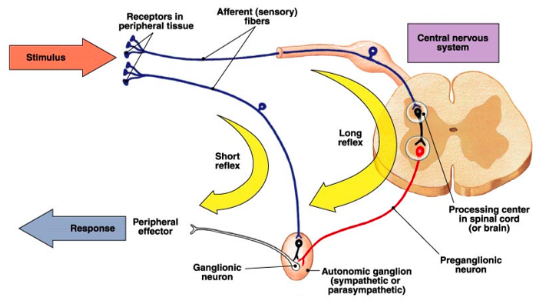
Autonomic Nervous System:
both maintenance and activity of specific organs/tissues.
performance.
conservation and rebuilding.
activation of the SNS or the PSNS depending on the circumstances.
responses.
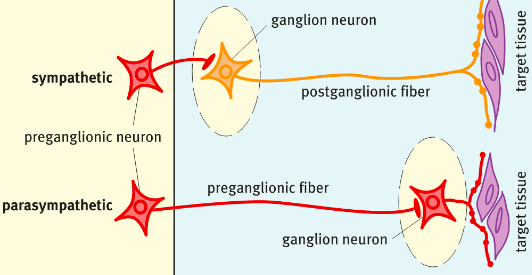
cardiac muscle, smooth muscle and glands.
heavily myelinated axons that extend in spinal or cranial nerves down to skeletal
muscles, the ANS uses a two-neuron chain to its effectors.
spinal cord and their axons are lightly myelinated thin fibres.
which sits outside the CNS.
effector tissue.
Sympathetic Nervous System (SNS)
Sympathetic Action | Target Tissue |
Increased HR | Heart |
Increased BP | Blood Vessels |
Relaxes and opens airways | Lungs |
Mobilises energy stores | Muscle and Fat cells |
Increases blood flow to muscles | Blood Vessels |
Stimulates sweating | Sweat glands |
Shunts blood to heart, lungs and brain | Blood vessels |
Suppresses digestion | Gastrointestinal (GI) system |
Relaxes bladder and rectum | Urinary and GI systems |
Action of the SNS on target tissues
the release of the action hormone adrenaline or noradrenaline directly to tissues,
orchestrating a coordinated response of many organs.
into the bloodstream.
ganglia near the CNS.
synapse in the sympathetic chain ganglia.
prevertebral ganglia (located away from spinal cord).
cord.
tissues such as sweat glands, hair erectors, blood vessels, heart and lungs (stellate
ganglion), salivary glands and pupils (cervical ganglia).
their effector tissue.
Parasympathetic Nervous System (PSNS)
Parasympathetic Action | Target Tissue |
Decreased HR | Heart |
Decreased BP | Acts via heart rather than directly on vessels |
Closes off airways | Lungs |
Constriction of pupils | Eyes |
Boost digestive system | GI system |
Contracts bladder and rectum | Urinary and GI systems |
Action of PSNS on target tissues:
(ACh) to target tissues.
○ eg. boosting the digestive system occurs through improving GI smooth muscle activity, GI blood flow, digestive enzyme secretion and GI hormone secretion helping nutrient absorption and use.
brainstem nuclei, and the sacral outflow from the lateral horn of the sacral spinal
cord.
(to the lacrimal and salivary glands), 9 (to the parotid gland) and 10 (to the vagus nerve to viscera).
Neurotransmitters of the ANS
adrenaline/noradrenaline.
using ACh on nicotinic ACh receptors.
tissues.
adrenaline/noradrenaline to act on adrenergic receptors on target tissue.
muscarinic receptors on target tissue.
Receptos:
act as ligand-gated ion channels for the cation Na+, K+ and Ca2+.
through depend on the subunit makeup.
PSNS at the first synapse.
nAChRs on skeletal muscle as part of the somatic nervous system.
tissue distribution allows them to have different actions.
the parasympathetic functions.
cells to elicit their function.
reside in, eg. activation of alpha receptors leads to vasoconstriction, GI shutdown
etc; activation of beta receptors leads to vasodilation, increased cardiac output etc.
function.
Smooth Muscle - the quiet effector cells
cells.
muscle cells produce a slow, prolonged contraction that makes it an excellent cell for
its homeostatic role.
control access to specific areas of the body.
products.
are ideal to maintain key bodily functions.
modulate activity.
skeletal muscles.
muscle.
contractile activity via pacemaking: leak channels (K+) which periodically depolarise.
actual contractions (Ca2+ influx).
channels that allow communication between cells.
contraction.
entry is enough.
calcium levels.
●
Autonomic reflexes
● Several CNS nuclei communicate to ANS ganglia to influence autonomic functions in a combined behavioural, autonomic and endocrine response.
● It is the solitary nucleus of the dorsal medulla that receives most of the visceral sensory input from the vagus nerve.
● It then feeds information up to the autonomic integration centres.
● These areas include the parabrachial nuclei (arousal and thermoregulation), the periaqueductal grey (nociception), the hypothalamus (neuroendocrine and physiological functions).
WEEK 8 - Cardiovascular Physiology I
The cardiomyocyte
contractions of the atria and ventricles to allow the pumping of blood around the
body.
and this has major impacts on whole body physiology.
intercalated disks.
action potentials from cell-to-cell.
propagate like a wave throughout the chamber and depolarise all of the cells - this is
called a functional syncytium.
the ventricles by a thick band of fibrous tissue meaning the heart has two functional
syncytia - the atria and the ventricles.
ventricular depolarisation.
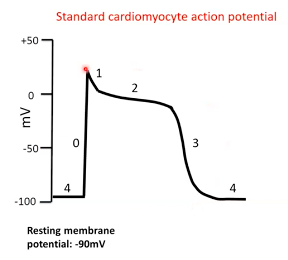
system to enable proper cardiac function. Resting membrane potential in cardiomyocytes
●
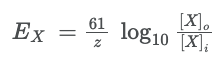
![]()
The cardiac pacemaker cells and spontaneous depolarisation
cardiac cells that form pacemakers.
intervals (~100/sec).
under normal healthy conditions.
act as the normal pacemaker.
pacemaker albeit with an overall lower rate of depolarisations (leading to lower HR).
the transmission of atrial action potentials/depolarisations to the ventricles via the Bundle of His.
Pacemaker cells
Phase
contribute
4 = pacemaker potential
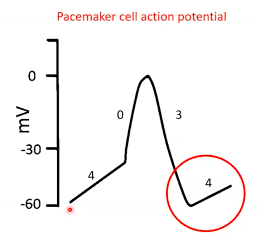
Phase 0 = depolarisation
● Voltage gated Ca2+ channels open at -40mV. ● Ca2+ influx.
● Membrane depolarises.
Phase 3 = repolarisation
● Ca2+ channels close. ● K+ channels open.
● K+ efflux.
● Membrane repolarises.
Summary:
Contractile cardiomyocyte depolarisation and cardiac contraction
and cannot spontaneously depolarise.
membrane voltage is held constant at approx. -90mV.
potential... which is generated at regular intervals by pacemaker cell depolarisation
in the SA node.
potential generated by pacemaker cells activates adjacent contractile cardiomyocytes and they continue to spread the action potential like a mexican wave through the atria in a cell-to-cell manner.
favours movement of K+ out of the cell.
over ~200ms. The plateau phase is maintained by a balance between
continues K+ efflux and Ca2+ influx.
greater efflux of K+ and membrane repolarization.
○ Phase 4 - resting phase is re-established, and ion concentrations are rapidly brought back to normal by ion pumps/exchangers.
How do the action potentials cause muscle contraction?
300ms.
depolarised (+tve membrane potential).
take place within a cardiomyocyte:
Video:
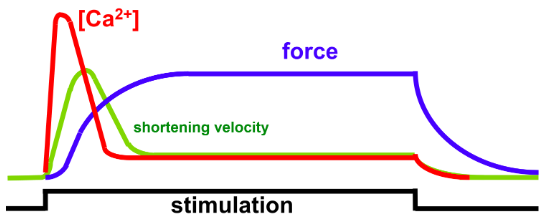
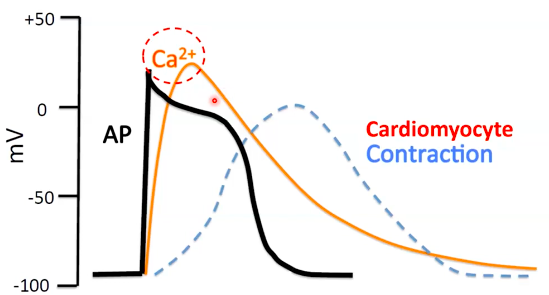
Excitation contraction coupling and the refractory period Cardiomyocyte depolarisation
Excitation-contraction coupling (ECC)
The more calcium that is present in the cell, the more powerful the contraction AND The longer the calcium hangs around, the longer the contraction.
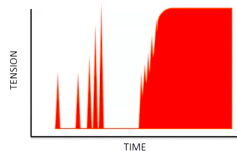
Refractory period
● Effective/absolute refractory period
● Relative refractory period
What happens if we try to stimulate more action potential in the refractory period? Tetanic Contractions
● Skeletal muscle
stimulation.
stops the valves from opening and the
heart from filling.
Summary: Refractoriness and non-tetanisation
before the heart can contract again.
Control of frequency and force of contraction
depolarisation.
a coordinated sequence to generate contraction in an efficient way for blood to be
pumped.
the:
muscle).
Video:
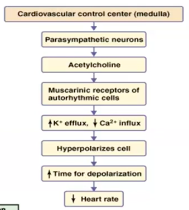
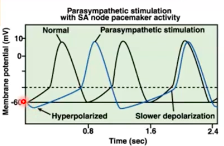
Parasympathetic and sympathetic modulation of HR Cardiac Automaticity - signal generation
● Cardiac action potentials are initiated in collections of specialised cardiac cells that form pacemakers.
○ Physiological pacemaker under normal conditions. ● AV NODE
○ Can take over as pacemaker if SA node is damaged.
Multiple autonomic nervous system inputs PNS (vagus)
signal is strong enough).
SNS
PNS - decreasing heart rate
WEEK 9 CV2: control of cardiac output and blood pressure
Cardiac output
pressure necessary to provide adequate blood flow to the organs.
to generate sufficient pressure to propel blood from the left ventricle, through the
aortic valve and into the aorta.
pulmonary valve and into the pulmonary artery to perfuse the lungs.
Factors influencing venous return
venous system forcing blood back towards the heart (venous valves prevent
backflow).
muscles, propelling blood back through the venous system to the heart.
○ This is because average resting HR
is ~60bpm and SV is ~80mL/beat
(60x80=4800)
to ~35L/min and this is driven by the work/oxygen requirements of working skeletal muscle.
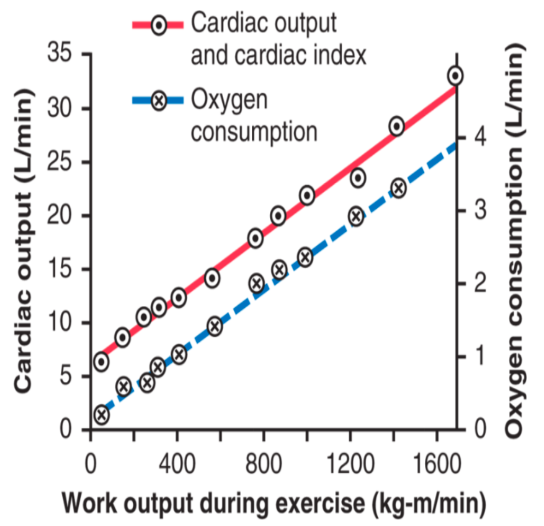
Regulation of HR (and CO)
increase from the resting state and therefore can contribute a greater relative
increase in CO when required.
depolarization of the pacemaker cells in the SA node.
systems have direct inputs into the heart to regulate HR.
The Bainbridge Reflex
increased HR.
distend the right atrium, but denervation of the vagus nerve to the heart eliminated
these effects.
arterial stretch receptors.
via the vagus nerve to the heart, leading to increased HR.
○ Increased venous pressure -> pressure in vena cava increases -> pressure in right atrium increases -> stretch receptors activated -> signal to the medullary control centre to decrease PNS activity -> increased HR.
Regulation of Stroke Volume (and CO)
every time the heart beats.
○ These are preload, afterload and contractility.
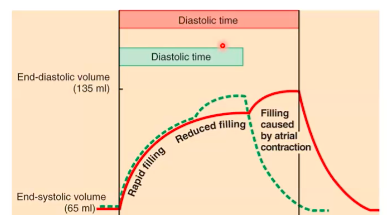
Preload
greater the force of the contraction.
Preload continued (video)
generated in the subsequent contraction.
(ie stronger contraction). Mechanism of length-tension relationship
● It has been proposed that placing the sarcomere under more tension increases the sensitivity of troponin C to Ca2+.
○ Ie it increases the excitation-contraction coupling efficiency. Preload
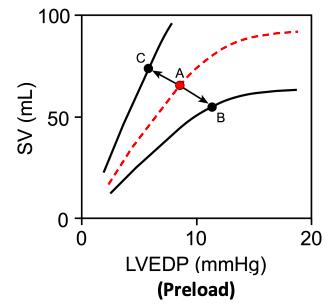
The frank-starling curve
● THis is a ventricular function curve, and clearly shows the frank-starling mechanism
at work.
●
Implications of the Frank-starling mechanism
flow to the tissues.
● When HR increases, time available for filling decreases.
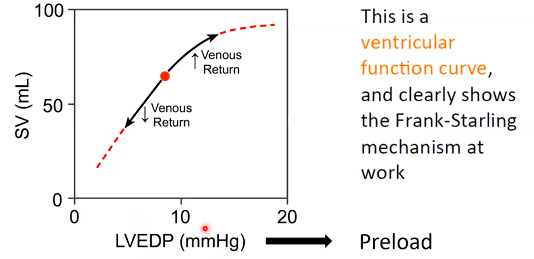
relatively greater importance.
Afterload
○ Eg when lifting a weight.
● The afterload of the left ventricle is closely related to aortic pressure.
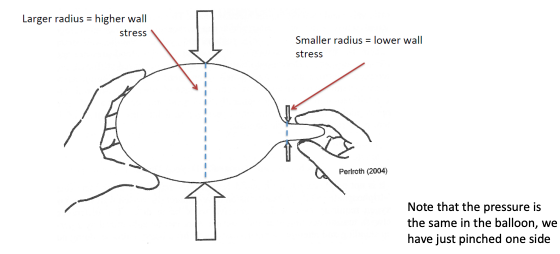
The ventricles have muscle arranged in roughly a sphere shape
related to the tension in the ventricular wall.
says thats:
Analogy - balloon filled with air
● An increase in radius increases wall stress
Afterload and Cardiac Function
during each ventricular contraction. The frank-starling curve: effect of afterload
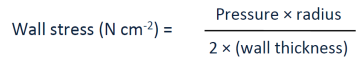
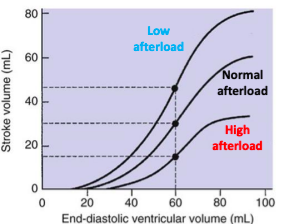
What can change afterload?
eject blood).
○ Eg. dilated cardiomyopathy ● Wall thickness
○ Eg. ventricular hypertrophy decreases wall stress and is a compensatory mechanism for when afterload increases.
Contractility - The performance of the ventricular myocytes at a given preload and afterload. Contractility of cardiac muscle
phase/contraction.
The Frank-starling curve: effect of contractility
function curve.
any given preload.
any given preload.
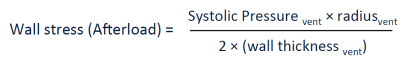
Cardiac output provides arteries with BP
pressure differences present between the large arteries (high pressure) and large
veins (low pressure).
time it beats.
at ~120mmHg during systole and dropping down to ~ 80mmHg during diastole in a
resting, healthy individual (this is where the 120/80 mmHg comes from).
arteries and arterioles that supply capillary networks within organs
○ These arteries/arterioles are typically called the resistance vessels and
collectively they determine the total peripheral resistance (TPR).
blood pressure is much lower compared with the arterial system.
as it enters the right atrium.
Mean arterial blood pressure (MAP)
cardiovascular function and for assessment of organ perfusion.
sometime also called systemic vascular resistance [SVR]), and central venous pressure (CVP) according to the following relationship, which is based upon the relationship between flow, pressure and resistance (Ohm’s law):
![]()
![]()
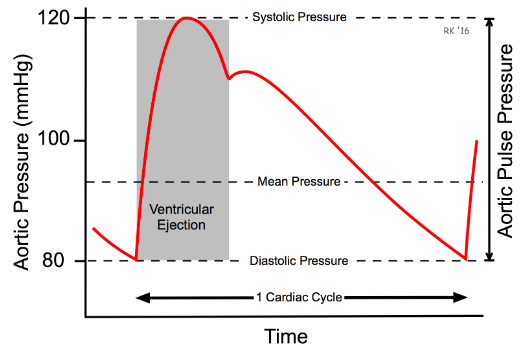
average (100mmHg when time is ignored) of the systolic and diastolic pressures.
Regulation of mean arterial blood pressure (MAP)
blood vessels during a single cardiac cycle.
perfusion (blood flow) of the organs of the body at any given time (normal range is
between 70 and 100mmHg).
pressure alone (because it also relies on TPR and thus tells us about peripheral
blood flow).
arteries, kidneys, and brain to keep them functioning.
consequences and these will manifest acutely within vital organs. Problems when MAP is too high:
Problems when MAP is too low:
● Low MAP can be life-threatening as well.
keep MAP regular at all costs.
Physiological feedback loops - 3 major parts 1. Sensor
○ Detects a change in a physical or chemical property that (eg. vascular stretch in a baroreceptor).
2. Control centre
3. Effector
muscle relaxation).
Physiological feedback and short-term regulation of blood pressure
○ The vasomotor centre in the brain stem (medulla).
3. Multiple effectors
Short-term control of MAP Baroreceptors - direct sensors

![]()
by inhibiting vasoconstriction.
systems.
Baroreceptor reflex
neurons and inhibits the PNS neurons in the medulla.
quickly correct the fall in BP.
Chemoreceptors - indirect sensors
causes peripheral vasoconstriction, increased HR and increased SV.
Chemoreceptor reflex
stimulation of the heart, which increases the HR.
stimulation of the heart, which increases the HR and SV.
stimulation of blood vessels, which increases vasoconstriction.
Summary:
to ensure organ perfusion.
activate/deactivate the cardiovascular centre as needed.
to changes in O2 and CO2.
Long-term control of MAP
of blood volume.
cardiovascular system and we see blood pressure increasing.
system and therefore blood pressure decreases.
renal system.
tightly regulate blood volume by controlling excretion rates of Na+ and water in particular.
●
volume/pressure homeostasis.
WEEK 10 CV3: Control of blood flow and special properties of the macro- and micro- circulation.
Haemodynamics and control of blood flow
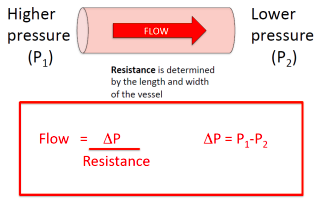
● How constriction and dilation impacts the flow of blood through arteries can be explained by Ohm’s law and Poiseuille’s law that describe the relationship between radius, resistance and flow.
Basic haemodynamics - Ohm’s and Poiseuille’s laws
○ During ventricular systole, blood is ejected from the ventricles at very high pressure.
○ The bulk flow of blood in the cardiovascular system is from high to low pressure.
○ Resistance is:
The flow through a tube is (pressure difference x radius to the power of 4) divided by (viscosity x tube length).
In the body:
heart beat-heart beat.
Vessel radius is a very powerful determinant of both resistance and flow

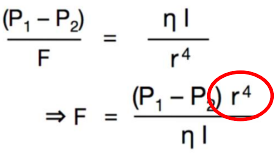

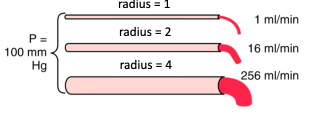
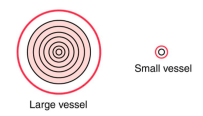
○ The further the blood is located from the vessel wall the faster the flow.
Using the whole circulation as an example
● In this case, the flow is the entire output of the heart =
cardiac output.
peripheral circulation.
Control of blood flow in the body
use, but dispensable organs that get blood flow when there is enough to go around.
○ The radius of the blood vessels it is flowing through.
they can handle an order of magnitude more blood flow because thier resistance to
flow has decreased dramatically.
where blood flows in the body - these arterioles are often referred to as ‘resistance vessels.’
In the body, six primary factors can modulate the radius of resistance arterioles to regulate how much blood flow they receive:
1. Special receptors
○ Example: skeletal muscle arterioles dilate in response to moderate levels of
adrenaline or insulin. 2. Metabolic control
○ Example: diffusion of metabolic waste products (adenosine, ADP, CO2, lactate, K+ etc) from active cells causes arteriolar vasodilation.
3. Oxygen deficiency (low pO2)
○ Example: oxygen removal from blood by active cells reduces local blood
oxygen levels -> stimulates arteriolar vasodilation. 4. Sympathetic nervous system

○ Example: SNS has direct inputs into most blood vessels and SNS outflow can alter constriction/dilation in arteries and veins to regulate blood flow to individual organs.
5. Nitric oxide
○ Example: shear stress (force exerted by blood on endothelial cells = blood
viscosity x blood velocity/internal diameter) in arteries and arterioles causes their lining endothelial cells to release Endothelium Derived Relaxing Factors (EDRF) like nitric oxide -> nitric oxide is a potent vasodilator.
6. Longer-term control mechanisms
○ Example: capillary density changes (eg. in exercise trained skeletal muscles)
meaning the arterioles need to supply more flow to that tissue -> arteriolar
vasodilation.
Autoregulation and rationing of cardiac output
Regulation of blood flow by metabolism
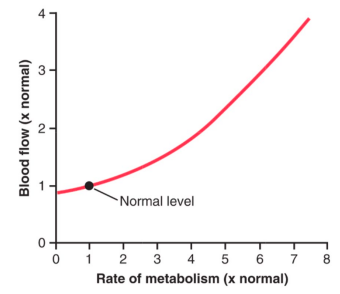
‘Autoregulation’ of blood flow
● Why is flow not a passive reflection of pressure changes in certain organs?
○ Because some organs require constant blood flow to maintain their functions despite changes is systemic blood pressure
● Two mechanisms are responsible for this in those organs:
prominent in arterioles.
release of vasoactive metabolites and low O2 tension.
Even if systemic arterial pressure changes, blood flow in some organs remains stable
● This is called autoregulation and it is achieved by automatic changes in vascular tone in response to blood pressure.
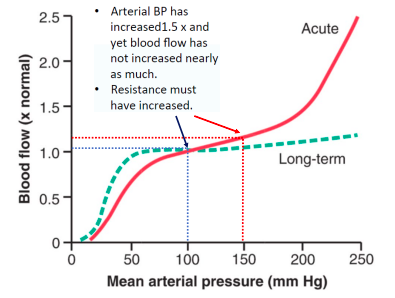
Total systemic blood flow (CO) is the sum of blood flow to all organs
demands for blood/nutrients.
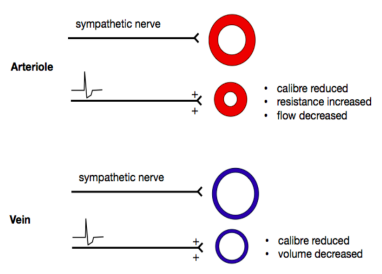
Sympathetic nervous system innervates the vasculature to regulate in this instance
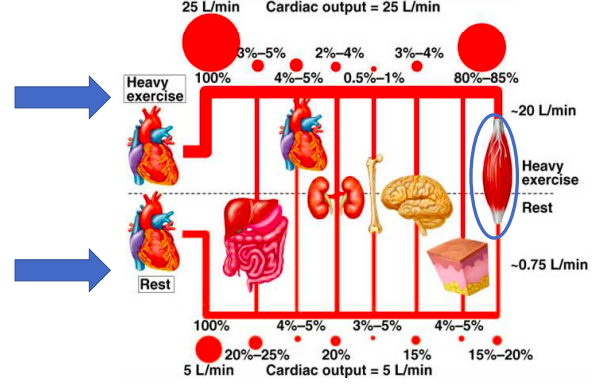
What if cardiac output is limited?
maintain perfusion.
and reduced perfusion.
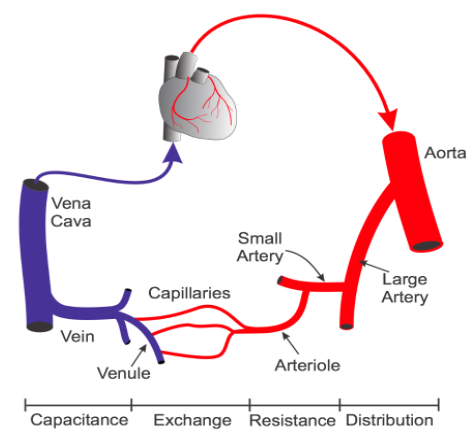
Special properties arteries, veins and capillaries
branch into successively smaller arterial vessels until
they become capillaries.
continue to join with other veins until they enter either the superior or inferior vena cava that bring the blood back into the right atrium of the heart.
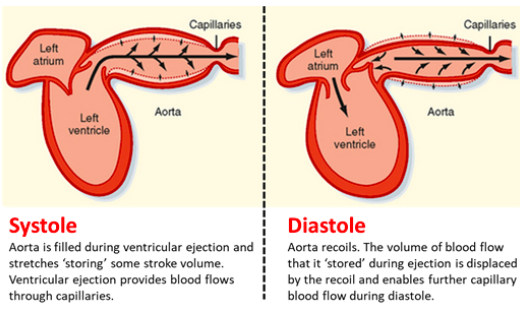
Functions of the aorta and large arteries
blood to the systemic arterial system, dampens the pulsatile pressure that results
from the intermittent outflow from the left ventricle.
flow is a function of the aortic compliance - the ability of the aorta to stretch when it fills with blood during systole and then recoil back to its resting state by actively contracting to continue pushing blood forward.
involved in the regulation of arterial blood pressure as well as blood flow within organs.
basement membrane, are termed capillaries.
Functions of the capillary network
● Capillaries represent the smallest vessels (~5-10um in diameter) within the
microcirculation and arguably perform the most important role of the cardiovascular systems:
○ They facilitate nutrient exchange between blood and tissues they supply.
constriction/dilation controls capillary flow.
each downstream capillary equally.
themselves have cells wrapped around them that can constrict/dilate individual capillaries - these cells are called pericytes and they are similar to smooth muscle cells.
through. The kidneys are the primary site in the body where these capillaries are found.
Starlings forces and capillary filtration
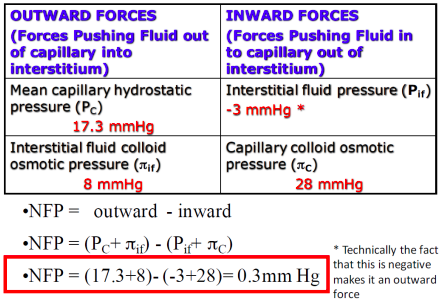
Movement of solutes or filtration across capillaries depends on 2 things:
1. The balance of hydrostatic and osmotic pressure across the capillary
○ This is called the Net Filtration Pressure (NFP) = total pressure promoting
fluid movement to/from capillary
2. The surface area and permeability coefficient (Kf) of the capillary
Starling’s forces: pushing fluid in and out of capillaries
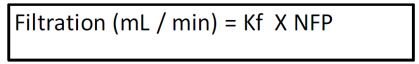
So does that mean very little fluid flow to or from capillaries?
Yes and No
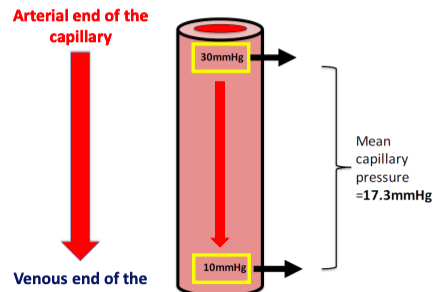
Yes: The average capillary hydrostatic pressure is ~17.3mmHg. Therefore, there is not much net movement of fluid to and from the interstitial space.
No: The arterial end of the capillary is at higher pressure than 17.3
The venous end of the capillary is at lower pressure than 17.3
Blood flows from high-low pressure
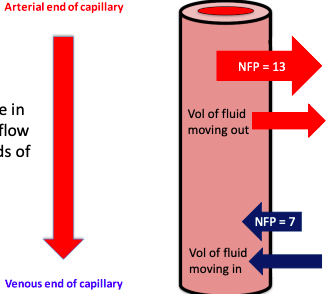
Net Filtration Pressure - Arterial end of capillary
Net Filtration Pressure - Venous end of capillary
The amount of filtration at each end is determined by the NFP and the permeability (Kf)
Notice the difference in pressure/amount of flow between the two ends of the capillary
~90% of the fluid leaving the capillary at the arterial end comes back into the venous end.
Venous end of capillaries have a higher Kf Filtration (mL/min) = Kf x NFP


If 90% of the fluid comes back into the capillary, where does the remaining 10% go?
● To quantify: 10% = ~2ml/min
● Over the course of 24 hours = ~3000mL or 3L
● If this fluid was not removed, you would lose a lot of blood volume very quickly.
● The excess fluid left in the interstitial space (~3000mL/day) is drained away by the
lymphatic system.
Functions of the vena cava and veins
exchange vessels, particularly for large macromolecules as well as fluid.
intervals - this means blood flow in the venous system is unidirectional.
again appears.
constricting, and serve an important function regulating capillary pressure.
- ie. the site where most of the blood volume is found and where regional blood volume is regulated.
○ For example, constriction of the veins decreases venous volume and increases venous pressure, which alters cardiac output by increasing venous return (preload).
● The final venous vessels are the inferior and superiour vena cava, which carry the blood back to the right atrium of the heart.
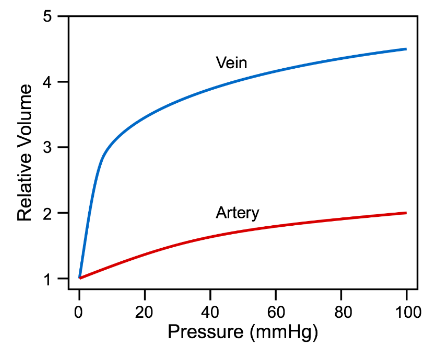
Arterial and venous compliance and capacitance
depicted:
decreases as pressure increases.
vessels become “stiffer” at higher pressures and volumes).
the compliance of a vein is about 10 to 20 times greater than an artery.
Exercise, venous return and the skeletal muscle venous pump
is the muscle venous pump.
flow away from the limb and back toward the heart.
muscles surrounding them contract, and they become decompressed as the muscles
relax.
stages of compression and decompression (ie. there is pumping).
venous segment.
towards the heart.
flow that enhances venous return.
relax to keep the body in balance.
pressure and venous return, and lowers venous and capillary pressures in the feet and lower limbs.
The lymphatic system and vessels
that reside near the normal capillary bed in all tissues within the body.
fluid/protein/bacteria etc. into the lymphatic vessel.
from tissues to the larger lymph vessels and ultimately back into the systemic circulation.
- specialised structures that also contain a large number of lymphocytes and
macrophages that monitor the lymph for pathogens.
not lost in the interstitial space within tissues.
interstitial space and returns this to the systemic circulation at the subclavian vein.
day.
Regulation of lymphatic flow rate
What controls movement of fluid into lymph vessels?
What happens if we exceed this max lymph flow rate?
capacity:
●
What factors can cause oedema?
overload).
![]()
![]()
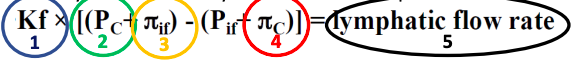
WEEK 11
Renal System 1 - Basic Renal Functions and Transport Processes in the Nephron Intro
from the body by forming and excreting urine.
deadly consequences for the rest of the body.
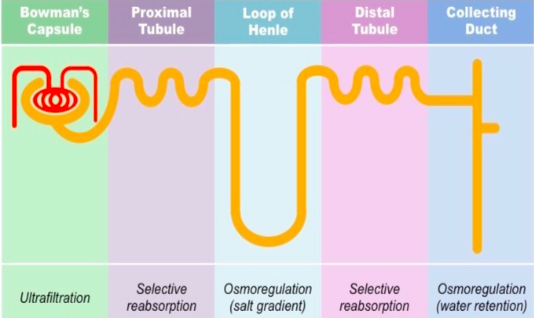
Excretory functions of the kidneys
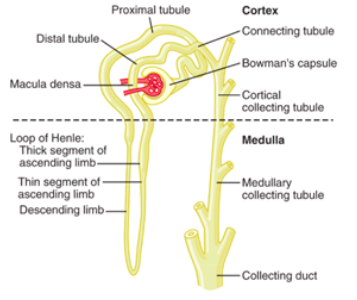
discrete sections:
the distal end to form a collecting tubule that eventually turns into a collecting duct and delivers urine to the calyxes and eventually the ureters.
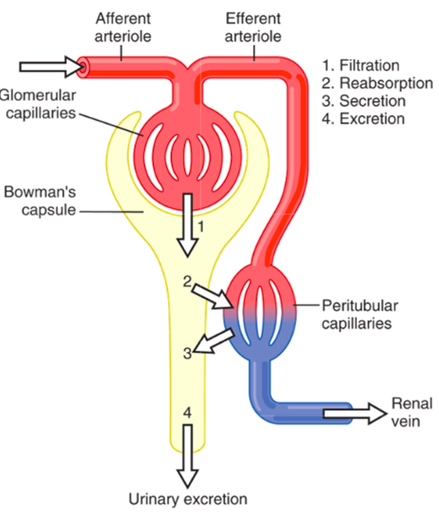
Events that determine renal excretion
into the Bowman’s capsule from the glomerulus.
the nephron tubules back into the body.
peritubular capillaries that surround the nephron into the tubular network.
● Therefore the amount of any substance that is excreted from the body is determined using the following equation:
○ Excretion = Amount filtrated - Amount reabsorbed + amount secreted
Filtration
● The process where blood is filtered from the glomerular capillaries into the
Bowman’s Capsule through a serious of pores that make up the filtration barrier.
●
Glomerular filtration barrier Anatomy of the barrier:
extensions (pedicles).
layers.
Glomerular filtration barrier: size
size).
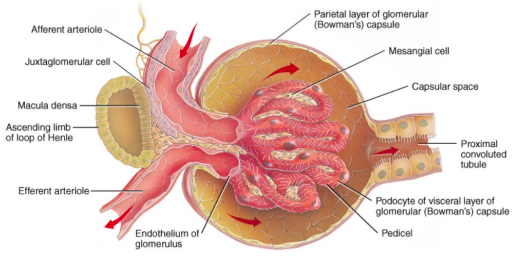
Glomerular filtration barrier: charge
prevents their passage into the filtrate.
○ In disease states (eg. glomerular nephritis or diabetic nephropathy) this
barrier lsoses its selectivity allowing protein into urine -> proteinuria. Glomerular filtration
filtrate as plasma.
Glomerular filtration rate (GFR)
plasma entering the kidneys per minute - ie. renal plasma flow is ~600mL/min.
the glomerular filtration rate (GFR).
nephrons.
volume is filtered 60 times per day.
○ This provides the kidneys an enormous capacity to regulate fluid and
electrolyte levels in the body to ensure homeostasis.
regulated to ensure there is not too much or too little getting to the nephron where
filtration happens.
afferent and efferent arterioles.
regulate how much filtrate is formed by the kidneys at any given time.
Control of glomerular filtration rate (GFR)
● GFR is dependent on 2 factors:
1. The surface area and permeability of the filtration barrier
○ Expressed as the constant Kf
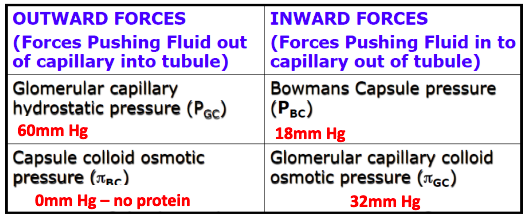
2. The Net Filtration Pressure (NFP)
○ THe balance of hydrostatic and osmotic pressures at work across the filtration
barrier. GFR and NFP
NFP = outward - inward
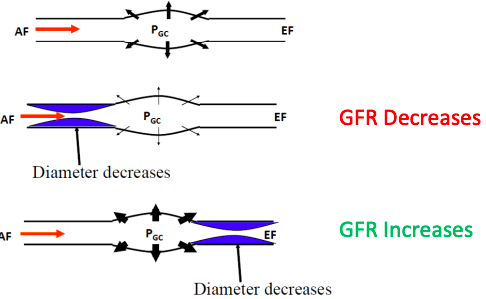
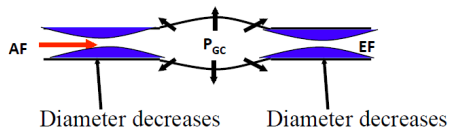
Modifying GFR through arterial constriction
What happens if both vessels constrict?
Summary of arteriolar control of GFR
● The majority of physiological control of GFR occurs through alteration of the
glomerular capillary hydrostatic pressure (PGC)
Renal Autoregulation
ensure constant renal blood flow and GFR despite potential changes in systemic
blood pressure - this is autoregulation.
blood flow despite changes in systemic blood pressure.
influences and therefore is intrinsic to the organ, although these influences can
modify the response.
pressure.
○ Afferent and efferent arteriolar resistance are altered to keep GFR steady.
● Why is this useful?
○ Allows the kidneys to maintain their excretory capacity and blood volume
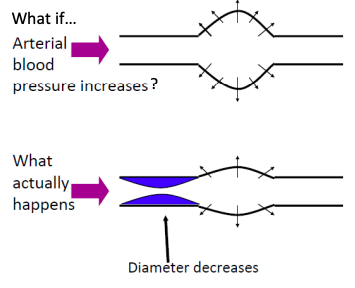
homeostasis even when systemic blood pressure changes. Renal Autoregulation: Myogenic mechanism
Vessel diameter unchanged, hydrostatic pressure increases, GFR increases.
Afferent arteriole constricts due to smooth muscle stretch-feedback, hydrostatic pressure and GFR largely unchanged.
Renal autoregulation: tubulo-glomerular mechanism
very close to the afferent and efferent arterioles.
through the DCL.
fluid has decreased.
receive a signal which then causes them to produce more renin.
arteriolar constriction and increases back pressure inside capillaries.
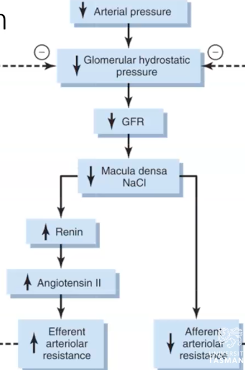
● Macula densa talks directly to afferent arterial to decrease resistance and increase dilation, increasing GHS.
Renal Autoregulation
● Myogenic mechanism
○ Very fast to act and accounts for ~50% of renal autoregulation. ● Tubulo-glomerular feedback mechanism
○ Slower to act and accounts for ~35-40% of renal autoregulation.
● Renal blood vessels can respond without SNS/humoral input to ensure a steady
GFR despite changes in systemic pressure.
Reabsorption and secretion
through re-absorptive processes.
on secretory processes.
network within the nephron (excluding bowman's capsule).
most move between the tubule/capillaries by using either transcellular (through cells via transporters or ion channels) or paracellular (between cells using tight junctions) or both.
Principles of tubular handling of filtered substances
nephron, not every substance that is filtered can be reabsorbed and/or secreted.
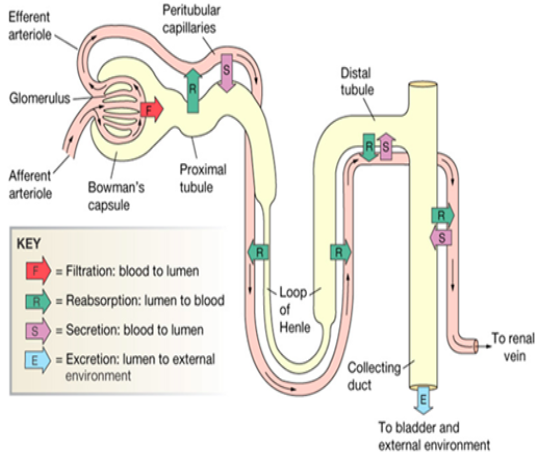
beyond what is filtered from the blood alone.
this substance from the peritubular capillaries and into the tubules.
filtered nutrients.
blood (~145mmol/L).
concentration, very large amounts of Na+ are filtered all the time.
control of how much Na+ is excreted from the body (depends on homeostatic
needs),
reabsorbed from the filtrate (allowing more to leave the body).
has multiple physiological inputs that can regulate Na+ balance.
from the filtrate. We can also switch on the active tubular secretion if further
K+ excretion is required.
the body and the body has multiple physiological inputs that can regulate K+ balance.
● Water (H2O)
of urine per day.
percent (1-2L) can be reabsorbed or not depending on water homeostasis
requirements.
filtered H2O.
has multiple physiological inputs that can regulate water balance.
The limits of tubular transport
far exceeds our total blood volume (by a factor of 30) and thus most of what is
filtered must be reabsorbed back into the body to maintain homeostasis.
there are two limiting factors to be aware of:
number of transporters available that can move that substance from the tubular
lumen back into the body. Therefore reabsorption can be ‘Transport Limited’
maximum concentration gradient that can be achieved and maintained between the tubular lumen and the interstitial space. Therefore reabsorption can be ‘Gradient Limited’
Transport limits and osmotic diuresis
Reabsorption of some substances is rate-limited by transporter capacity
Glucose transport in the nephron
○ Note - blood flow prevents glucose build up in interstitial space, so conc. Gradient is maintained to favour glucose movement into the bloodstream.
Glucose is a good example of Tm limited transport
● At normal glucose concentrations (~5mM) all of the filtered glucose is reabsorbed.
○ Although some SGLT2 inhibitors (reduce Tm) are used to reduce glucose levels in people with T2D.
Gradient limited transport - eg. Na+ in the proximal tubule
● ●
●
Some substances (typically ions) do not have a Tm
Upper limits of transport is defined by the concentration gradient that can be sustained across the tubule.
Eg. as Na+ is reabsorbed in the proximal tubule,
○ Lumen [Na+] decreases while interstitial [Na+] increases
○ The tight junctions in the proximal tubules are slightly leaky to Na+
○ The gradient limit for Na+ is only 2 mmol/L, once the interstitial [Na+] exceeds
tubular lumen [Na+] by more than 2mmol/L, Na+ starts to leak back into the
tubules through paracellular pathways (tight junctions become leaky).
○ Under normal circumstances in the proximal tubule, water follows Na+ so that
the lumen [Na+] doesn’t change and is not anywhere near the gradient limit.
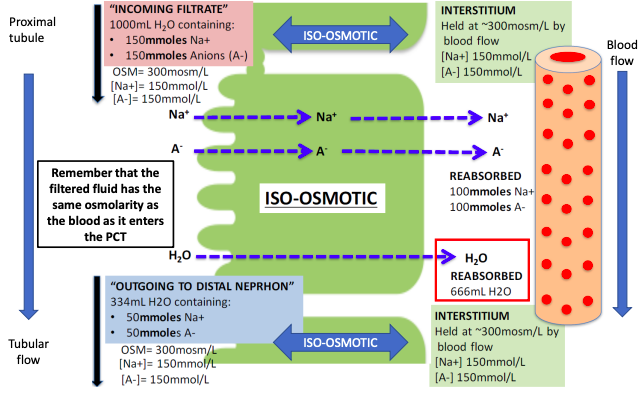
This is ●
●
the normal situation... what happens if we stuff things up with an ‘osmotic diuretic’ What if we intravenously infuse a patient with a solute that stays in the extracellular fluid, is freely filtered but poorly reabsorbed in the kidney.
Eg. Mannitol
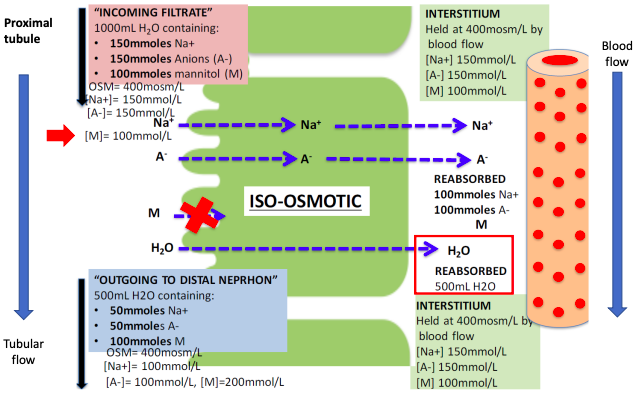
Osmotic diuresis - mannitol/water
○ 1000ml fluid into tubule from glomerulus
○ 334ml fluid out of distal nephron as urine (65% reabsorbed)
○ 1000ml fluid filtered into tubule from glomerulus.
○ 500ml fluid out of distal nephron as urine (50% reabsorbed)
Osmotic diuresis - mannitol and Na+
○ [Na] in interstitium and peritubular capillaries is 150 mmol/L
○ [Na] in tubular fluid going out of distal nephron as urine is 150 mmol/L
○ [Na] in interstitium and peritubular capillaries is 150 mmol/L
○ [Na] in tubular fluid going out of distal nephron as urine is 100 mmol/L
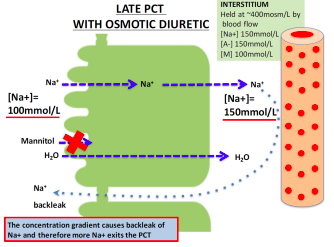
Late PCT with Osmotic Diuretic
Osmotic diuresis
increased volume of urine, quickly causing dehydration.
reabsorbed out of the lumen.
water and Na+ out with it to increase urine output.
○ This is why uncontrolled diabetics urinate a lot and are always thirsty.
Take home message: there are limits on tubular transport
Or
these transport limits.
WEEK 12 - Renal System 2 - Water, Sodium and Potassium homeostasis
Variation in water and solute handling across the nephron
processes available to each segment.
● Most of the physiological control of water, Na+ and K+ occurs in the distal nephron and collecting duct.
●
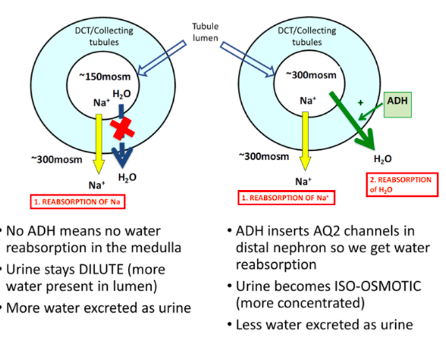
Antidiuretic Hormone (ADH)
blood volume.
on principal cells to increase water reabsorption in the distal nephron (inserts
aquaporin 2 channels into cells).
becomes permeable.
Counter-current mechanism and renal interstitial solute concentration
ducts, loops of henle, vasa recta (capillaries) and the interstitial space.
of urine concentration and other specialised kidney functions.
Video 1
The role of the counter-current mechanism and ADH in urine formation
What mechanism produces the concentration gradient in the renal medulla?
● There are 2 main factors:
concentration of interstitium to increase.
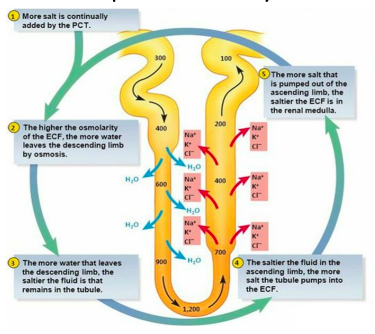
1. Loop of Henle permeability to water and solute
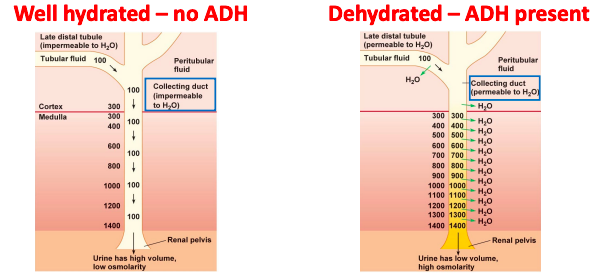
2. Medullary blood flow
○ 1. Medullary blood flow is much slower than in the cortex
■ While useful for concentrating urine, this does increase the susceptibility of the medulla to ischemic injury.
○ 2. The arrangement of the medullary blood supply into the descending and ascending vasa recta limits the washout of the medulla hyperosmolarity.
Countercurrent Mechanism Countercurrent exchange
which is taken back by ascending limb of vasa recta.
medulla.
of vasa recta maintains the osmolality gradient established by LOH.
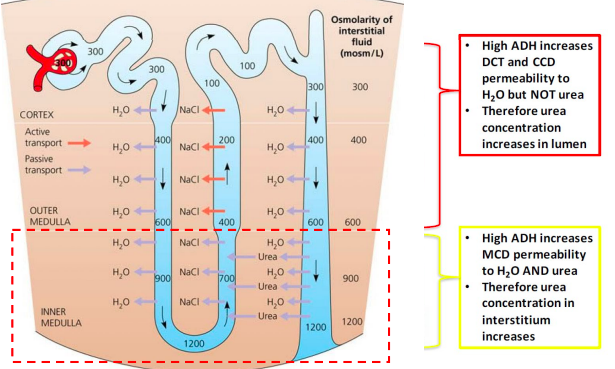
Urea and the osmolarity in the medulla
from NaCl.
Recycling of urea in the distal nephron
How much urine volume do you need to produce each day for excretory homeostasis?
excreted by the kidney daily.
per day or as much as 15L (min ADH effect) per day.
concentration of the urine that can be achieved in the renal medulla according to the following equation:
○
loss from the skin, respiratory tract, and GIT, when water is not available to drink.
concentration of 1200 mOsm/L explains why severe dehydration occurs if one
attempts to drink seawater. Can you drink seawater to survive?
contributes about 600mOsm/L.
body of 1200 milliosmoles of NaCl ingested in addition to all the other soltes.
Water homeostasis
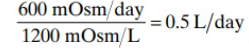
Video 2
Physiological Regulation of body water Basic negative feedback loops in physiology
● These mechanisms have two components:
specific set-point (eg. vascular stretch).
original disturbance (tries to restore homeostasis).
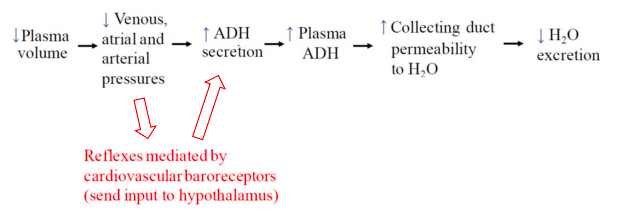
Body water homeostasis
water sensors are:
Osmoreceptors in the hypothalamus Baroreceptors (mechano-receptor)
water effectors are:
Posterior pituitary release of ADH (signal from hypothalamus)
ADH increases water permeability in the distal nephron (and increased inner medullar urea absorption).
Activation of the thirst centre increases thirst (and thus water intake)
○
Blood volume, baroreceptors and ADH release
Interplay of the osmoreceptor, thirst and ADH
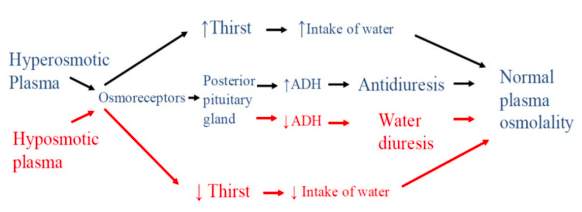
Which is the more powerful mediator of ADH release?
Secretion of ADH
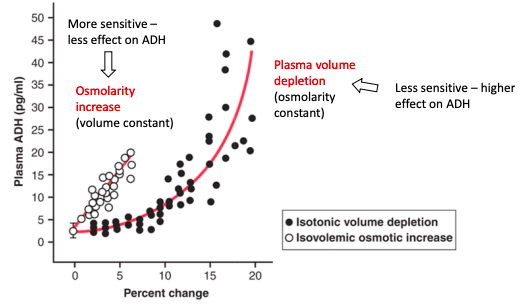
● ADH secretion depends on both plasma osmolarity and volume: 1. Plasma osmolarity
○ Very sensitive: responds to 1-2% change 2. Plasma volume
together and become synergistic (eg. dehydration, blood loss with fluid shift from
extracellular space).
hypotonic fluid replacement; eg. diarrhoea with normal water intake) and the result
will be determined largely by how low blood volume is.
Water homeostasis and thirst
dilate or concentrated urine via ADH).
homeostasis...
Sodium Homeostasis
motivated behavioural response that drives us to seek and ingest salt-containing
foods and fluids.
Why is salt so important?
a concentration of ~145 mmol/L.
chloride (Cl-) make up most of the plasma osmolarity (~300mOsm/L) under normal
physiological conditions.
plasma osmolarity.
(thirst) and output of water (kidneys, ADH status) and mediated by the osmoreceptors.
ECF volume.
therefore increased BP.
ECF volume (and thus BP)... because plasma osmolarity is monitored closely by the
osmoreceptors, water balance is adjusted to maintain a constant 300mOsm/L.
therefore low blood pressure because osmolarity is held constant.
volume and therefore blood pressure.
angiotensin aldosterone system and the atrial natriuretic peptide system.
Video 3
Control of Na+ in the body by the RAAS and ANP Renal regulation of Na+ excretion
● As Na+ is freely filtered at the glomerulus and actively reabsorbed, but not secreted then:
Negative feedback looks for Na+:
1. A sensor ->
○ 1. Extra-renal pressure sensors
■ Baroreceptors (activate CNS)
○ 2. Intrarenal pressure sensors
■ Juxtaglomerular cells (release renin)
○ 3. Indirect pressure sensor (NaCl delivery)
■ Macula desna (renal autoregulation/GFR control)
2. An effector ->
■ AND/OR
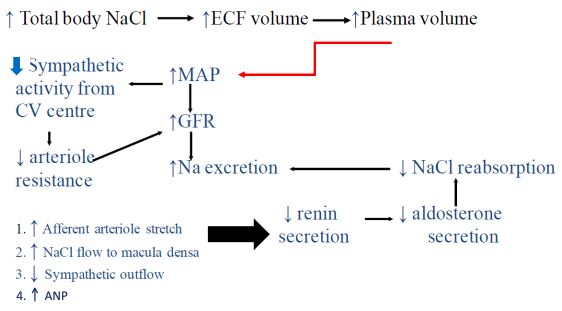
Regulation of Na+ by increased GFR
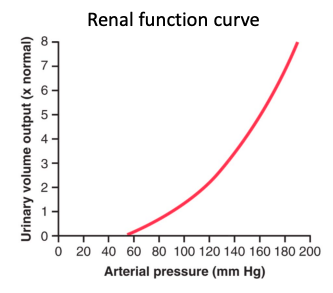
● Increasing arterial pressure causes a corresponding rise
in filtrate formation and therefore renal water output. ○ This is called pressure diuresis
● Increasing arterial pressure also causes a corresponding increase in Na+ filtration and therefore output.
○ This is called pressure natriuresis
● Therefore if pressure goes up, Na+ excretion goes up.
Regulation of Na+ by reduced GFR
Regulation of Na+ by reabsorption
● There are a number of stimuli that increase tubular reabsorption of Na+ (and
therefore Na+ in the body).
increase reabsorptive processes).
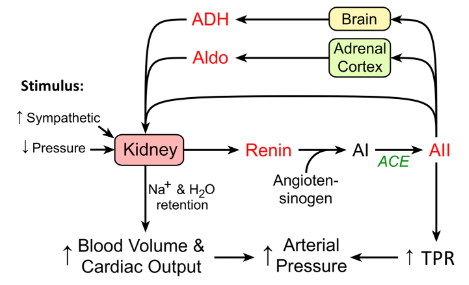
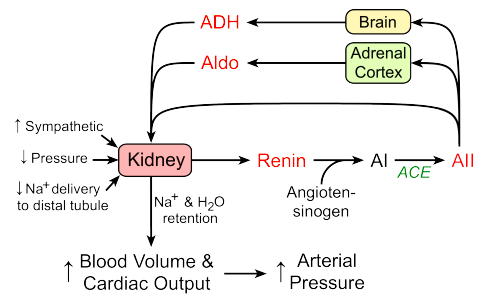
Renin angiotensin system in Na+ (and BP) homeostasis
Effect of angiotensin II on Na+ and water handling
causing Na+ reabsorption utilising H+ cycle
space at the expense of K+.
reabsorption in the proximal nephron..
Effect of aldosterone on Na+ and water handling
ATPase
● Remember that Na+ and water can be handled independently in the distal nephron
so while aldosterone will increase Na+ reabsorption, water will follow only if ADH is
also present.
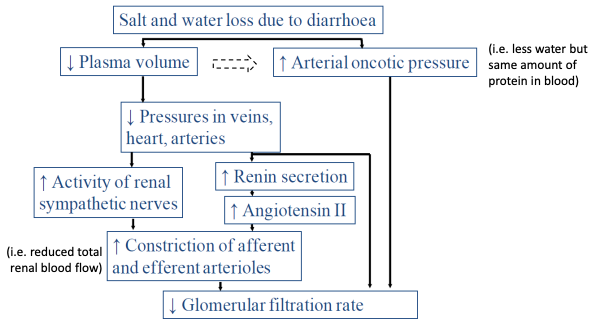
What is we need to increase Na+ excretion?
○ Results in less water and Na+ reabsorption. What happens when we increase total body sodium?
(eg high salt diet)
Summary
volume
determines blood pressure.
Potassium Homeostasis
(~140mmol/L) within cells.
depolarisation/repolarisation in excitable cells like neurons and cardiomyocytes.
3.5-5mmol/L.
excreted from the body via the renal system.
How do we regulate potassium homeostasis?
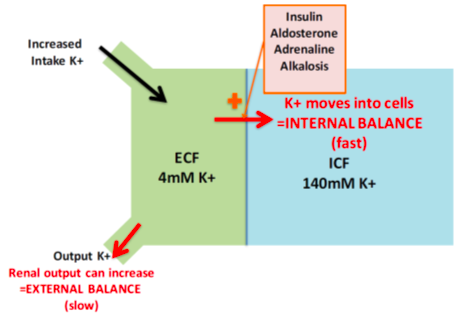
then adding 100 mmol of K+ would increase the ECF K+ concentration by more than 3.5mmol/L.
○ We would almost double our plasma K+ concentration from ~4-7.5mmol/L if nothing else happened.
○ this is where the renal system comes in and helps regulate the output of K+ to match the input from the diet.
●
Video 4
Physiological control of body K+ occurs in the distal nephron
How ●
How ●
● How ●
● How ●
●
is K+ excretion regulated
As with other solutes, K+ is freely filtered, actively reabsorbed and variably secreted:
Reabsorption of K+ in the PCT is constant whether it's hypokalemia or hyperkalemia.
○ Ie. 65% of filtered load is reabsorbed
Reabsorption of K+ is passive through both paracellular and intracellular routes. K+ os reabsorbed in the ascending loop of Henle
Thick ascending limb
movement back into interstitium
Loop diuretics act here to directly cause hypokalemia
is K+ reabsorbed in the distal nephron?
Intercalated cells can reabsorb some K+ in exchange for H+
When we need to conserve K+, the processes in the distal nephron shuts down K+ secretion by principal cells, leading net reabsorption by intercalated cells
Three main factors influence K+ secretion by principal cells in the distal nephron: 1. Aldosterone status
Effect of aldosterone
Effect of plasma K+ concentration
secretion.
Effect of tubular flow rate on K+
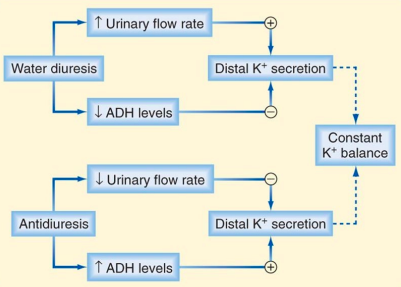
Does water status affect K+ homeostasis?
● Less ADH: decreased collecting duct permeability to ions
● More ADH: increased collecting duct permeability to ions.
Effect of high Na+ diet on K+ balance
in principal cells.
increasing K+ secretion.
Summary of potassium homeostasis
reabsorption and especially secretion.
○ Aldosterone
Summary of Renal Physiology 2
pressure because water will follow Na+ in the body.
systems to normalise Na+ and these work largely by activating thirst or changing
renal handling of Na+ via RAAS or ANP.
are able to depolarise and repolarise.
consequences and so K+ concentrations in the ECF are closely monitored and maintained at low concentrations by modulating the renal excretion rate of K+.
Week 13: Acid Base Physiology
Reintroduction - Acids and Bases
pH goes up or down quite quickly.
concentration of H+ in the ECF to ensure constant pH.
Video 1: reintroduction to Acids and Bases
● Acid: substance that is capable of releasing a proton.
○ hydrogen/proton DONOR
○ HCL->H++Cl-
● Base: substance that can accept/take a proton
Strong vs Weak Acids and Bases
○ HClH++Cl-
○ H2CO3 H+ + HCO3 - Equilibrium Constant (Ka)
○ Counter-intuitive with pH change
●
Control of pH in the body
○ Within cells pH is a little more acidic (~7.2) due to production of H+.
● Control of [H+] or pH in the body is a result of the balance between:
■ AND
Acid production in the body
○
Processes that decrease plasma [H+]
● Three levels of defence: 1. Buffer Systems
○ Works incredibly quick (fraction of a second) 2. Respiratory Control
○ Eg. removal of volatile acids by breathing out
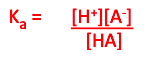
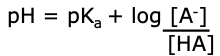
![]()
○ Works in minutes 3. Renal Compensation
Buffer Systems in Acid-Base Homeostasis
forming a new molecule.
back into solution if the amount of H+ starts to decrease.
of H+ and the buffer in the solution.
systems (respiratory or kidney) can kick in and remove the excess H+. Video 2: A Theoretical Experiment - Buffering H+
The bicarbonate buffer system is the most important buffer in the body
bicarbonate in the plasma and interstitial fluid) to momentarily form carbonic acid that then quickly breaks down into water and CO2 according to:
○
physiology:
○
partial pressure of CO2 in the expressed air.
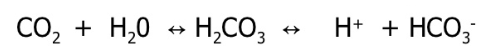
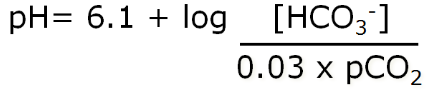
● We have physiological control over the pCO2 (lungs) and the HCO3- (kidneys).
The Respiratory System in Acid-Base Homeostasis
regulating the blood levels of carbonic acid (H2CO3).
H2CO3 in the blood are in equilibrium.
additional H2CO3, which dissociates further into H+ and HCO3- thus lowering blood
pH.
how much CO2 is removed from the system
●
activates the respiratory centre.
kicks in and the equation favours the right hand side more.
because of the need for O2 supply.
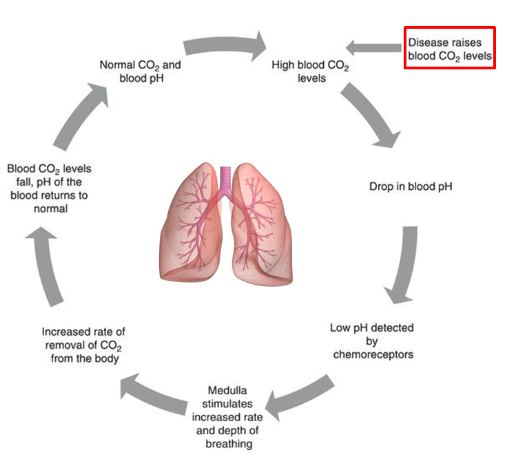
H+ being released and thus form more CO2 and H2O.
thus preventing a pH change.
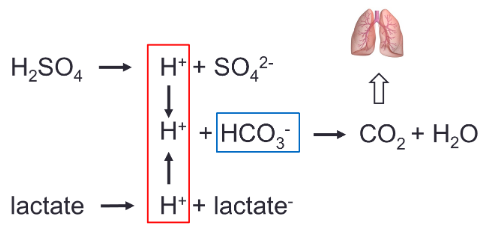
●
started.
and we would not be able to regulate pH.
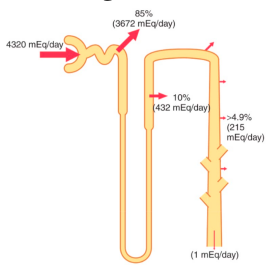
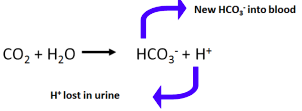
The Renal System in Acid-Base Homeostasis
they act quickly.
concentration of HCO3- and H+ in the body.
body.
doing so, they regulate the ratio of HCO3- to CO2 and thus pH:
●
course of a day, ~4320 mmoles of HCO3- is filtered.
take long to deplete our body HCO3- levels.
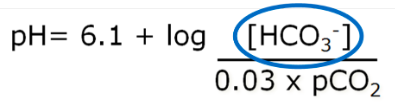
● The nephron can increase or decrease reabsorption of HCO3- to suit the body’s needs by changing what is happening in the PCT, DCT and collecting ducts.
VIDEO 3: Regulation of HCO3- and H+ in the Kidneys
○
H+ formed in the process
○ ● How
○ The answer is to bind the H+ (buffer it). ● Urinary Buffers and H+ excretion
much H+ can you excrete in urine?
used up.
much H+ can you excrete in urine?
● How
production.
○ Therefore, the 70 moles of H+ made in the process must be
secreted/excreted (because we make HCO3- by secreting H+).
○ ○
Breakdown of amino acids (protein metabolism) produces NH4+ Ammonia is toxic and converted to either urea which is excreted by the kidneys or glutamine which is converted back to NH4+ in PCT.
on a Na+ - H+ antiport carrier.
capillary blood via a symport carrier.
achieve this maximum.
○ 1. Reabsorption of filtered HCO3- (PCT_
■ Once HCO3- is fully reabsorbed, any further tubular H+ secretion
results in:
○ 2. Synthesis of new HCO3- (DCT/CD). Secreted H+ binds to urinary buffers.
■ Once urinary buffers are used up:
○ 3. Further H+ secretion and HCO3- generation occurs via ammonia (NH4+)
secretion/excretion (PCT). ● 2nd take home message:
○ 2. The amount of NH4+ excreted ○-
○ 3. The amount of HCO3- lost in the urine.
Acid Base Pathophysiology and Compensation to maintain pH Definitions:
- glutamine handling
The 4 pathological processes that can disturb pH:
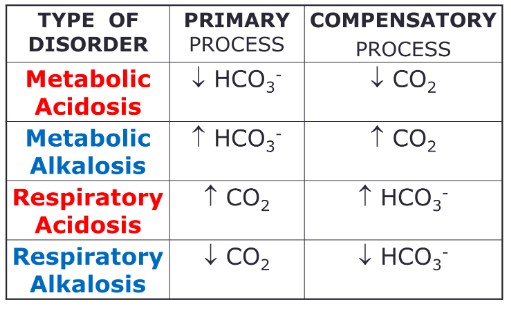
1. Metabolic Acidosis:
buffering of H+.
●
excretion ammonia.
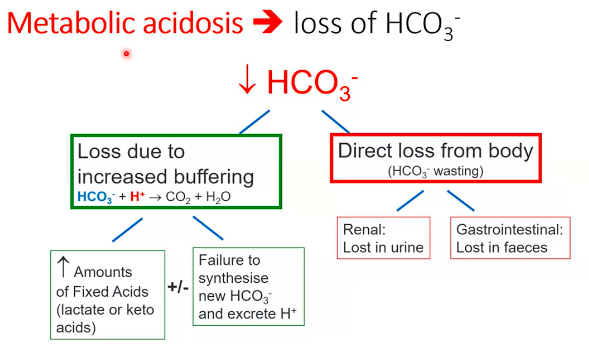
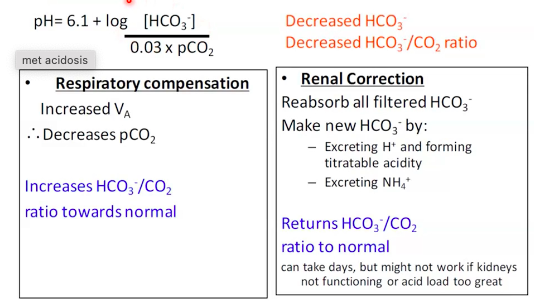
●
2. Metabolic Alkalosis
of HCO3- (antacids/renal problems).
■ ECF volume depletion
■ Hypokalaemia
■ Hyperaldosteronism (RAAS)
● Case: Excessive Vomiting
dehydration.
○
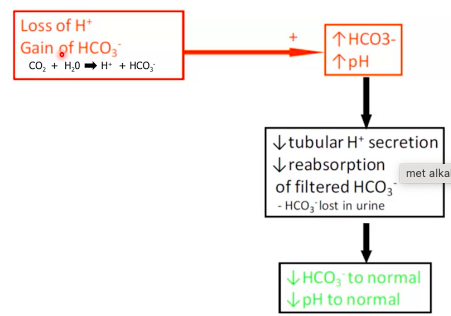

○
increased H+ secretion.
●
■ But limited by need to maintain oxygenation ● Renal correction:
■ But mechanism doesn’t work if volume depleted, decreased K+, aldosterone, etc.
3. Respiratory Acidosis
● The issue with respiratory acidosis is always a gain of CO2 in the body.
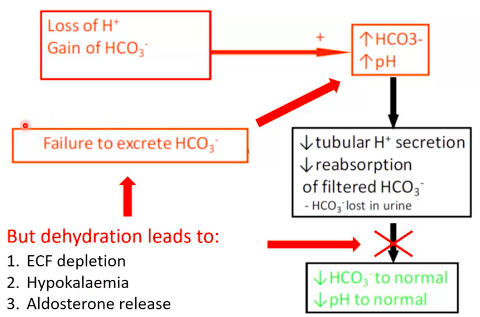
4. Respiratory Alkalosis
due to hyperventilation from a panic attack, stress/anxiety or certain drug use.
the alkalosis.
Summary
● Acid base disturbances are a major challenge in the body and manifest as a change
in the pCO2 to HCO3- ratio.
●
CHP207 Human Physiology
Week 1:
Ventilation and FRC Compliance
Compliance
● Ventilation requires:
○ Change in thoracic volume (P1V1=P2V2)
■ Normal Breathing = active inspiration, passive expiration
Ptp, greater lung V).
cavity with thin layer of pleural fluid with a negative pressure of -4mmHg
● Ventilation is affected by: ○ Compliance
○ Airway resistance
Pulmonary Compliance


volume
○ Specific compliance = compliance/lung volume
(FRC)
● Lung compliance is affected by:
expansion
ions.
surface tension between alveoli of different sizes
throughout the breathing cycle.
becomes more thinly spread, increasing surface
tension and decreasing rate of inflation
more surfactant therefore decrease surface tension
and slowing contraction of the alveoli
reduces surface tension and decreases the amount of
elastic recoil.

the lung at any pressure.
required to expand the lung.
Week 2
Airway Resistance
mouth and alveoli) and resistance to flow.
them open and allow easy passage of air to and from the alveoli ○ Trachea
■ Has cartilage rings, some smooth muscle ○ Bronchi
■ Irregular cartilage plates offer some rigidity and allow sufficient motion for expansion and contraction, smooth muscle.
○ Bronchioles
the alveoli
mainly pulmonary epithelium, some fibrous tissue and smooth
muscle fibres).
● Resistance to airflow in the airways
○ **exists only when air is moving
respiratory passage (resistance).
○ Poiseuille’s law: R=8ln/πr(^4)

○ Where l=length, n=gas viscosity, and r=the radius of the tube.
increases from the trachea (2-3cm2) to the alveoli (50-100m(^2))
the number of parallel terminal bronchioles (65 000) which air must pass through
the cross sectional area of the airways, therefore the velocity of the air moving through the airways decreases as it travels from the trachea to the periphery of the lung.
Elastic Recoil & Dynamic Airway Compression
● R is greatest upon expiration
○ Recoil of the lung during expiration is dependent on the condition of the lung parenchyma
○ Bernoulli principle - P will decrease as we move towards the mouth (a
constriction).
Dynamic Airway Compression ● During expiration:
● Forced breathing increases that pressure (from
muscles pushing)
pressure in the airways, compression of the airway may occur
If disease:
○ Can occur in diseased individual which is when gas is trapped in alveoli
because of the increase intrapleural pressures
Bronchial Smooth Muscle Tone
● Smooth muscle in the airways is under control of efferent fibres of the ANS
■ Released by mast cells, cause bronchoconstriction Work of Breathing

Week 3:
Pulmonary Diffusion & Gas Transport Atmospheric Air
Alveolar Air
The composition of alveolar air is not the same as that of the atmosphere because:
the air.
Therefore the composition of gas exchange as it is inhaled from the atmosphere, through the respiratory passage and to the alveoli.
The alveolar Partial Pressure of Oxygen (ideally 104mmHg) and carbon dioxide (40mmHg) is determined by:
Ventilation (VA) and perfusion (Q) Regional differences in ventilation
Matching Ventilation and Perfusion (VA/Q)
gas exchange and influence alveolar partial pressures.
○ Alveolar ventilation of ~5L/min and pulmonary blood flow of ~5L/min ● A mismatched can result in:
without undergoing gas exchange remains only partially oxygenated. This in effect ‘dilutes’ the oxygenated blood entering the left atrium.
■ In addition, some venous blood from coronary and bronchial circulation is added to the arterial blood diluting it slightly with venous blood (ie. normal anatomical features and venous admixture - slightly similar to dead space).
venous admixture
respiratory dead space.
category.
Diffusion across the Respiratory membrane:
following equation:
●
Anatomical Factors:
Factors relating to respiratory gases:
Partial pressure gradient:
What is the normal partial pressure gradient across the respiratory membrane for PO2 and PCO2?
PO2= 40 mm Hg and a PCO2= 45 mm Hg???
Diffusion constant: CO2 is 23 times more stable than O2, which molecule has a larger molecular weight? CO2
Diffusion Gradients
gradient.




Gas Transport Oxygen
Alveolar air:
Po2 = 105mmHg Pco2 = 40mmHg
Oxygenated Blood: Po2 = 100mmHg Pco2 = 40mmHg
Systemic tissue cells: Po2 = 40mmHg Pco2 = 45mmHg
Deoxygenated blood: Po2 = 40mmHg Pco2 = 45mmHg
Transport of Oxygen
● Only 3% of oxygen travels dissolved in the blood
○ Henry’s law
Hb+O2 -> Hb-O2
(4 times resting O2 requirement).

Haemoglobin (Hb):
○ Two alpha and two beta chains
Hb ranges from 0-4
○ Pulse oximetry (absorbance of light to determine saturation)
molecule which increases its affinity for O2, and therefore enhances binding
Oxygen-Hb Dissociation curve
saturation
saturated
(P50 for Hb) At lungs:




○ 50ml/L O2 delivered to the tissues for a ~60mmHg change in PO2
Factors which ‘shift’ the curve
● There are a number of factors which will change the
affinity between O2 and Hb, and therefore shift the curve left/right
○ Shift in the curve will change the P50 ● These include CO2, pH, temperature and
2,3-diphosphoglycerate (2,3 DPG or BPG). These factors facilitate
loading/off-loading of O2 at the lungs or tissue. Bohr Effect: pH and CO2
● At the tissues, the increase in blood CO2 and H+ enhances the release of O2 from the blood into the tissues
○ Decreased affinity -> right shift, increase P50 (increased oxygen off-loading) ● At the lungs, decrease in CO2 and H+ enhances oxygenation
○ Increased affinity -> left shift, decrease P50 (increased oxygen loading)
Hypoxia: low O2 at tissues
aerobic metabolism
Carbon Dioxide

● We carry CO2 in 3 forms:
CO2 Transport:
● 7% dissolved in plasma
○ It is this form that contributes to PaCO2
●
carbonic anhydrase (CA)
○ Facilitates CO2 loading at tissues, offloading at lungs
Carbon Dioxide dissociation curve
More linear relationship between PCO2 and CO2 in blood
Much steeper than the oxygen dissociation curve

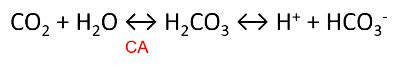

At tissue:
At Lungs:




Bohr Effect: pH and CO2
● Bohr effect - how CO2 and H+ affect Hb’s affinity for O2?
○ At the tissues, the increase in blood CO2 and H+ enhances the release of oygen from the blood into the tissues.
■ Decreased affinity -> right shift, increase P50
○ At the lungs, decrease in CO2 and H+ enhances oxygenation
■ Increased affinity -> left shift, decrease P50 ● This occurs because:
affinity of Hb for O2. Haladane effect
carbonic acid are buffered by Hb
carry H+ ions
becomes a more effective buffer -> ‘haldane effect.’

excess H+ ions and displaces carbon dioxide
Carbon Dioxide and regulation of acid-base balance
Excretion of CO2 by the lungs is an important mechanism in the regulation of acid-base balance
CO2 in the blood:
●
dioxide (VCO2) and inversely proportional to PCO2.
○ Eg 5L/min = 0.863 x 200ml/min / 40mmHg
Hypercapnia = elevated PaCO2 Hypercapnia may be caused by:
Week 4: Ventilation/ Perfusion Matching
● For efficient and adequate gas exchange to occur, ventilation must be matched to perfusion.

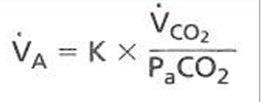
V/Q Matching
● In the lungs as a whole, ventilation and perfusion are well matched (ventilation is
~5L/min matched to cardiac output of ~5L/min). However there are regional differences in both ventilation and perfusion which lead to natural regions of mis-match.
Ventilation (V)
● There is a mismatch in ventilation due to gravity and the alveolar being smaller and
tightly packed in the bottom of the lung. This means that ventilation is greatest at the
bottom of the lung due to greater compliance of the alveoli and a larger SA. Perfusion (Q)
● In an upright subject, ventilation increases more slowly than blood flow in the apex of the lung to the base. Hence, the V/Q ratio at the apex of the lung is much greater than 1. Whereas the V/Q ratio at the base of the lung is much less than 1.
V/Q ratio and pulmonary gas exchange Ventilation
● Apex
● Base
Perfusion
● Apex

● Base

● VA/Q
○ No VA but normal Q (VA/Q=0), where units
ratios can range across extremes from:
contribute to the venous admixture
become part of the respiratory dead space.
within the ‘normal’ category.
Regional Differences Alveolar PO2/PCO2
(under-perfused) has higher PO2 and lower PCO2 than at the base, which is relatively under-ventilated (over-perfused).
Effect of Exercise on V/Q ratio
● Exercise, by recruiting more alveoli and more pulmonary capillaries throughout the
whole of the lung, but more especially in the apical regions, produces a smaller range of V/Q ratios.
Disease and V/Q mismatch
production).
○ Embolism or thrombosis.

EG. V/Q mismatch
● V/Q mismatch and PCO2/PO2 and chemoreceptor response
○
Control of Breathing Medullary Respiratory Centre
○ The most important of these are:
■ Dorsal respiratory group (DRG) ● Inspiratory centre
■ Ventral respiratory group (VRG) ● Expiratory centre
● The DRG comprises entirely of inspiratory neurons, whereas the VRG comprises both inspiratory and expiratory neurons.
VRG and forced breathing
forced expiration.
VRG expiratory neurons.
Pontine Respiratory Centre
medullary respiratory centre and fine-tunes respiratory rhythm.
phase of breathing.

● The apneustic centre in the lower pons prolongs inspiratory activity
○ The pneumotaxic centre usually keeps the apneustic centre suppressed.
Factors which alter ventilation
● THere are a number of factors which can influence the respiratory centre and alter
breathing pattern:
Central and Peripheral Chemoreceptors
The chemical control of breathing is one of the main regulators for our ventilatory pattern helping us to achieve our goal of maintaining arterial partial pressures and pH.
● Central chemoreceptors respond to increase in CO2 and H+ (low pH).
○
○ Doesn’t respond to O2.
Aortic Body and Carotid Body:
glomus cells.
their neurotransmitters out.
potential down the neuron.
potentials being sent along the neuron.
and therefore CO2 builds up in the cell, setting off the vesicles. Additionals points:
● Chemoreceptors only detect/respond to the dissolved for of each gas - ie. PO2 and PCO2 - they do not detect changes in the oxygen and/or carbon dioxide that is bound to Hb.
response. Essentially the rate of firing from the chemoreceptors will decrease (“quieten”).
Chemical Control of Breathing
● Among the most important inputs to the brainstem control centres originate in the
chemoreceptors
○ Centrally located in the medulla
■ Changes in PCO2, H+
○ Peripherally located in the aortic and carotid bodies
■ Changes in PO2, PCO2, H+ Peripheral Chemoreceptors
forms a branch of the glossopharyngeal cranial nerve (IX) which projects to the
medulla.
Peripheral Chemoreceptors: PO2
ventilation?
PO2, we still have a high range of % saturation and can safely deliver enough
oxygen to cells.
drops drastically.
Peripheral Chemoreceptors: PCO2 and H+
● Peripheral chemoreceptors respond to PCO2 in arterial blood.
○ They respond more rapidly than central chemoreceptors, but overall provide
only 25-30% of the total ventilatory response to PCO2.
● Unlike the central chemoreceptors, peripheral chemoreceptors respond to changes in arterial [H+].
○ Eg. ketoacidosis.
● Unlike O2, HCO2 only needs to change by 1mmHg to get a response.
PACO2 and ventilation
linear.
there is a significant increase in the slope of ventilatory response to CO2. Chronic effects of Hypercapnia
~1/5th its initial effect due to renal compensation which normalises pH.
○ Eg. COPD - increased PCO2, with normal blood pH.
Week 5
Nervous System Membrane Potential
would take to move a charged particle (eg ion) from point A to point B.
particles in that area and there you are going up a concentration gradient.
charges to positive voltages.
force trying to drive them inside the cell.
Potential
Potential DIfference
Charged particles can move in a conductor
Important Points:
voltage.
The Equilibrium Potential
respectively).
respectively).

+40mV.
far fewer of them.
than Na+.
○
amount of substance and temperature.
amount of a substance and its electric charge.
were allowed to flow.
○ To calculate where the membrane potential actually gets to, you need to know
how easily the ion flows, as the bigger a proportion of the total ionic flux an ion makes up, the closer the resting potential will be to that ions equilibrium potential.
The resting membrane potential and Action potentials Electrochemical Gradient
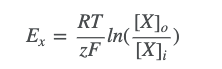

outside.
to draw it back in.
enough potassium going out to reach equilibrium potential.
Nernst Equation
● At the equilibrium potential for ion X (Ex), the two balance:
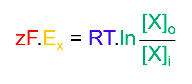
○ ○ ○ ○ ○ ○ ○
●
Z: valence for ion
F: faraday’s constant (96,500 coulombs per mole) R: universal gas constant (8.314 J deg-1 mole-1)
T: absolute temp (kelvins; 0oC = 273K, 37oC = 310K) [X]o = concentration of ions inside cell
[X]i = concentration of ions inside cell
- rearranged formula to find equilibrium potential
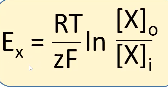
Equilibrium potential:
Eg:
RT/F = 27 “mV” Potassium: [K+] = 4mM
[K+] = 150mM Z = +1
Hint: think of cells inside sea water, salt is high in water, therefore NaCl is high outside cell.


Calculating Resting membrane Potential
as the reciprocal of resistance.
and low resistance.
●
Important Points:
to the flow due to concentration gradient.
membrane potential of a cell.
ion channels.
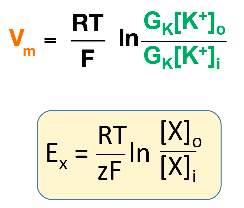
![]()
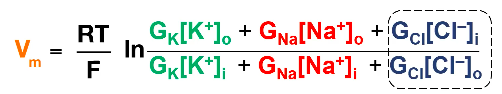
Action Potential
(H+H = Hodgkin + Huxley)
must be associated with the flow of an ion with an equilibrium potential of +40mV or
above - Na+ (+40mV) and Ca2+ (+120mV).
happens if you remove Ca2+ so therefore action potential must involve Na+.
increase in the conductance of an ion with an equilibrium of -70mV or lower - K+
(-90mV) and Cl- (-70mV).
K+ involved.
conductive to Na+ ions.
and become very permeable to K+
What currents cause the action potential?
Voltage Gated ion channels:
potential to depolarise a little bit, this then causes a few voltage gated sodium channels to open (activate).
○ After being open for a millisecond
or two, the inactivation gate slams shut, putting the gate into an inactivated state.


1. They conduct K+ not Na+
2. They do not inactivate: if the cell is more depolarised than about -40mV, these
channels open.
3. Importantly, these channels open more slowly than Na+ channels.



Relative Refractory Period
further than usual from threshold.
to excite to threshold.
larger than usual stimuli.
Important Points:
channels.
Week 6 - Nervous System II
Neurotransmission
other neurons.
to the synapse.
initial segment.
channels, this not only depolarises the axon initial segment, but it also beings to
weaky depolarise nearby parts of the axon.
sodium channels to cause the action potential to begin there too.
sodium channels to open, and so on, down the length of the dendrite.
What happens when the action potential gets to the end of the axon?
presynaptic terminal.
channels to open, allowing Ca2+ to enter the presynaptic terminal.
them to eject their contents into the synaptic cleft, the small (20nm wide) gap between the presynaptic terminal and the postsynaptic cell.
● The time delay between when Ca2+ influx occurs and synaptic release happens is as brief as 100μs.
● This is notable bc Ca2+ diffuses very slowly within the cell due to the presence of a huge variety of proteins which bind calcium.
● This means that in order for release to happen this quickly, the neurotransmitter containing vesicles must be physically very close to the voltage gated Ca2+ channels.

Synaptotagmin - the calcium sensor of the vesicle, once calcium activates synaptotagmin, the process of exocytosis begins.
The SNARE Complex - a complex of proteins which tether the vesicle to the membrane and force it into the membrane to cause release, once synaptotagmin is activated by Ca2+.
● Once released into the synapse, the neurotransmitters simply diffuse across the synapse.
● Diffusion can be very slow over long distances, however, given the minute distance the neurotransmitters have to diffuse over the synapse (~20nm), the diffusion is almost instant on a biological time scale, taking only a few nanoseconds.
A = Action potential
B = Ca2+ entering the cell just after the action potential starts (measured in Amps).

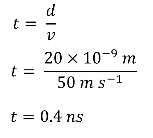
Overview
Glutamate Neurotransmission
(there are approx 100 in total).
Glutamate Receptors
● Glutamate acts on glutamate receptors, which fall into 3 broad classes:
3. Metabotropic glutamate receptors - which commonly have the name mGluR with receptor number.
AMPA/KA Receptors
acid (KA) allowed the identification of two receptors found on almost all neurons.
close to 0mV.
and GluA4 subunits, and typically made up of 2x GluA1 + 2x GluA2 OR 2x GluA2 + 2x
GluA3.
NMDA receptors
GluN2C, GluN2D, GluN3A and GluN3B subunits.
and hence have an equilibrium potential near 0mV.
Ca2+ is about 10% that of Na+/K+, so it doesn’t really effect their equilibrium potential
but the permeability is enough to provide a massive source of calcium).
become significantly conductive when the cell is depolarised beyond -40mV.
receptor is impermeable to Mg2+ and hence it essentially plugs the NMDA receptor.
receptor, allowing Ca2+ (and Na+/K+) to enter/exit the cell.
allows strong excitatory input to leave a biochemical “footprint.”
Ca2+ will enter the cell, modulating Ca2+ sensitive enzymes/proteins.
Metabotropic Glutamate Receptors (mGluRs)
trimeric protein which splits in two when activated.
beta/gamma subunits module ion channels.
GABA Neurotransmission
amino acid glutamine.
GABAA Receptors
binding - in this case chemical GABA].
receptor different properties.
and are often expressed in different locations and drug designers had high hopes for
being able to treat specific diseases by targeting different types of GABAA receptors.
membrane becomes more permeable to Cl-.
○ That depends on the equilibrium potential for Cl- in that cell.

● Does this mean that GABA has no action in these neurons?
○ No, It means that if any other source of depolarisation is impacting on the cell,
then the action of the GABAA receptors will work to bring the membrane
potential back to the Cl- equilibrium potential/resting membrane potential.
cell to the AP threshold.
GABAB Receptors
their ligand, they activate the G-protein biochemical cascade.
differs on where they are located.
gated calcium channels, meaning that when the action potential invades the presyaptic terminal, less Ca2+ enters the terminal, and hence less neurotransmitter is released.
Neurotransmitter Clearance
from the synaptic cleft.
gradient, so the concentration of neurotransmitters outside the synapse must be kept
close to 0.
neurotransmitters, or by transporters, which pump the neurotransmitters into a cell.
transporter of glutamate is particularly noteworthy.
○ Once glutamate is released, it is taken up by glutamate transporters on the
processes of astrocytes (type of glial cell) that surrounds excitatory synapses.
Week 7 - Nervous System III
Coordinated Action of the Nervous System
movement and control of involuntary responses which can maintain organ function.
recived in the brain and activation of the somatic motor system to
initiate a motor action.
system which control involuntary responses.
■ Effector actions include reflexes, coordination and optimisation which
tunes the body to suit certain behaviours or actions.
○ Endocrine - these responses are unto the control of the hypothalamus that
can control endocrine output.
■ Effector actions include systemic control and whole organism action as
a result of secretion of hormones that influence function. Overview of Nervous System:

Autonomic Nervous System:
both maintenance and activity of specific organs/tissues.
performance.
conservation and rebuilding.
activation of the SNS or the PSNS depending on the circumstances.
responses.
cardiac muscle, smooth muscle and glands.
heavily myelinated axons that extend in spinal or cranial nerves down to skeletal
muscles, the ANS uses a two-neuron chain to its effectors.
spinal cord and their axons are lightly myelinated thin fibres.
which sits outside the CNS.
effector tissue.

Sympathetic Nervous System (SNS)
Sympathetic Action | Target Tissue |
Increased HR | Heart |
Increased BP | Blood Vessels |
Relaxes and opens airways | Lungs |
Mobilises energy stores | Muscle and Fat cells |
Increases blood flow to muscles | Blood Vessels |
Stimulates sweating | Sweat glands |
Shunts blood to heart, lungs and brain | Blood vessels |
Suppresses digestion | Gastrointestinal (GI) system |
Relaxes bladder and rectum | Urinary and GI systems |
Action of the SNS on target tissues
the release of the action hormone adrenaline or noradrenaline directly to tissues,
orchestrating a coordinated response of many organs.
into the bloodstream.
ganglia near the CNS.
synapse in the sympathetic chain ganglia.
prevertebral ganglia (located away from spinal cord).
cord.
tissues such as sweat glands, hair erectors, blood vessels, heart and lungs (stellate
ganglion), salivary glands and pupils (cervical ganglia).
their effector tissue.
Parasympathetic Nervous System (PSNS)
Parasympathetic Action | Target Tissue |
Decreased HR | Heart |
Decreased BP | Acts via heart rather than directly on vessels |
Closes off airways | Lungs |
Constriction of pupils | Eyes |
Boost digestive system | GI system |
Contracts bladder and rectum | Urinary and GI systems |
Action of PSNS on target tissues:
(ACh) to target tissues.
○ eg. boosting the digestive system occurs through improving GI smooth muscle activity, GI blood flow, digestive enzyme secretion and GI hormone secretion helping nutrient absorption and use.
brainstem nuclei, and the sacral outflow from the lateral horn of the sacral spinal
cord.
(to the lacrimal and salivary glands), 9 (to the parotid gland) and 10 (to the vagus nerve to viscera).
Neurotransmitters of the ANS
adrenaline/noradrenaline.
using ACh on nicotinic ACh receptors.
tissues.
adrenaline/noradrenaline to act on adrenergic receptors on target tissue.
muscarinic receptors on target tissue.
Receptos:
act as ligand-gated ion channels for the cation Na+, K+ and Ca2+.
through depend on the subunit makeup.
PSNS at the first synapse.
nAChRs on skeletal muscle as part of the somatic nervous system.
tissue distribution allows them to have different actions.
the parasympathetic functions.
cells to elicit their function.
reside in, eg. activation of alpha receptors leads to vasoconstriction, GI shutdown
etc; activation of beta receptors leads to vasodilation, increased cardiac output etc.
function.
Smooth Muscle - the quiet effector cells
cells.
muscle cells produce a slow, prolonged contraction that makes it an excellent cell for
its homeostatic role.
control access to specific areas of the body.
products.
are ideal to maintain key bodily functions.
modulate activity.
skeletal muscles.
muscle.
contractile activity via pacemaking: leak channels (K+) which periodically depolarise.
actual contractions (Ca2+ influx).
channels that allow communication between cells.
contraction.
entry is enough.
calcium levels.
●
Autonomic reflexes
● Several CNS nuclei communicate to ANS ganglia to influence autonomic functions in a combined behavioural, autonomic and endocrine response.
● It is the solitary nucleus of the dorsal medulla that receives most of the visceral sensory input from the vagus nerve.
● It then feeds information up to the autonomic integration centres.
● These areas include the parabrachial nuclei (arousal and thermoregulation), the periaqueductal grey (nociception), the hypothalamus (neuroendocrine and physiological functions).


WEEK 8 - Cardiovascular Physiology I
The cardiomyocyte
contractions of the atria and ventricles to allow the pumping of blood around the
body.
and this has major impacts on whole body physiology.
intercalated disks.
action potentials from cell-to-cell.
propagate like a wave throughout the chamber and depolarise all of the cells - this is
called a functional syncytium.
the ventricles by a thick band of fibrous tissue meaning the heart has two functional
syncytia - the atria and the ventricles.
ventricular depolarisation.
system to enable proper cardiac function. Resting membrane potential in cardiomyocytes
●
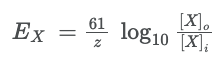
![]()
The cardiac pacemaker cells and spontaneous depolarisation
cardiac cells that form pacemakers.
intervals (~100/sec).
under normal healthy conditions.
act as the normal pacemaker.
pacemaker albeit with an overall lower rate of depolarisations (leading to lower HR).
the transmission of atrial action potentials/depolarisations to the ventricles via the Bundle of His.
Pacemaker cells

Phase
contribute
4 = pacemaker potential

Phase 0 = depolarisation
● Voltage gated Ca2+ channels open at -40mV. ● Ca2+ influx.
● Membrane depolarises.
Phase 3 = repolarisation
● Ca2+ channels close. ● K+ channels open.
● K+ efflux.
● Membrane repolarises.
Summary:
Contractile cardiomyocyte depolarisation and cardiac contraction
and cannot spontaneously depolarise.
membrane voltage is held constant at approx. -90mV.
potential... which is generated at regular intervals by pacemaker cell depolarisation
in the SA node.
potential generated by pacemaker cells activates adjacent contractile cardiomyocytes and they continue to spread the action potential like a mexican wave through the atria in a cell-to-cell manner.
favours movement of K+ out of the cell.
over ~200ms. The plateau phase is maintained by a balance between
continues K+ efflux and Ca2+ influx.
greater efflux of K+ and membrane repolarization.
○ Phase 4 - resting phase is re-established, and ion concentrations are rapidly brought back to normal by ion pumps/exchangers.
How do the action potentials cause muscle contraction?
300ms.
depolarised (+tve membrane potential).
take place within a cardiomyocyte:
Video:
Excitation contraction coupling and the refractory period Cardiomyocyte depolarisation
Excitation-contraction coupling (ECC)
The more calcium that is present in the cell, the more powerful the contraction AND The longer the calcium hangs around, the longer the contraction.
Refractory period
● Effective/absolute refractory period

● Relative refractory period
What happens if we try to stimulate more action potential in the refractory period? Tetanic Contractions
● Skeletal muscle
stimulation.
stops the valves from opening and the
heart from filling.
Summary: Refractoriness and non-tetanisation
before the heart can contract again.
Control of frequency and force of contraction
depolarisation.
a coordinated sequence to generate contraction in an efficient way for blood to be
pumped.
the:
muscle).
Video:
Parasympathetic and sympathetic modulation of HR Cardiac Automaticity - signal generation
● Cardiac action potentials are initiated in collections of specialised cardiac cells that form pacemakers.

○ Physiological pacemaker under normal conditions. ● AV NODE
○ Can take over as pacemaker if SA node is damaged.
Multiple autonomic nervous system inputs PNS (vagus)
signal is strong enough).
SNS
PNS - decreasing heart rate
WEEK 9 CV2: control of cardiac output and blood pressure
Cardiac output
pressure necessary to provide adequate blood flow to the organs.
to generate sufficient pressure to propel blood from the left ventricle, through the
aortic valve and into the aorta.
pulmonary valve and into the pulmonary artery to perfuse the lungs.


Factors influencing venous return
venous system forcing blood back towards the heart (venous valves prevent
backflow).
muscles, propelling blood back through the venous system to the heart.
○ This is because average resting HR
is ~60bpm and SV is ~80mL/beat
(60x80=4800)
to ~35L/min and this is driven by the work/oxygen requirements of working skeletal muscle.

Regulation of HR (and CO)
increase from the resting state and therefore can contribute a greater relative
increase in CO when required.
depolarization of the pacemaker cells in the SA node.
systems have direct inputs into the heart to regulate HR.
The Bainbridge Reflex
increased HR.
distend the right atrium, but denervation of the vagus nerve to the heart eliminated
these effects.
arterial stretch receptors.
via the vagus nerve to the heart, leading to increased HR.
○ Increased venous pressure -> pressure in vena cava increases -> pressure in right atrium increases -> stretch receptors activated -> signal to the medullary control centre to decrease PNS activity -> increased HR.
Regulation of Stroke Volume (and CO)
every time the heart beats.
○ These are preload, afterload and contractility.
Preload
greater the force of the contraction.
Preload continued (video)
generated in the subsequent contraction.
(ie stronger contraction). Mechanism of length-tension relationship
● It has been proposed that placing the sarcomere under more tension increases the sensitivity of troponin C to Ca2+.
○ Ie it increases the excitation-contraction coupling efficiency. Preload
The frank-starling curve
● THis is a ventricular function curve, and clearly shows the frank-starling mechanism
at work.
●
Implications of the Frank-starling mechanism
flow to the tissues.
● When HR increases, time available for filling decreases.


relatively greater importance.
Afterload
○ Eg when lifting a weight.
● The afterload of the left ventricle is closely related to aortic pressure.
The ventricles have muscle arranged in roughly a sphere shape
related to the tension in the ventricular wall.
says thats:
Analogy - balloon filled with air
● An increase in radius increases wall stress
Afterload and Cardiac Function
during each ventricular contraction. The frank-starling curve: effect of afterload
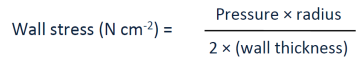


What can change afterload?
eject blood).
○ Eg. dilated cardiomyopathy ● Wall thickness
○ Eg. ventricular hypertrophy decreases wall stress and is a compensatory mechanism for when afterload increases.
Contractility - The performance of the ventricular myocytes at a given preload and afterload. Contractility of cardiac muscle
phase/contraction.
The Frank-starling curve: effect of contractility
function curve.
any given preload.
any given preload.
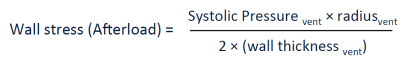

Cardiac output provides arteries with BP
pressure differences present between the large arteries (high pressure) and large
veins (low pressure).
time it beats.
at ~120mmHg during systole and dropping down to ~ 80mmHg during diastole in a
resting, healthy individual (this is where the 120/80 mmHg comes from).
arteries and arterioles that supply capillary networks within organs
○ These arteries/arterioles are typically called the resistance vessels and
collectively they determine the total peripheral resistance (TPR).
blood pressure is much lower compared with the arterial system.
as it enters the right atrium.
Mean arterial blood pressure (MAP)
cardiovascular function and for assessment of organ perfusion.
sometime also called systemic vascular resistance [SVR]), and central venous pressure (CVP) according to the following relationship, which is based upon the relationship between flow, pressure and resistance (Ohm’s law):
![]()
![]()

average (100mmHg when time is ignored) of the systolic and diastolic pressures.
Regulation of mean arterial blood pressure (MAP)
blood vessels during a single cardiac cycle.
perfusion (blood flow) of the organs of the body at any given time (normal range is
between 70 and 100mmHg).
pressure alone (because it also relies on TPR and thus tells us about peripheral
blood flow).
arteries, kidneys, and brain to keep them functioning.
consequences and these will manifest acutely within vital organs. Problems when MAP is too high:
Problems when MAP is too low:
● Low MAP can be life-threatening as well.
keep MAP regular at all costs.
Physiological feedback loops - 3 major parts 1. Sensor
○ Detects a change in a physical or chemical property that (eg. vascular stretch in a baroreceptor).
2. Control centre
3. Effector
muscle relaxation).
Physiological feedback and short-term regulation of blood pressure
○ The vasomotor centre in the brain stem (medulla).
3. Multiple effectors
Short-term control of MAP Baroreceptors - direct sensors

![]()
by inhibiting vasoconstriction.
systems.
Baroreceptor reflex
neurons and inhibits the PNS neurons in the medulla.
quickly correct the fall in BP.
Chemoreceptors - indirect sensors
causes peripheral vasoconstriction, increased HR and increased SV.
Chemoreceptor reflex
stimulation of the heart, which increases the HR.
stimulation of the heart, which increases the HR and SV.
stimulation of blood vessels, which increases vasoconstriction.
Summary:
to ensure organ perfusion.
activate/deactivate the cardiovascular centre as needed.
to changes in O2 and CO2.
Long-term control of MAP
of blood volume.
cardiovascular system and we see blood pressure increasing.
system and therefore blood pressure decreases.
renal system.
tightly regulate blood volume by controlling excretion rates of Na+ and water in particular.
●
volume/pressure homeostasis.
WEEK 10 CV3: Control of blood flow and special properties of the macro- and micro- circulation.
Haemodynamics and control of blood flow

● How constriction and dilation impacts the flow of blood through arteries can be explained by Ohm’s law and Poiseuille’s law that describe the relationship between radius, resistance and flow.
Basic haemodynamics - Ohm’s and Poiseuille’s laws
○ During ventricular systole, blood is ejected from the ventricles at very high pressure.
○ The bulk flow of blood in the cardiovascular system is from high to low pressure.
○ Resistance is:
The flow through a tube is (pressure difference x radius to the power of 4) divided by (viscosity x tube length).
In the body:
heart beat-heart beat.
Vessel radius is a very powerful determinant of both resistance and flow


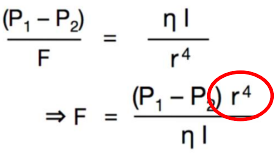



○ The further the blood is located from the vessel wall the faster the flow.
Using the whole circulation as an example
● In this case, the flow is the entire output of the heart =
cardiac output.
peripheral circulation.
Control of blood flow in the body
use, but dispensable organs that get blood flow when there is enough to go around.
○ The radius of the blood vessels it is flowing through.
they can handle an order of magnitude more blood flow because thier resistance to
flow has decreased dramatically.
where blood flows in the body - these arterioles are often referred to as ‘resistance vessels.’
In the body, six primary factors can modulate the radius of resistance arterioles to regulate how much blood flow they receive:
1. Special receptors
○ Example: skeletal muscle arterioles dilate in response to moderate levels of
adrenaline or insulin. 2. Metabolic control
○ Example: diffusion of metabolic waste products (adenosine, ADP, CO2, lactate, K+ etc) from active cells causes arteriolar vasodilation.
3. Oxygen deficiency (low pO2)
○ Example: oxygen removal from blood by active cells reduces local blood
oxygen levels -> stimulates arteriolar vasodilation. 4. Sympathetic nervous system


○ Example: SNS has direct inputs into most blood vessels and SNS outflow can alter constriction/dilation in arteries and veins to regulate blood flow to individual organs.
5. Nitric oxide
○ Example: shear stress (force exerted by blood on endothelial cells = blood
viscosity x blood velocity/internal diameter) in arteries and arterioles causes their lining endothelial cells to release Endothelium Derived Relaxing Factors (EDRF) like nitric oxide -> nitric oxide is a potent vasodilator.
6. Longer-term control mechanisms
○ Example: capillary density changes (eg. in exercise trained skeletal muscles)
meaning the arterioles need to supply more flow to that tissue -> arteriolar
vasodilation.
Autoregulation and rationing of cardiac output
Regulation of blood flow by metabolism

‘Autoregulation’ of blood flow
● Why is flow not a passive reflection of pressure changes in certain organs?
○ Because some organs require constant blood flow to maintain their functions despite changes is systemic blood pressure
● Two mechanisms are responsible for this in those organs:
prominent in arterioles.
release of vasoactive metabolites and low O2 tension.
Even if systemic arterial pressure changes, blood flow in some organs remains stable
● This is called autoregulation and it is achieved by automatic changes in vascular tone in response to blood pressure.

Total systemic blood flow (CO) is the sum of blood flow to all organs
demands for blood/nutrients.
Sympathetic nervous system innervates the vasculature to regulate in this instance


What if cardiac output is limited?
maintain perfusion.
and reduced perfusion.
Special properties arteries, veins and capillaries
branch into successively smaller arterial vessels until
they become capillaries.
continue to join with other veins until they enter either the superior or inferior vena cava that bring the blood back into the right atrium of the heart.
Functions of the aorta and large arteries
blood to the systemic arterial system, dampens the pulsatile pressure that results
from the intermittent outflow from the left ventricle.
flow is a function of the aortic compliance - the ability of the aorta to stretch when it fills with blood during systole and then recoil back to its resting state by actively contracting to continue pushing blood forward.
involved in the regulation of arterial blood pressure as well as blood flow within organs.


basement membrane, are termed capillaries.
Functions of the capillary network
● Capillaries represent the smallest vessels (~5-10um in diameter) within the
microcirculation and arguably perform the most important role of the cardiovascular systems:
○ They facilitate nutrient exchange between blood and tissues they supply.
constriction/dilation controls capillary flow.
each downstream capillary equally.
themselves have cells wrapped around them that can constrict/dilate individual capillaries - these cells are called pericytes and they are similar to smooth muscle cells.
through. The kidneys are the primary site in the body where these capillaries are found.
Starlings forces and capillary filtration
Movement of solutes or filtration across capillaries depends on 2 things:
1. The balance of hydrostatic and osmotic pressure across the capillary
○ This is called the Net Filtration Pressure (NFP) = total pressure promoting
fluid movement to/from capillary
2. The surface area and permeability coefficient (Kf) of the capillary
Starling’s forces: pushing fluid in and out of capillaries
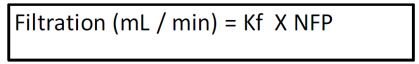

So does that mean very little fluid flow to or from capillaries?
Yes and No
Yes: The average capillary hydrostatic pressure is ~17.3mmHg. Therefore, there is not much net movement of fluid to and from the interstitial space.
No: The arterial end of the capillary is at higher pressure than 17.3
The venous end of the capillary is at lower pressure than 17.3
Blood flows from high-low pressure

Net Filtration Pressure - Arterial end of capillary
Net Filtration Pressure - Venous end of capillary
The amount of filtration at each end is determined by the NFP and the permeability (Kf)
Notice the difference in pressure/amount of flow between the two ends of the capillary
~90% of the fluid leaving the capillary at the arterial end comes back into the venous end.
Venous end of capillaries have a higher Kf Filtration (mL/min) = Kf x NFP



If 90% of the fluid comes back into the capillary, where does the remaining 10% go?
● To quantify: 10% = ~2ml/min
● Over the course of 24 hours = ~3000mL or 3L
● If this fluid was not removed, you would lose a lot of blood volume very quickly.
● The excess fluid left in the interstitial space (~3000mL/day) is drained away by the
lymphatic system.
Functions of the vena cava and veins
exchange vessels, particularly for large macromolecules as well as fluid.
intervals - this means blood flow in the venous system is unidirectional.
again appears.
constricting, and serve an important function regulating capillary pressure.
- ie. the site where most of the blood volume is found and where regional blood volume is regulated.
○ For example, constriction of the veins decreases venous volume and increases venous pressure, which alters cardiac output by increasing venous return (preload).
● The final venous vessels are the inferior and superiour vena cava, which carry the blood back to the right atrium of the heart.
Arterial and venous compliance and capacitance
depicted:
decreases as pressure increases.
vessels become “stiffer” at higher pressures and volumes).
the compliance of a vein is about 10 to 20 times greater than an artery.

Exercise, venous return and the skeletal muscle venous pump
is the muscle venous pump.
flow away from the limb and back toward the heart.
muscles surrounding them contract, and they become decompressed as the muscles
relax.
stages of compression and decompression (ie. there is pumping).
venous segment.
towards the heart.
flow that enhances venous return.
relax to keep the body in balance.
pressure and venous return, and lowers venous and capillary pressures in the feet and lower limbs.
The lymphatic system and vessels
that reside near the normal capillary bed in all tissues within the body.
fluid/protein/bacteria etc. into the lymphatic vessel.
from tissues to the larger lymph vessels and ultimately back into the systemic circulation.
- specialised structures that also contain a large number of lymphocytes and
macrophages that monitor the lymph for pathogens.
not lost in the interstitial space within tissues.
interstitial space and returns this to the systemic circulation at the subclavian vein.
day.
Regulation of lymphatic flow rate
What controls movement of fluid into lymph vessels?
What happens if we exceed this max lymph flow rate?
capacity:
●
What factors can cause oedema?
overload).
![]()
![]()
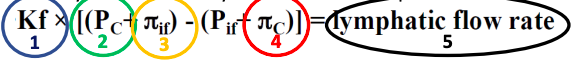
WEEK 11
Renal System 1 - Basic Renal Functions and Transport Processes in the Nephron Intro
from the body by forming and excreting urine.
deadly consequences for the rest of the body.
Excretory functions of the kidneys
discrete sections:
the distal end to form a collecting tubule that eventually turns into a collecting duct and delivers urine to the calyxes and eventually the ureters.

Events that determine renal excretion
into the Bowman’s capsule from the glomerulus.
the nephron tubules back into the body.
peritubular capillaries that surround the nephron into the tubular network.
● Therefore the amount of any substance that is excreted from the body is determined using the following equation:
○ Excretion = Amount filtrated - Amount reabsorbed + amount secreted
Filtration
● The process where blood is filtered from the glomerular capillaries into the
Bowman’s Capsule through a serious of pores that make up the filtration barrier.
●
Glomerular filtration barrier Anatomy of the barrier:
extensions (pedicles).
layers.
Glomerular filtration barrier: size
size).


Glomerular filtration barrier: charge
prevents their passage into the filtrate.
○ In disease states (eg. glomerular nephritis or diabetic nephropathy) this
barrier lsoses its selectivity allowing protein into urine -> proteinuria. Glomerular filtration
filtrate as plasma.
Glomerular filtration rate (GFR)
plasma entering the kidneys per minute - ie. renal plasma flow is ~600mL/min.
the glomerular filtration rate (GFR).
nephrons.
volume is filtered 60 times per day.
○ This provides the kidneys an enormous capacity to regulate fluid and
electrolyte levels in the body to ensure homeostasis.
regulated to ensure there is not too much or too little getting to the nephron where
filtration happens.
afferent and efferent arterioles.
regulate how much filtrate is formed by the kidneys at any given time.
Control of glomerular filtration rate (GFR)
● GFR is dependent on 2 factors:
1. The surface area and permeability of the filtration barrier
○ Expressed as the constant Kf
2. The Net Filtration Pressure (NFP)
○ THe balance of hydrostatic and osmotic pressures at work across the filtration
barrier. GFR and NFP
NFP = outward - inward
Modifying GFR through arterial constriction
What happens if both vessels constrict?
Summary of arteriolar control of GFR
● The majority of physiological control of GFR occurs through alteration of the
glomerular capillary hydrostatic pressure (PGC)



Renal Autoregulation
ensure constant renal blood flow and GFR despite potential changes in systemic
blood pressure - this is autoregulation.
blood flow despite changes in systemic blood pressure.
influences and therefore is intrinsic to the organ, although these influences can
modify the response.
pressure.
○ Afferent and efferent arteriolar resistance are altered to keep GFR steady.
● Why is this useful?
○ Allows the kidneys to maintain their excretory capacity and blood volume
homeostasis even when systemic blood pressure changes. Renal Autoregulation: Myogenic mechanism
Vessel diameter unchanged, hydrostatic pressure increases, GFR increases.
Afferent arteriole constricts due to smooth muscle stretch-feedback, hydrostatic pressure and GFR largely unchanged.
Renal autoregulation: tubulo-glomerular mechanism
very close to the afferent and efferent arterioles.
through the DCL.
fluid has decreased.
receive a signal which then causes them to produce more renin.
arteriolar constriction and increases back pressure inside capillaries.


● Macula densa talks directly to afferent arterial to decrease resistance and increase dilation, increasing GHS.
Renal Autoregulation
● Myogenic mechanism
○ Very fast to act and accounts for ~50% of renal autoregulation. ● Tubulo-glomerular feedback mechanism
○ Slower to act and accounts for ~35-40% of renal autoregulation.
● Renal blood vessels can respond without SNS/humoral input to ensure a steady
GFR despite changes in systemic pressure.
Reabsorption and secretion
through re-absorptive processes.
on secretory processes.
network within the nephron (excluding bowman's capsule).
most move between the tubule/capillaries by using either transcellular (through cells via transporters or ion channels) or paracellular (between cells using tight junctions) or both.
Principles of tubular handling of filtered substances
nephron, not every substance that is filtered can be reabsorbed and/or secreted.

beyond what is filtered from the blood alone.
this substance from the peritubular capillaries and into the tubules.
filtered nutrients.
blood (~145mmol/L).
concentration, very large amounts of Na+ are filtered all the time.
control of how much Na+ is excreted from the body (depends on homeostatic
needs),
reabsorbed from the filtrate (allowing more to leave the body).
has multiple physiological inputs that can regulate Na+ balance.
from the filtrate. We can also switch on the active tubular secretion if further
K+ excretion is required.
the body and the body has multiple physiological inputs that can regulate K+ balance.
● Water (H2O)
of urine per day.
percent (1-2L) can be reabsorbed or not depending on water homeostasis
requirements.
filtered H2O.
has multiple physiological inputs that can regulate water balance.
The limits of tubular transport
far exceeds our total blood volume (by a factor of 30) and thus most of what is
filtered must be reabsorbed back into the body to maintain homeostasis.
there are two limiting factors to be aware of:
number of transporters available that can move that substance from the tubular
lumen back into the body. Therefore reabsorption can be ‘Transport Limited’
maximum concentration gradient that can be achieved and maintained between the tubular lumen and the interstitial space. Therefore reabsorption can be ‘Gradient Limited’
Transport limits and osmotic diuresis
Reabsorption of some substances is rate-limited by transporter capacity
Glucose transport in the nephron
○ Note - blood flow prevents glucose build up in interstitial space, so conc. Gradient is maintained to favour glucose movement into the bloodstream.
Glucose is a good example of Tm limited transport
● At normal glucose concentrations (~5mM) all of the filtered glucose is reabsorbed.
○ Although some SGLT2 inhibitors (reduce Tm) are used to reduce glucose levels in people with T2D.
Gradient limited transport - eg. Na+ in the proximal tubule
● ●
●
Some substances (typically ions) do not have a Tm
Upper limits of transport is defined by the concentration gradient that can be sustained across the tubule.
Eg. as Na+ is reabsorbed in the proximal tubule,
○ Lumen [Na+] decreases while interstitial [Na+] increases
○ The tight junctions in the proximal tubules are slightly leaky to Na+
○ The gradient limit for Na+ is only 2 mmol/L, once the interstitial [Na+] exceeds
tubular lumen [Na+] by more than 2mmol/L, Na+ starts to leak back into the
tubules through paracellular pathways (tight junctions become leaky).
○ Under normal circumstances in the proximal tubule, water follows Na+ so that
the lumen [Na+] doesn’t change and is not anywhere near the gradient limit.

This is ●
●
the normal situation... what happens if we stuff things up with an ‘osmotic diuretic’ What if we intravenously infuse a patient with a solute that stays in the extracellular fluid, is freely filtered but poorly reabsorbed in the kidney.
Eg. Mannitol

Osmotic diuresis - mannitol/water
○ 1000ml fluid into tubule from glomerulus
○ 334ml fluid out of distal nephron as urine (65% reabsorbed)
○ 1000ml fluid filtered into tubule from glomerulus.
○ 500ml fluid out of distal nephron as urine (50% reabsorbed)
Osmotic diuresis - mannitol and Na+
○ [Na] in interstitium and peritubular capillaries is 150 mmol/L
○ [Na] in tubular fluid going out of distal nephron as urine is 150 mmol/L
○ [Na] in interstitium and peritubular capillaries is 150 mmol/L
○ [Na] in tubular fluid going out of distal nephron as urine is 100 mmol/L
Late PCT with Osmotic Diuretic
Osmotic diuresis
increased volume of urine, quickly causing dehydration.
reabsorbed out of the lumen.
water and Na+ out with it to increase urine output.
○ This is why uncontrolled diabetics urinate a lot and are always thirsty.
Take home message: there are limits on tubular transport
Or
these transport limits.
WEEK 12 - Renal System 2 - Water, Sodium and Potassium homeostasis
Variation in water and solute handling across the nephron
processes available to each segment.

● Most of the physiological control of water, Na+ and K+ occurs in the distal nephron and collecting duct.

●
Antidiuretic Hormone (ADH)
blood volume.
on principal cells to increase water reabsorption in the distal nephron (inserts
aquaporin 2 channels into cells).
becomes permeable.

Counter-current mechanism and renal interstitial solute concentration
ducts, loops of henle, vasa recta (capillaries) and the interstitial space.
of urine concentration and other specialised kidney functions.
Video 1
The role of the counter-current mechanism and ADH in urine formation
What mechanism produces the concentration gradient in the renal medulla?
● There are 2 main factors:
concentration of interstitium to increase.
1. Loop of Henle permeability to water and solute


2. Medullary blood flow
○ 1. Medullary blood flow is much slower than in the cortex
■ While useful for concentrating urine, this does increase the susceptibility of the medulla to ischemic injury.
○ 2. The arrangement of the medullary blood supply into the descending and ascending vasa recta limits the washout of the medulla hyperosmolarity.
Countercurrent Mechanism Countercurrent exchange
which is taken back by ascending limb of vasa recta.
medulla.
of vasa recta maintains the osmolality gradient established by LOH.
Urea and the osmolarity in the medulla
from NaCl.
Recycling of urea in the distal nephron

How much urine volume do you need to produce each day for excretory homeostasis?
excreted by the kidney daily.
per day or as much as 15L (min ADH effect) per day.
concentration of the urine that can be achieved in the renal medulla according to the following equation:
○
loss from the skin, respiratory tract, and GIT, when water is not available to drink.
concentration of 1200 mOsm/L explains why severe dehydration occurs if one
attempts to drink seawater. Can you drink seawater to survive?
contributes about 600mOsm/L.
body of 1200 milliosmoles of NaCl ingested in addition to all the other soltes.
Water homeostasis
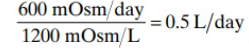
Video 2
Physiological Regulation of body water Basic negative feedback loops in physiology
● These mechanisms have two components:
specific set-point (eg. vascular stretch).
original disturbance (tries to restore homeostasis).
Body water homeostasis
water sensors are:
Osmoreceptors in the hypothalamus Baroreceptors (mechano-receptor)
water effectors are:
Posterior pituitary release of ADH (signal from hypothalamus)
ADH increases water permeability in the distal nephron (and increased inner medullar urea absorption).
Activation of the thirst centre increases thirst (and thus water intake)
○
Blood volume, baroreceptors and ADH release

Interplay of the osmoreceptor, thirst and ADH

Which is the more powerful mediator of ADH release?
Secretion of ADH
● ADH secretion depends on both plasma osmolarity and volume: 1. Plasma osmolarity
○ Very sensitive: responds to 1-2% change 2. Plasma volume
together and become synergistic (eg. dehydration, blood loss with fluid shift from
extracellular space).
hypotonic fluid replacement; eg. diarrhoea with normal water intake) and the result
will be determined largely by how low blood volume is.
Water homeostasis and thirst
dilate or concentrated urine via ADH).
homeostasis...
Sodium Homeostasis
motivated behavioural response that drives us to seek and ingest salt-containing
foods and fluids.

Why is salt so important?
a concentration of ~145 mmol/L.
chloride (Cl-) make up most of the plasma osmolarity (~300mOsm/L) under normal
physiological conditions.
plasma osmolarity.
(thirst) and output of water (kidneys, ADH status) and mediated by the osmoreceptors.
ECF volume.
therefore increased BP.
ECF volume (and thus BP)... because plasma osmolarity is monitored closely by the
osmoreceptors, water balance is adjusted to maintain a constant 300mOsm/L.
therefore low blood pressure because osmolarity is held constant.
volume and therefore blood pressure.
angiotensin aldosterone system and the atrial natriuretic peptide system.
Video 3
Control of Na+ in the body by the RAAS and ANP Renal regulation of Na+ excretion
● As Na+ is freely filtered at the glomerulus and actively reabsorbed, but not secreted then:
Negative feedback looks for Na+:
1. A sensor ->
○ 1. Extra-renal pressure sensors
■ Baroreceptors (activate CNS)
○ 2. Intrarenal pressure sensors
■ Juxtaglomerular cells (release renin)
○ 3. Indirect pressure sensor (NaCl delivery)
■ Macula desna (renal autoregulation/GFR control)
2. An effector ->
■ AND/OR
Regulation of Na+ by increased GFR
● Increasing arterial pressure causes a corresponding rise
in filtrate formation and therefore renal water output. ○ This is called pressure diuresis
● Increasing arterial pressure also causes a corresponding increase in Na+ filtration and therefore output.
○ This is called pressure natriuresis
● Therefore if pressure goes up, Na+ excretion goes up.
Regulation of Na+ by reduced GFR
Regulation of Na+ by reabsorption
● There are a number of stimuli that increase tubular reabsorption of Na+ (and
therefore Na+ in the body).


increase reabsorptive processes).
Renin angiotensin system in Na+ (and BP) homeostasis
Effect of angiotensin II on Na+ and water handling
causing Na+ reabsorption utilising H+ cycle
space at the expense of K+.
reabsorption in the proximal nephron..
Effect of aldosterone on Na+ and water handling
ATPase
● Remember that Na+ and water can be handled independently in the distal nephron
so while aldosterone will increase Na+ reabsorption, water will follow only if ADH is
also present.
What is we need to increase Na+ excretion?
○ Results in less water and Na+ reabsorption. What happens when we increase total body sodium?
(eg high salt diet)


Summary
volume
determines blood pressure.
Potassium Homeostasis
(~140mmol/L) within cells.
depolarisation/repolarisation in excitable cells like neurons and cardiomyocytes.
3.5-5mmol/L.
excreted from the body via the renal system.
How do we regulate potassium homeostasis?
then adding 100 mmol of K+ would increase the ECF K+ concentration by more than 3.5mmol/L.
○ We would almost double our plasma K+ concentration from ~4-7.5mmol/L if nothing else happened.
○ this is where the renal system comes in and helps regulate the output of K+ to match the input from the diet.
●
Video 4
Physiological control of body K+ occurs in the distal nephron

How ●
How ●
● How ●
● How ●
●
is K+ excretion regulated
As with other solutes, K+ is freely filtered, actively reabsorbed and variably secreted:
Reabsorption of K+ in the PCT is constant whether it's hypokalemia or hyperkalemia.
○ Ie. 65% of filtered load is reabsorbed
Reabsorption of K+ is passive through both paracellular and intracellular routes. K+ os reabsorbed in the ascending loop of Henle
Thick ascending limb
movement back into interstitium
Loop diuretics act here to directly cause hypokalemia
is K+ reabsorbed in the distal nephron?
Intercalated cells can reabsorb some K+ in exchange for H+
When we need to conserve K+, the processes in the distal nephron shuts down K+ secretion by principal cells, leading net reabsorption by intercalated cells
Three main factors influence K+ secretion by principal cells in the distal nephron: 1. Aldosterone status
Effect of aldosterone
Effect of plasma K+ concentration
secretion.
Effect of tubular flow rate on K+
Does water status affect K+ homeostasis?
● Less ADH: decreased collecting duct permeability to ions
● More ADH: increased collecting duct permeability to ions.
Effect of high Na+ diet on K+ balance
in principal cells.
increasing K+ secretion.
Summary of potassium homeostasis
reabsorption and especially secretion.
○ Aldosterone

Summary of Renal Physiology 2
pressure because water will follow Na+ in the body.
systems to normalise Na+ and these work largely by activating thirst or changing
renal handling of Na+ via RAAS or ANP.
are able to depolarise and repolarise.
consequences and so K+ concentrations in the ECF are closely monitored and maintained at low concentrations by modulating the renal excretion rate of K+.
Week 13: Acid Base Physiology
Reintroduction - Acids and Bases
pH goes up or down quite quickly.
concentration of H+ in the ECF to ensure constant pH.
Video 1: reintroduction to Acids and Bases
● Acid: substance that is capable of releasing a proton.
○ hydrogen/proton DONOR
○ HCL->H++Cl-
● Base: substance that can accept/take a proton
Strong vs Weak Acids and Bases
○ HClH++Cl-
○ H2CO3 H+ + HCO3 - Equilibrium Constant (Ka)
○ Counter-intuitive with pH change
●
Control of pH in the body
○ Within cells pH is a little more acidic (~7.2) due to production of H+.
● Control of [H+] or pH in the body is a result of the balance between:
■ AND
Acid production in the body
○
Processes that decrease plasma [H+]
● Three levels of defence: 1. Buffer Systems
○ Works incredibly quick (fraction of a second) 2. Respiratory Control
○ Eg. removal of volatile acids by breathing out
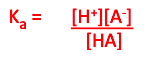
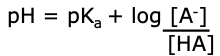
![]()
○ Works in minutes 3. Renal Compensation
Buffer Systems in Acid-Base Homeostasis
forming a new molecule.
back into solution if the amount of H+ starts to decrease.
of H+ and the buffer in the solution.
systems (respiratory or kidney) can kick in and remove the excess H+. Video 2: A Theoretical Experiment - Buffering H+
The bicarbonate buffer system is the most important buffer in the body
bicarbonate in the plasma and interstitial fluid) to momentarily form carbonic acid that then quickly breaks down into water and CO2 according to:
○
physiology:
○
partial pressure of CO2 in the expressed air.
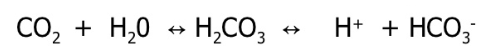
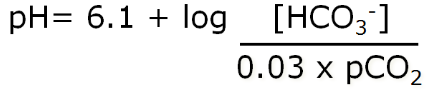
● We have physiological control over the pCO2 (lungs) and the HCO3- (kidneys).
The Respiratory System in Acid-Base Homeostasis
regulating the blood levels of carbonic acid (H2CO3).
H2CO3 in the blood are in equilibrium.
additional H2CO3, which dissociates further into H+ and HCO3- thus lowering blood
pH.
how much CO2 is removed from the system
●
activates the respiratory centre.
kicks in and the equation favours the right hand side more.
because of the need for O2 supply.
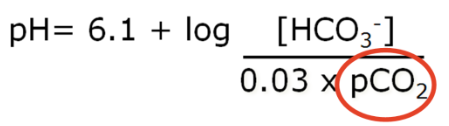

H+ being released and thus form more CO2 and H2O.
thus preventing a pH change.

●
started.
and we would not be able to regulate pH.
The Renal System in Acid-Base Homeostasis
they act quickly.
concentration of HCO3- and H+ in the body.
body.
doing so, they regulate the ratio of HCO3- to CO2 and thus pH:
●
course of a day, ~4320 mmoles of HCO3- is filtered.
take long to deplete our body HCO3- levels.
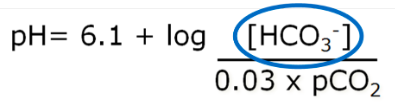
● The nephron can increase or decrease reabsorption of HCO3- to suit the body’s needs by changing what is happening in the PCT, DCT and collecting ducts.
VIDEO 3: Regulation of HCO3- and H+ in the Kidneys
○

H+ formed in the process

○ ● How
○ The answer is to bind the H+ (buffer it). ● Urinary Buffers and H+ excretion
much H+ can you excrete in urine?
used up.
much H+ can you excrete in urine?
● How
production.
○ Therefore, the 70 moles of H+ made in the process must be
secreted/excreted (because we make HCO3- by secreting H+).
○ ○
Breakdown of amino acids (protein metabolism) produces NH4+ Ammonia is toxic and converted to either urea which is excreted by the kidneys or glutamine which is converted back to NH4+ in PCT.
on a Na+ - H+ antiport carrier.
capillary blood via a symport carrier.
achieve this maximum.
○ 1. Reabsorption of filtered HCO3- (PCT_
■ Once HCO3- is fully reabsorbed, any further tubular H+ secretion
results in:
○ 2. Synthesis of new HCO3- (DCT/CD). Secreted H+ binds to urinary buffers.
■ Once urinary buffers are used up:
○ 3. Further H+ secretion and HCO3- generation occurs via ammonia (NH4+)
secretion/excretion (PCT). ● 2nd take home message:
○ 2. The amount of NH4+ excreted ○-
○ 3. The amount of HCO3- lost in the urine.
Acid Base Pathophysiology and Compensation to maintain pH Definitions:
- glutamine handling
The 4 pathological processes that can disturb pH:
1. Metabolic Acidosis:
buffering of H+.
●
excretion ammonia.


●
2. Metabolic Alkalosis
of HCO3- (antacids/renal problems).
■ ECF volume depletion
■ Hypokalaemia
■ Hyperaldosteronism (RAAS)
● Case: Excessive Vomiting
dehydration.
○


○
increased H+ secretion.
●
■ But limited by need to maintain oxygenation ● Renal correction:
■ But mechanism doesn’t work if volume depleted, decreased K+, aldosterone, etc.
3. Respiratory Acidosis
● The issue with respiratory acidosis is always a gain of CO2 in the body.

4. Respiratory Alkalosis
due to hyperventilation from a panic attack, stress/anxiety or certain drug use.
the alkalosis.
Summary
● Acid base disturbances are a major challenge in the body and manifest as a change
in the pCO2 to HCO3- ratio.
●
