Chapter 5: Chemical Bonds and Compounds
Lewis Symbols and the Octet Rule
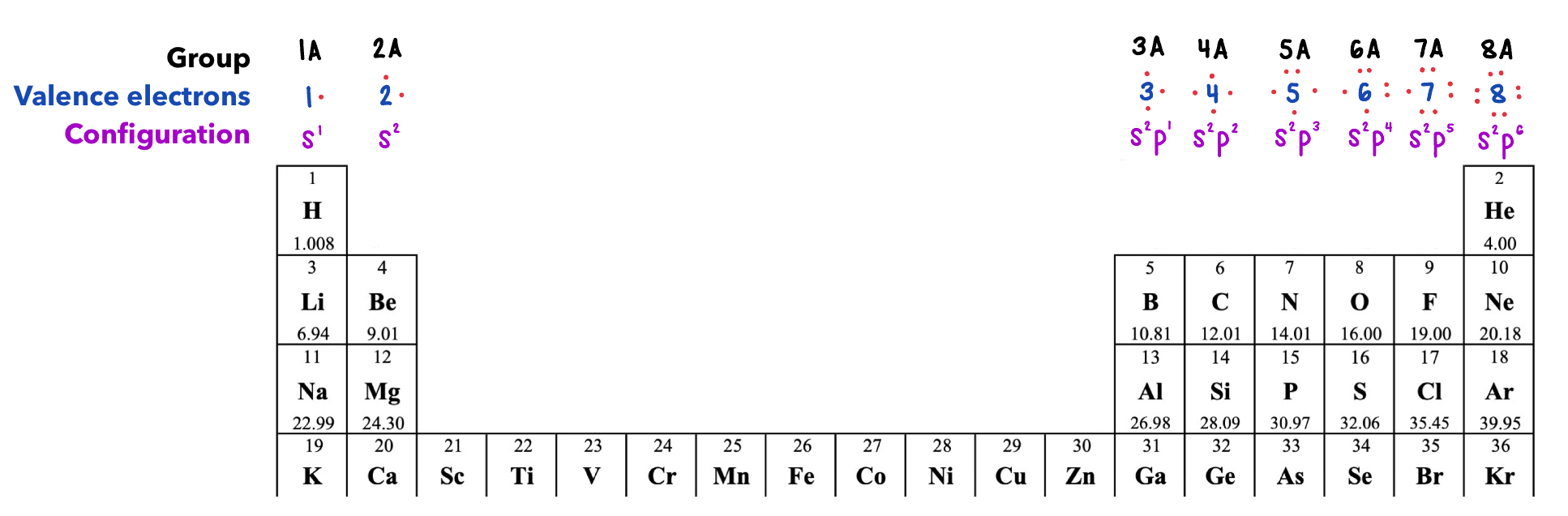
Valence electrons
Valence electron - electrons in the highest occupied energy level of an atom
n level
The valence level involves only s and p sub-levels
Up to 8 electrons can occupy the valence level.
Lewis Symbols
Lewis dot symbols - Represent valence electrons as dots around atomic symbol.
Rules:
If you have 1-4 valence electrons, there is a single dot on each side.
If you have 5-8 valence electrons, start pairing up electrons.
8A has a filled valence electron shell.
The Octet Rule
Octet Rule - An atom is stabilized by having its valence energy level filled.
Noble gases fulfill the octet rule (8A).
Other atoms fulfill the octet rule by:
Gaining or losing electrons.
Sharing electrons.
Some atoms do not obey the octet rule when forming compounds.
Helium has a max of 2 electrons.
Ions
Ions - Atoms or groups of atoms that have an overall charge.
Charges are indicated by using a superscript to the right of the ion.
Monatomic = 1 atom
Cations
Cations - Positively charged ions.
Can have variable charges.
Main group metals fulfill the octet rule by forming cations.
Loses an electron.
Fixed charges to know: Ag2+, Cd2+, Zn2+
Cations | Charge |
|---|---|
Group 1A | 1+ |
Group 2A | 2+ |
Group 3A | 3+ (only for Aluminum) |
Transition metals form cations
Typical charges are 1+, 2+, 3+, or 4+
Some metals form multiple charged ions.
p-block metals also do this.
Naming Cations
Metal cations have the same name as the neutral metal.
Metals with variable charges use Roman numbers to indicate the charge.
Examples:
Ag+ = Silver
Pb2+ = Lead (II)
Pb4+ = Lead (IV)
Anions
Most non-metals fulfill the octet rule by gaining electrons, forming anions.
Anions have negative charges.
Anions have fixed charges and NEVER have Roman numerals.
Anions | Charge |
|---|---|
Group 5A | 3- |
Group 6A | 2- |
Group 7A | 1- |
Naming Anions
Change ending to -ide
Nonmetals = ide
Atom | Anion Symbol | Anion Name |
|---|---|---|
Chlorine | Cl- | Chloride |
Oxygen | O2- | Oxide |
Sulfur | S2- | Sulfide |
Nitrogen | N3- | Nitride |
Polyatomic Ions
Polyatomic Ions - Groups of atom with a charge.
Formula and Charge | Name |
|---|---|
Ammonium | |
Nitrate | |
Nitrite | |
Carbonate | |
Bicarbonate (Hydrogen carbonate) | |
Phosphate | |
Hydrogen phosphate | |
Acetate | |
Hydroxide | |
Cyanide | |
Peroxide | |
Sulfate | |
Sulfite | |
Bisulfate (Hydrogen sulfate) | |
Perchlorate | |
Chlorate | |
Chlorite | |
Hypochlorite | |
Chromate | |
Dichromate | |
Permanganate |
Oxyanions
Oxyanions - Contain Oxygen
Usually end with -ate
More than one oxyanion:
More oxygen atoms -ate
Fewer oxygen atoms -ite
Ionic Bonds and Compounds
Polyatomic anion = metal cation
Polyatomic cation = nonmetal anion
Ionic bond - An attraction between oppositely charged ions.
Ionic compound - Composed of charged ions.
Metal cations and nonmetal anions form ionic compounds
Oppositely charged ions
Don’t form molecules
Polyatomic cation and polyatomic anion
There is a total charge of 0 in ionic compounds.
Compound Notation
No charges in the final compound formula.
Ions are always listed with the cation first and the anion second.
Cation (Anion)
Example: CrO
Subscripts show how many of each atom.
Subscripts are placed after the atom that they are used for.
Parenthesis are used when there are multiple of the same polyatomic ion.
Ionic Compound Structure
Ionic lattice - an array of positive and negative ions.
Whenever you have a negative, a positive will always be next to it.
Chemical Formulas
Empirical formula - the smallest whole number ratio of atoms (always used for ionic compounds).
Formula unit - the smallest number of ions necessary to form a compound.
If you can simplify all subscripts, you have to do so.
Subscripts for polyatomic ions do NOT change.
Only use parenthesis if there is more than 1 polyatomic ion.
Naming Ionic Compounds: Fixed Cation Charges Examples
NaCl = Sodium chloride
Chlorine has 1 element, so it ends in -ide
MgCl2 = Magnesium chloride
Chlorine has 1 element, so it ends in -ide
MgSO4 = Magnesium sulfate
SO4 has 2 elements, so it ends in -ate
Naming Ionic Compounds: Variable Charge Metals
The charge can change.
Use the anion to figure out the charge on the cation.
Covalent Bonding
Covalent bond - valence electrons shared between two nonmetal atoms.
Goal is to have a filled valence shell.
Sharing electrons can result in filled valence shell.
Diatomic Molecules
Diatomic molecules can NEVER EXIST ALONE.
They must be paired with an identical element or something else.
7 elements that form diatomic molecules:
H2
N2
O2
F2
Cl2
Br2
I2
Double and Triple Bonds
Double covalent bond - 2 shared pairs of electrons
Triple covalent bond - 3 shared pairs of electrons
Covalent Compounds
Covalent compounds fulfill the octet rule by sharing electrons.
Between nonmetal atoms
Usually form molecules
Molecular formula (do not simplify)
Bonding electrons - electrons in bonds
Nonbonding electrons - unshared electrons that are not in bonds, aka lone pairs.
Binary Covalent Compounds
Binary covalent compounds - 2 different nonmetals
Prefixes indicate the number of atoms present.
The leftmost element on the periodic table is named first.
The second element is an anion (-ide).
Atoms | Prefix |
|---|---|
1 | mono- |
2 | di- |
3 | tri- |
4 | tetra- |
5 | penta- |
6 | hexa- |
7 | hepta- |
8 | octa- |
9 | nona- |
10 | deca- |
Aqueous Solutions
Aqueous solution - a homogenous mixture, in which the main component is water.
Soluble - able to dissolve in water.
Insoluble - unable to dissolve in water.
Electrolyte Solutions
Electrolyte solutions conduct electricity because of ions in water.
Dissociation
Dissociation - ions are pulled apart in an aqueous solution.
Covalent Molecules
Covalent molecules - do not dissociate into ions.